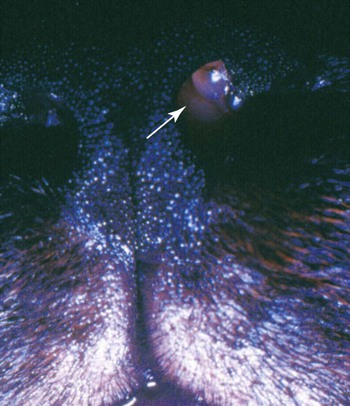
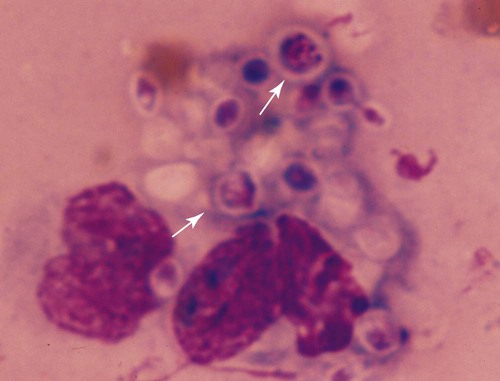
Twenty-five species of Trichosporon have been identified either in soil or on plants in the environment, as commensals of the skin or mucosae of the gastrointestinal or respiratory tract, or as disease-producing organisms. Based on classical microbiologic methods, causative agents of disease in people were identified as Trichosporon beigelii; however, genomic analysis has indicated that these isolates comprise at least eight species (Trichosporon asahii [= T. beigelii and some previously reported as Trichosporon cutaneum], Trichosporon asteroides, Trichosporon cutaneum, Trichosporon inkin, Trichosporon mucoides, Trichosporon ovoides, Trichosporon pullulans, and Trichosporon loubieri).27,47a As for isolates from domestic animals, T. pullulans, T. asahii, Trichosporon domesticum, Trichosporon loubieri, or unspecified Trichosporon spp. have caused infections in reported cases in cats. T. cutaneum was isolated from a dog with skin problems.22 Using polymerase chain reaction amplification, Trichosporon spp. DNA was amplified from the duodenal contents of dogs without and with chronic enteropathies, at a greater frequency in clinically ill dogs.40 T. asahii was associated with outbreaks of mastitis in dairy herds.11 T. inkin, a skin commensal, is recognized as the agent of white piedra, a nodular mycosis of hair shafts affecting people, monkeys, and horses in temperate to tropical climates. A synergistic coryneform bacterial infection is often present in this condition, suggesting a pathogenic co-infection.37 T. asahii and T. mucoides have been linked to the syndrome of human summer-type hypersensitivity pneumonitis in Japan.29,33 T. asahii has been found in the urine of human patients with indwelling urinary catheters and may be an opportunist in causing urinary tract infections.21 T. mucoides was incriminated as a cause of nosocomial infection in children associated with a faulty bronchoscope.39 T. asahii has been responsible for most systemic infections in immunocompromised people, especially those treated for hematologic malignancies, with human immunodeficiency virus/acquired immunodeficiency syndrome, hemochromatosis, intravascular implants or catheters, end-stage renal failure or hereditary phagocyte deficiencies, or receiving glucocorticoid immunosuppression or renal or bone marrow transplants. Biochemical and genetic studies of isolates from humans indicate that cutaneous, mucosal, and environmental isolates differ from those that cause disseminated illness.19,23,23 Trichosporon spp. are opportunistic pathogens that cause cutaneous and visceral infections in immunocompromised hosts. Most cases of trichosporonosis in people have been disseminated and fatal, especially in patients with severe immunosuppression who were also neutropenic. Many patients had received multiple or broad-spectrum antibacterials for documented or presumed bacterial infections, whereas others had neoplastic diseases or organ transplants. Presumably, the fungus invades mucosal surfaces of the respiratory, gastrointestinal, or urogenital tracts of immunosuppressed hosts, with subsequent dissemination. A few cases of valvular endocarditis caused by Trichosporon spp. have been reported.3,18 Phenotypic switching, a process by which pathogenic species of these yeasts vary their surface characteristics and enzyme production, may be one way they avoid host defense mechanisms.15 One cat was reported to have a fever, an inspiratory stertor, and a protruding unilateral nasal mass (Fig. 64-1) similar to that caused by Cryptococcus neoformans.14 Later spread to regional lymph nodes and aerogenous spread to pulmonary tissues was suspected. In another cat, a large mass in the nasal passage was associated with marked turbinate destruction as visualized by nasal computerized axial tomography.17 A third cat had a chronic ulcerative subcutaneous lesion at the site of a bite wound.7 A fourth cat suffered from chronic hematuria and dysuria as a result of chronic cystitis complicated by the yeast infection.7 A fifth cat in which T. domesticum was isolated also had persistent lower urinary tract signs of hematuria and dysuria.37 In a sixth cat, with a nasal mass caused by T. loubieri, fluconazole therapy for 6 months was successful in achieving therapeutic resolution.38 Mere culture of Trichosporon spp. from cutaneous or mucosal surfaces can be misleading because the organism is a normal constituent of the endogenous microflora in these areas. In cytologic specimens made from impression smears of granulomatous lesions, round to oval encapsulated fungal organisms can be visualized (Fig. 64-2). Organisms may be free or within macrophages and may show pseudohyphal forms or broad-based budding. Trichosporon spp. are less pleomorphic, being spherical to oval, and usually have a thin polysaccharide capsule, in comparison to Cryptococcus spp. Biopsy with histopathologic confirmation of host reaction and invasion of the deeper tissues by characteristic fungal elements is more specific for documentation of pathogenicity. In cats with nasal involvement, radiographic imaging findings may be similar to those for other fungal infections.17 In animals with urinary tract infections, yeasts may be observed microscopically in the urine sediment. In disseminated infections, Trichosporon spp. can be confused with Candida spp. in cytologic and histologic specimens. T. asahii produces a heat-stable cell-wall antigen that is similar to the capsular polysaccharide of C. neoformans. The latex agglutination test, used to detect cryptococcal capsular polysaccharide antigen (see Chapter 59), has been used to diagnose disseminated T. asahii infections in humans and in experimentally infected rabbits.28,45 Pretreatment of sera with pronase, which presumably disrupts immune complexes and nonspecific protein binding to the fungal capsule, increased the sensitivity of the antigen detection test. Changes in the cell-wall conformation that develop with antifungal therapy may make the test results negative despite a continued presence of the organism.27,47a In a cat with nasal infection caused by T. pullulans, cryptococcal antigen test findings of serum were negative before clinical evidence of dissemination or before treatment.14 Biochemical and physiologic features do not consistently distinguish between members of this genus.35,36 Polymerase chain reaction has been used to develop genus-specific primers for detection of Trichosporon spp. in clinical specimens and culture.37,43 Pathogenic Trichosporon spp. can be detected with this method, whereas DNA sequences of other similar pathogenic yeasts such as Malassezia spp., Candida spp., and C. neoformans are not amplified. Genomic analysis is the most accurate means of identifying individual species.6,36,36
Trichosporonosis
Etiology
Pathogenesis
Clinical Findings
Diagnosis
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree