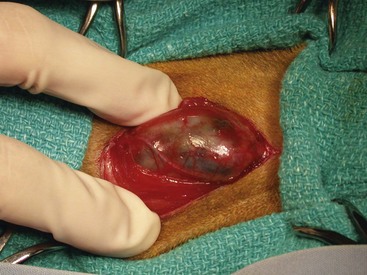
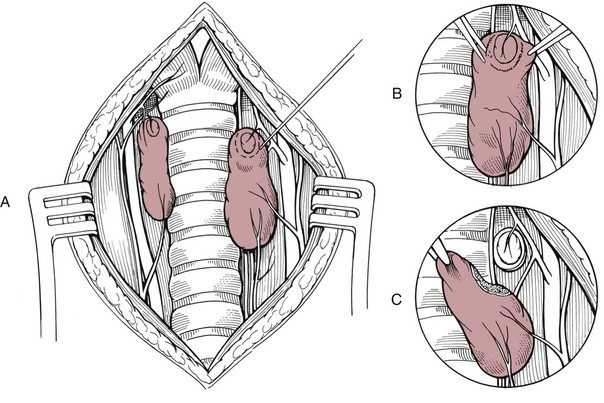
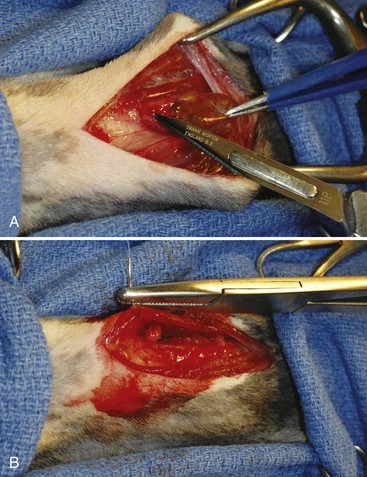
Chapter 121 Dogs and cats have two thyroid glands (Figure 121-1), which are sometimes referred to as lobes.26 The glands are lateral and, at times, somewhat ventral to the trachea, spanning the initial five to eight tracheal rings. In dogs, thyroid gland size varies with breed and individual body size. In an adult medium-size breed, the thyroid gland is approximately 5 cm in length, 1.5 cm in width, and 0.5 cm in thickness. The cranial pole of the right cranial gland is at the level of the caudal border of the cricoid cartilage of the larynx, and the caudal pole extends to the fifth tracheal ring. The left gland is slightly more caudal, extending from the third through the eighth tracheal rings. In some dogs, there is a narrow communication between the two glands. This bridge, composed of glandular parenchyma, called isthmus glandularis, passes across the trachea ventrally. In cats, the thyroid glands are tan and flat and measure 10 mm long, 3 to 5 mm wide, and 1 to 2 mm thick.14,34 To its dorsolateral aspect, the right gland is in close proximity to the common carotid artery, internal jugular vein, vagosympathetic trunk (all components of the carotid sheath), and tracheal duct (lymphatic vessel). The dorsolateral boundary of the left gland is the esophagus.26 Components of the carotid sheath are displaced by the esophagus on the left side; therefore, the left thyroid gland is not in contact with the carotid sheath. Of clinical importance is the presence of the laryngeal recurrent nerves in the cervical area. The laryngeal recurrent nerves are positioned dorsally and tend to be medial to the thyroid glands. Of lesser clinical importance is the presence of the pararecurrent laryngeal and pharyngoesophageal nerves in the cervical area, which can make accurate identification of the recurrent laryngeal nerves more difficult. The vascular supply of the thyroid gland is mostly through cranial and caudal thyroid arteries.26 The cranial thyroid artery is the first major branch of the common carotid artery. It approaches the thyroid from the cranial pole and runs dorsal to the gland to anastomose with the caudal thyroid artery. The caudal thyroid artery typically arises from the brachiocephalic artery and approaches the thyroid gland from the caudal pole. From the vessel formed by anastomosis of thyroid arteries, many branches arise in various numbers and patterns that approach the dorsal and ventral surfaces of the gland. These dorsal and ventral branches bifurcate before entering the gland to supply its lateral and medial surfaces. In most cats, the caudal thyroid artery is absent.16,42 Venous return from the thyroid gland is primarily from cranial and caudal thyroid veins, which exit from respective poles.26 The cranial thyroid vein empties into the internal jugular vein at the level of the caudal margin of the larynx. The caudal thyroid vein empties into the internal jugular vein in the region of the caudal neck. Some dogs have an unpaired vessel near the midline of the trachea that receives a large tributary from the middle segment of the left thyroid gland and proceeds to enter the brachiocephalic vein. Lymphatic drainage is cranially through relatively large lymphatic trunks that end in the cranial deep cervical lymph node.26 There are also lymphatics caudally that drain into the caudal deep cervical lymph nodes. The cranial and caudal deep cervical lymph nodes can be absent in a good proportion of dogs. In these cases, lymph flows directly to the nearest lymph node, such as the medial retropharyngeal lymph node cranially. Lymphatic vessels ultimately drain into the right lymphatic duct on the right side and left tracheal duct on the left side.26 Innervation to the thyroid gland is provided by the thyroid nerve, a branch of the cranial laryngeal nerve that is itself a branch of the vagal nerve.26 The thyroid nerve runs in close association with the cranial thyroid artery. The parathyroid glands are small, ellipsoid disks that are anatomically intimately associated with the thyroid glands.26 Each thyroid gland is usually related to a pair of parathyroid glands. One of the parathyroid glands, often referred to as the external parathyroid gland, is usually found close to the cranial dorsolateral margin of each of the thyroid glands; however, the position of this parathyroid gland with respect to the thyroid gland varies.26 In some cases, the external parathyroid gland is not in contact with the thyroid gland, and in others, it is embedded into the surface of the thyroid and covered by the thyroid fascia. In rare individual dogs, it is found within the thyroid parenchyma. The second parathyroid gland is often embedded within each thyroid gland, generally in the caudal pole, at a depth that is variable. For this reason, this parathyroid gland has been referred to as the internal