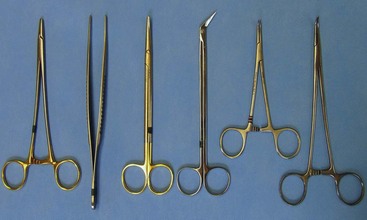
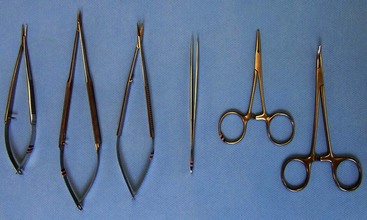
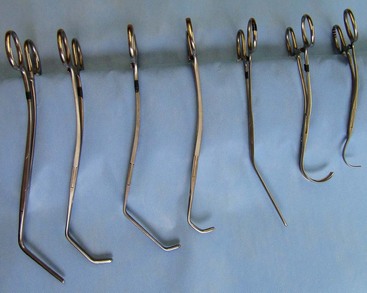
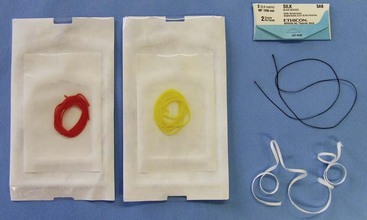
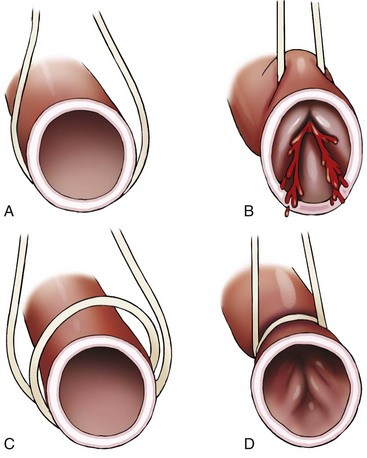
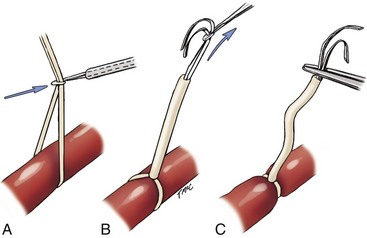
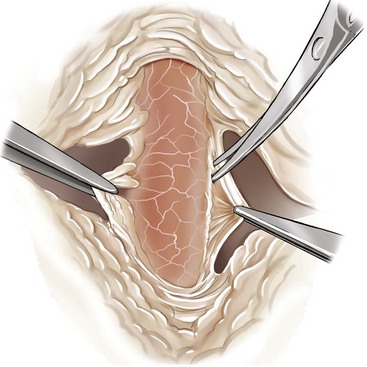
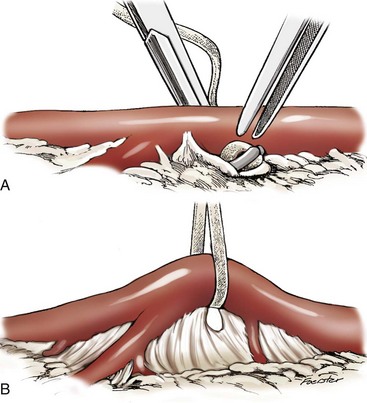
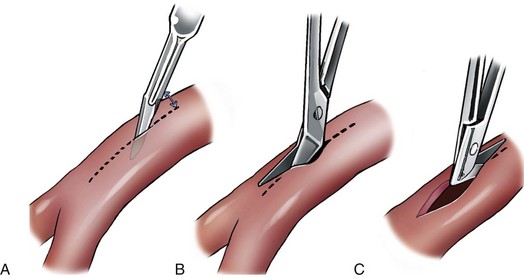
Chapter 108 Historically, permanent vascular interruption by ligation was the principal vascular surgical technique used by human and veterinary surgeons. In 1762, Richard Lambert described an arterial suturing technique using a wire or thread twisted about a pin for the treatment of a brachial arterial aneurysm associated with a bloodletting procedure.36,37 This technique, known as the Lambert-Hallowell procedure, was the first described vascular reconstructive effort. By 1882, Schede reported on the successful repair of a peripheral vein. Widespread success with vascular reconstruction, however, was not realized until development of more refined vascular suturing techniques decades later.37 The origins of successful vascular surgery can be traced to pioneering efforts of Dr. Alexis Carrel in the early 1900s. Although Kummell had performed the first end-to-end anastomosis of an artery in a person in 1889, Carrel described in 1902 a triangulation technique for more accurate end-to-end vascular anastomosis, and he advocated end-to-side vascular anastomosis for vessels having significant disparity of luminal size. Classic studies of Carrel and Dr. Charles Guthrie culminated in successful replantation of severed appendages, and microvascular surgical techniques were subsequently introduced after the first use of an operating microscope in 1921 by Dr. Carl Nylen for an otic surgery.29,38 Medical and surgical advances, including those in transfusion medicine, antithrombotic therapy, infection control and prevention, and surgical materials and instrumentation, have greatly and rapidly advanced the field of vascular surgery. The birth and advancement of endovascular surgery, with the development of minimally invasive catheterization-related procedures and prosthetic vascular grafts, stents, and thrombotic coils, have allowed for the successful treatment of previously inoperable conditions and decreases in morbidity and mortality associated with many risk-laden surgical procedures.46 With careful instruction and practice, the veterinary surgeon can apply the basic principles of vascular, microvascular, and endovascular surgery to the treatment of numerous conditions of small animal patients. Although a comprehensive discussion of the entire vascular anatomy of small animal patients is beyond the scope of this chapter, knowledge of the basic anatomy of the vascular trees and of an individual artery and vein is critical for successful vascular surgeons. An artery is composed of three layers called tunics. The outermost layer of an individual artery is the tunica externa or tunica adventitia. It is composed of connective tissue fibroblasts and collagen and should be dissected from the cut end of vessels involved in reconstruction to avoid inadvertent infolding of the adventitia into the lumen of the anastomosis. Deep to the adventitial layer is the tunica media, which is composed of smooth muscle cells and elastic tissue. The innermost layer of an artery is termed the tunica intima and is composed chiefly of endothelial cells.4 The intima is quite fragile and must be handled carefully by the surgeon to avoid intimal damage, which may lead to activation of the clotting cascade and thrombosis.15 Arteries are found in all locations in the body, excluding the hair, epidermis, nails, cartilage, and cornea. The arterial tree originates as the aorta at the base of the left ventricle of the heart. The smallest arterial branches perfuse the tissues of the trunk, enclosed organs, and the periphery of the body and its covering of dermis.19 For dissection purposes, it is important to note that the branch of an artery is smaller than the larger parent artery from which it arises. However, if one artery branches into two arteries concurrently, as in the terminal aorta at the level of the iliacs, the combined area of the branching vessels is generally greater than that of the parent vessel. Additionally, when dissecting an arterial vessel, one should note that its course is generally linear and straight, although arteries in highly mobile areas or those perfusing tissues prone to engorgement may be tortuous. Finally, knowledge of the presence of collateral circulation from anastomoses or communications between branches of the arterial tree is of great importance to vascular surgeons because it may allow sacrifice of some vessels.19 Veins are broadly categorized as systemic and pulmonary. Unlike systemic veins, pulmonary veins and the umbilical vein carry oxygenated blood as they traverse from the lungs to the heart or from the dam to the fetus, respectively. The portal vein is a specialized systemic vein that returns blood from the portal venous system of the abdomen to the liver. Peripheral systemic veins are divided into superficial and deep veins and venous sinuses. Superficial veins are found in subcutaneous tissues and return blood from these areas to the deep veins. For dissection consideration, note that deep veins generally accompany arteries as they course through the body and are frequently contained within the same surrounding perivascular sheath. Smaller arteries may have one or two veins in accompaniment, but larger arteries generally only have one accompanying vein.20 Venous sinuses are contained only within the skull and are therefore not discussed in detail in this chapter. Veins are generally more thin walled and collapsible than arteries and are also composed of three main layers, or tunics. The outermost layer is the tunica externa, or adventitia, which is composed of connective tissue fibroblasts and collagen. The tunica media of a vein is thinner than that of an artery and, although composed chiefly of smooth muscle cells, generally does not act to function in a contractile manner. The tunica intima is the innermost layer of the vein and is composed of endothelial cells, as in the artery. Most veins, although not contractile, contain infoldings of the tunica intima. These valves prevent backflow and pooling of blood from the effects of gravity.4 Venous dissection and reconstruction should proceed with caution, because the venous wall is thin and its intima also readily damaged with overly aggressive or careless handling.15 As with arteries, when performing venous anastomoses, the surgeon should be certain to dissect the venous adventitia from the distal portion of the vein to avoid inadvertent inclusion of the adventitia in the lumen of the anastomosis. Instruments, Suture, and Graft Materials Along with basic instrumentation required for soft tissue procedures, general vascular surgeons require only a small number of additional instruments, including a fine-pointed diamond-jawed needle holder, vascular (e.g., DeBakey) forceps, scissors for dissecting and opening vessels, atraumatic vascular clamps, and a right-angled forceps such as a mixter (Figure 108-1). A blunt-tipped mixter and Metzenbaum scissors are invaluable for dissecting the perivascular tissues around vessels. However, when cutting into a vessel or extending an incision made in a vessel with a scalpel blade, delicately pointed tips of Potts scissors, which come in straight, curved, or angled varieties, are ideal.15,38 Microvascular surgery requires more specialized instrumentation. Microvascular needle holders and curved and straight scissors, such as the Castroviejo and Vannas varieties, are ideal for maintaining a fine and delicate grip on small needles, incising and transecting small vessels and fine suture, and dissecting adventitia. Jeweler’s forceps have very fine, pointed tips that are well suited for accurate and delicate grasping of fine vascular and perivascular tissues. Sharp-sharp iris scissors and a variety of straight and curved mosquito hemostatic forceps can also aid in microvascular procedures (Figure 108-2). Additionally, an operating microscope offering 10× magnification is mandatory for many delicate microvascular procedures, such as anastomoses of small vessels. General vascular surgical procedures may also benefit from the addition of magnification. Surgical loupes providing magnification from 2.5× to 4.5× are readily available. Whereas standard, lighted headsets can adjust to fit a variety of surgeons, individual loupes can be customized to suit a particular surgeon’s ocular needs.37–39 Atraumatic vascular clamps in a variety of sizes and angles are also required for general and microvascular surgery (Figure 108-3). Discrepancies or differences in the depth of the surgical wound, exposure of the vessel to be dissected, angle of clamp application, and size of the operated vessel can be accommodated by choosing an appropriately sized and configured clamp. The Satinsky clamp has handles for ease of vascular manipulations during surgery and interdigitating, atraumatic, serrated teeth that exert a firm grip without slipping or crushing. It comes in a variety of sizes and is one of the most versatile clamps for peripheral vascular applications.37,38 Castaneda atraumatic neonatal clamps and Cooley atraumatic pediatric anastomosis clamps also have handles and fine teeth that are offset and interdigitating, allowing them to occlude the vessel wall without crushing the tissue.9 If exposure is limited or the transected end of a vessel needs to be manipulated, a standard bulldog clamp can be used. These clamps do not have handles, and the jaws are held in apposition by a springlike mechanism.38 Other clamps, such as the arteriotomy clamp, are held in apposition by manual application of force. The arteriotomy clamp is used to grasp an area of artery for excision in preparation for end-to-side vascular anastomosis (Figure 108-4). Figure 108-4 Arteriotomy clamp. The open tip allows for accurate assessment of the depth of the arteriotomy (inset). In addition to clamps, loops of Silastic, heavy-gauge suture, rubber catheters, and moistened umbilical tapes can be used to facilitate manipulation, exposure, and dissection of vessels (Figure 108-5). These materials can be fed around a vessel after an opening has been created in the perivascular connective tissues.37,38 Loops may also be loosely applied for retraction of vital structures such as nerves and vessels or doubly applied, as in a Potts loop, to provide secure and firm tamponade of the vessel with traction on the loop ends (Figure 108-6). Furthermore, these materials may be utilized to create a Rumel tourniquet. To make a Rumel tourniquet, the suture, tape, or Silastic is moistened; placed around the vessel; and passed through a short length of plastic catheter or extension tubing with the aid of a Rumel instrument (Figure 108-7). Occlusion of the vessel then occurs by sliding the plastic tubing toward the vessel and securing its position with a clamp applied to the remaining ends of the suture, tape, or Silastic. The occlusion is readily relieved by clamp removal and movement of the plastic tubing away from the vessel.37 The ideal suture for vascular surgery should have excellent knot-holding capabilities and ease of handling and stimulate minimal inflammatory reaction. Vascular suture should be swaged onto a ½ or Prosthetic vascular grafts and patches can be categorized as biologic or synthetic. Biologic grafts include those derived from native tissues such as jugular veins and other expendable vessels, of which there are few in small animal patients. Synthetic grafts can be further classified into textile and nontextile varieties. Textile grafts, such as knitted Dacron, were used commonly in the past but have been supplanted largely by the nontextile grafts because of problems associated with thrombogenicity and infectivity.39 Nontextile grafts are predominantly made from expanded polytetrafluoroethylene, a linear, thermoplastic polymer of carbon, oxygen, and fluorine. They are not knitted or woven but are made by an extrusion process into porous tubes or sheets.14,39 PTFE grafts are strong, conformable, and easy to handle; can be cut to various sizes and configurations; and may be customized based on the needs of the patient. These newer synthetic grafts are biologically inert and relatively thromboresistant and hydrophobic by virtue of an electronegative surface potential; however, thrombogenicity and infectivity of synthetic grafts remain relatively high compared with autologous tissues.5 After incising overlying skin, subcutaneous fat, and fascia or entering an associated cavity, hand-held or self-retaining retractors may be helpful in exposing the vessel of interest. Blood vessels are often contained within a perivascular sheath or are surrounded by a variable amount of adipose tissue and may not be readily discerned. After a vessel of interest has been identified, the surgeon should dissect as closely to the adventitia as possible, generally with a blunt-tipped, right-angled forceps or blunt-tipped fine Metzenbaum scissors. By initially dissecting directly over the surface of a vessel closest to the skin, the surgeon can generally avoid inadvertent disruption of branching collaterals. The adventitial surface of the artery is marked by a characteristic vaso vasorum pattern of perfusing vessels (Figure 108-8). After this plane has been located, the surgeon should readily identify the “white line” marking the proper plane of dissection. This adventitial white line is created when dissection and retraction adjacent to the vessel allow air to penetrate the areolar tissues interposed between adventitia and surrounding tissues (see Figure 108-8). Veins are generally recognizable by their white and bluish color and a ballotable sensation noted on direct finger pressure over the vessel. Dissection of a vascular segment can then be facilitated by passage of suture, tape, or Silastic under the vessel and, with application of traction and countertraction, restricting points of attachment may be released (Figure 108-9). Any intervening collateral branches are identified and ligated, cauterized, or preserved.37,38 Once the desired length of artery or vein has been dissected and exposed, the vessel must be controlled before any vascular interventions can proceed. Major inflow and outflow vessels to and from the selected segment should be isolated and substantial branches identified. Atraumatic devices for occlusion, such as vascular clamps; Potts loops or single loops of suture, tape or Silastic; or Rumel tourniquets may be applied. Clamps are usually chosen to occlude supplying vessels because of greater security and more reliable and stable occlusion; however, loops can be applied in a variety of configurations and, when doubly looped to incorporate a branching vessel, are quite secure. Additionally, Codman and Cooley clamps or curved Satinsky clamps can be applied parallel to the course of the vessel, resulting in partial or complete occlusion of the vessel to permit arteriotomy or venotomy within the confines of the clamp jaws.37 Most vascular procedures require interruption of blood flow for a time, and only the most brief and simplest of procedures can be performed without occlusion of blood flow.38 Therefore, the converse but interconnected processes of hemostasis and anticoagulation must be given due consideration in most vascular interventions. Careful and accurate hemostasis is one of Halsted’s tenets of surgery, and a surgeon exhibiting appropriate craftsmanship is generally able to limit blood loss by attention to detail and careful dissection.44 A variety of hemostatic tools are available to vascular surgeons to permanently interrupt blood flow, including Kelly, Crile, and mosquito hemostatic forceps; clips; sutures; staples; monopolar and bipolar cautery devices; and hemostatic sponges, gels, powders, textiles, and waxes. Temporary occlusion of blood flow can be accomplished with judicious use of atraumatic vascular clamps, sutures, tapes, polymeric silicone tubing, and specialty tourniquets as described above.37,39 When vessels are not simply ligated but explored, replaced, or reconstructed, the need for anticoagulation measures must be assessed. It has been the authors’ experience that in small animal patients, use of local anticoagulants is preferred over systemic administration. Many small animal patients in need of vascular reconstructive procedures present with concurrent disease processes that may adversely affect the coagulation cascade or platelet function, such as diabetes and other endocrine diseases, hepatic disease, uremia, trauma, sepsis, and others.26 One must carefully weigh the risk of systemic administration of anticoagulant compounds to the patient presented. Additionally, many anticoagulant medications commonly used in human patients, such as aspirin, present dangers other than hemorrhage to small animal patients, including hepatic, gastrointestinal, and renal compromise.24 To prevent clot formation in vessels undergoing simple incision or arterial or venous anastomosis, the authors have used with great success an ice-cold solution of 2% lidocaine, heparin, and 0.9% saline to thoroughly flush the isolated vessel segment via the arteriotomy or venotomy site. Accumulated thrombus may be removed with forceps; additionally, proximal inflow occlusion may be temporarily released to allow escape of clot thrombi and air emboli from the incision just before placement of the final sutures.38 Incorporating another of Halsted’s principles—gentle tissue handling—when dissecting and suturing the vessel wall prevents intimal damage that could lead to activation of the clotting cascade.15,26,44 Finally, when reconstructing small vessels, it is critical to dissect the adventitia from the ends of the cut vessels to prevent inadvertent inclusion of the adventitia in the lumen of the anastomosis, which could reduce the luminal diameter and obstruct blood flow, possibly leading to thrombus formation.39 Although systemic anticoagulative measures are less commonly used in veterinary vascular surgery than in humans, their use is justified in several instances, including cardiac surgery requiring bypass, adrenalectomy for adrenocortical tumors, placement of vascular autografts and prosthetics, and canine renal transplantation.* A technique for cardiopulmonary bypass in dogs has been described wherein patients received 300 U/kg of unfractionated heparin intravenously before cannulation to achieve an activated clotting time of more than 480 seconds (reference range; 60 to 120 sec).32 For adrenalectomy, a protocol to decrease the potential for thromboembolism has been described.28 During surgery, 35 U/kg of unfractionated heparin is given in plasma. Dosing continues for 3 to 4 days after surgery, starting at 35 U heparin/kg SC q12hr and tapering to 10 U/kg SC q12hr. The day after heparin administration has ceased, hetastarch is administered at 10 to 20 mL/kg over 6 hr. For vascular autograft placement, a single dose of unfractionated heparin (30 to 100 U/kg IV) may be given before or during surgery and may be continued postoperatively in some patients.9,25,33 One author utilized enoxaparin, a low-molecular-weight heparin (1.0 mg/kg SC q6hr), for 1 month after surgery to reduce the risk of venograft thrombosis.25 Use of unfractionated heparin (75 U/kg SC q6–12hr) for up to 5 days has also been reported.25,33 LMWH compounds such as enoxaparin have been used by the authors without ill effects; the drug (0.75 mg/kg SC q24hr) is given the day before the vascular procedure and continued for 7 days after surgery.23,34 Additionally, with increased use of erythropoietin analogues in cats with chronic renal failure, the authors have treated associated patients with thrombocytoses with low-dose aspirin therapy (20.25 mg/cat PO given once before vascular surgery). Permanent vascular interruption by ligation or use of another hemostatic device is the simplest of all vascular procedures. For veins and smaller arteries, single or double ligation using silk or absorbable suture or single ligation with the addition of a distal hemostatic clip should suffice. With the advent of vascular clips for stapling devices, which offer secure closure of large arterial vessels supplying organs such as lung or liver lobes, major arteries are rarely ligated and divided manually in small animal patients. Some vascular conditions, however, such as patent ductus arteriosus, present surgeons with little room for stapling devices or for clamp placement, vessel division, and end-on closure of vascular stumps. In these situations, a ligature placed at each end of the intervening vessel with a transfixation suture placed between obliterates the vessel lumen, decreases the risk of recanalization, and permits safe ligation in continuity (Figure 108-10). When ligating any vessel prone to recanalization or in situations when recanalization would be detrimental, a single transfixation suture that penetrates the lumen should be placed between two circumferential ligatures.37 Simple incision into a vessel may be used to introduce catheters for interventional procedures or cannulas for bypass procedures, to remove thrombi, or for treatment of specific vascular conditions.38 Vessel incisions may be longitudinal or transverse. Longitudinal incisions are useful in that they are readily extended to increase exposure, the incised parallel walls of the vessel are able to be retracted to increase exposure, and they can easily be manipulated to receive another vessel in an end-to-side anastomosis if a more aggressive approach is required. The vessel lumen, however, may be narrowed upon suture closure; even a minor amount of vessel narrowing, together with intimal exposure and the presence of foreign suture material in the vessel lumen, may be sufficient to elicit thrombus formation in smaller vessels. Therefore, in vessels smaller than 4 mm in diameter, a transverse incision is recommended.38,39 After control of blood flow to the area has been adequately obtained, a simple arteriotomy or venotomy should be initiated by incision through all layers of the vessel wall with a #11 blade placed at a 45-degree angle to the surface of the vessel. The surgeon should take care not to incise the opposing wall of the vessel. The incision may be extended with microvascular or angled Potts scissors as needed (Figure 108-11). A transverse incision commences similarly but should not extend beyond 180 degrees of the vessel circumference.37 Upon closure, one must be certain to excise excessive adventitial tissue from the ends of the incision to prevent its inadvertent inclusion in the closure and subsequent luminal compromise. Kunlin’s technique can also be used to produce a desired eversion of the tissue edges by starting the pattern with a horizontal mattress suture in each corner and gently grasping and everting the tissue edges with forceps during suture placement.38 Additionally, the surgeon should be certain to include all layers of the incised vessel wall, especially the intima, when placing sutures for closure but avoid inadvertent snaring of the opposing vessel wall. Sutures are generally placed 1 mm apart and 1 mm from the cut edge of the vessel except when vessels are enlarged, thickened, or diseased.39 Simple interrupted patterns and continuous patterns of suture placement have been described.37,38 Continuous suture patterns in human vascular surgery frequently commence at either end of the incision and proceed to the middle of the incision, where the ends are tied (Figure 108-12). The continuous suture line must be pulled and held taut throughout placement to avoid hemorrhage in areas that are slack. After flow has been restored, any suture line bleeding can usually be controlled with mild tamponade by finger pressure and patience. Occasionally, placement of a fine-gauge single simple interrupted suture may be required, or in cases of more serious bleeding, blood flow may need to be reoccluded to properly identify and address any leaks.37,38
Vascular Surgery
Anatomy
General Vascular and Microvascular Surgery
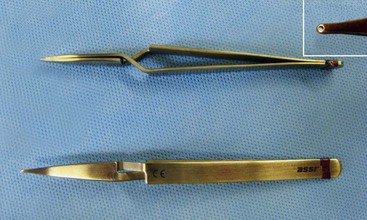
 circular, round, taper-point needle end. Caliber of the needle and suture should be as small as possible to minimize hemorrhage through suture holes.38,39 As a general reference, a range of 3-0 to 8-0 suture is typically used, progressing from larger to smaller vessels.37 For example, 3-0 suture might be chosen for a mitral valve annuloplasty in a small-breed dog, and 7-0 to 8-0 suture is typically chosen for vascular anastomosis in feline renal transplant procedures. Generally, vessels with an internal diameter of 4 to 6 mm can be sutured with 4-0 to 5-0 suture, vessels of 2 to 3 mm with 6-0 suture, and vessels of less than 2 mm with 7-0 to 8-0 suture.39 Although monofilament suture such as nylon or polypropylene is preferred for arterial work because of greater strength and durability and decreased tissue reactivity, braided multifilament sutures have excellent knot-holding capabilities and, when passed through mineral oil or bone wax, have improved ease of handling and exhibit less drag.38 These soft multifilament sutures are commonly used in the more delicate venous tissues.
circular, round, taper-point needle end. Caliber of the needle and suture should be as small as possible to minimize hemorrhage through suture holes.38,39 As a general reference, a range of 3-0 to 8-0 suture is typically used, progressing from larger to smaller vessels.37 For example, 3-0 suture might be chosen for a mitral valve annuloplasty in a small-breed dog, and 7-0 to 8-0 suture is typically chosen for vascular anastomosis in feline renal transplant procedures. Generally, vessels with an internal diameter of 4 to 6 mm can be sutured with 4-0 to 5-0 suture, vessels of 2 to 3 mm with 6-0 suture, and vessels of less than 2 mm with 7-0 to 8-0 suture.39 Although monofilament suture such as nylon or polypropylene is preferred for arterial work because of greater strength and durability and decreased tissue reactivity, braided multifilament sutures have excellent knot-holding capabilities and, when passed through mineral oil or bone wax, have improved ease of handling and exhibit less drag.38 These soft multifilament sutures are commonly used in the more delicate venous tissues.
Vascular Exposure and Control
Hemostasis and Anticoagulation
Vascular Incisions and Closures
< div class='tao-gold-member'>
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Vascular Surgery
Only gold members can continue reading. Log In or Register to continue