parathyroid gland. The internal parathyroid gland can also be found on the dorsolateral surface of the thyroid. Variations in location, number, and distribution of parathyroid glands are frequently reported. The vascular supply of the parathyroid glands is directly related to that of the thyroid glands.26 The external parathyroid glands receive blood supply by one or more branches from the cranial thyroid artery. In cats, the branch that supplies the external parathyroid gland may arise from the cranial thyroid artery after this artery has perforated the capsule of the thyroid gland.16 The internal parathyroid glands receive blood supply from vessels surrounding the thyroid parenchyma. Venous and lymphatic drainage are through corresponding structures in the thyroid glands. Innervation is the same as that of the thyroid. Ectopic thyroid tissue is common in dogs and cats. Functional accessory thyroid tissue can be found along the trachea, at the thoracic inlet, within the mediastinum, and along the thoracic portion of the descending aorta.26 Ectopic parathyroid tissue, however, is relatively uncommon. Only about 3% to 6% of dogs have been reported to have accessory parathyroid tissue. When present, this tissue can be found in the thorax associated with the thymus. Conversely, ectopic parathyroid tissue has been detected histologically in 35% to 50% of cats.15,24,31 Ionized calcium (Ca2+) is the physiologically active form of calcium and is regulated within a tight concentration range by the action of parathyroid hormone. As such, secretion of parathyroid hormone is regulated by serum ion calcium concentration. A high calcium concentration inhibits parathyroid hormone secretion (negative-feedback homeostatic control), and a low concentration stimulates its production. Parathyroid hormone has a relatively short half-life, which has been reported to be 3 to 5 minutes in humans;3,45 therefore, its effects are rapid. The majority of cats have a benign thyroid tumor described as multinodular adenomatous goiter, adenomatous hyperplasia, or adenoma. It is unknown if these are three distinct conditions or the same condition identified with different terms.10 Because they represent a benign process and have no clinical and prognostic differences, they are usually included under the term adenomatous hyperplasia.30,34 In approximately 1% to 4% of cats, hyperthyroidism is caused by a carcinoma of thyroid origin (Figure 121-2),10,14,34 although one recent study reported carcinoma in 25% of thyroid tissue examined.23 Histologic distinction between adenoma and adenocarcinoma can be difficult; features are similar because thyroid carcinomas typically do not show marked anaplasia, or pleomorphism. Histologic features that distinguish adenocarcinoma from benign adenoma in cats are degree of capsular and vascular invasion, local tissue (adipose or muscle) invasion, mitotic activity, regional lymph node involvement, and distant metastasis.6,25 The metastatic rate of carcinomas may be as high as 71%.6 Nonfunctional thyroid adenomas have been reported in cats,32 and nonfunctional carcinoma has also been reported but is infrequent.25 Approximately 70% of cats with hyperthyroidism have bilateral involvement of their glands; one study reported bilateral disease as high as 91%.30 Hyperfunctional ectopic tissue is found in 9% to 23% of cats, with the majority being in the chest.23,30 Most cats with hyperthyroidism present with clinical signs of polyphagia, weight loss, polydipsia and polyuria, increased activity and restlessness, and gastrointestinal signs (e.g., vomiting and diarrhea). Less common signs include respiratory signs, such as dyspnea or panting; tremors; seizures; and ventroflexion of the neck. About 14% of cats have apathetic hyperthyroidism with decreased appetite and lethargy, which is contradictory to the most common presentation.10 The prevalence of preexisting chronic renal insufficiency in cats with hyperthyroidism has been reported to be as high as 40%.53 Renal blood flow, glomerular filtration rate, and renal tubular resorptive and secretory capacities can be increased or decreased in cats with hyperthyroidism. Hyperthyroidism can mask preexisting chronic renal insufficiency: in up to 39% of treated cats, impaired renal function becomes apparent only after treatment of hyperthyroidism.53 Significant changes in kidney function occur within 4 weeks after treatment and none thereafter.54 Determining which cats have increased blood flow to the kidneys and which have decreased flow is challenging. Initially, cats with no azotemia and urine specific gravity greater than 1.035 were thought to have a reduced risk of manifesting renal insufficiency after treatment; however, these cats can still develop azotemia and, therefore, a suitable cutoff value for urine specific gravity cannot be given.53 Results of glomerular filtration rate, urine specific gravity, serum total T4, and urine retinol-binding protein-to-creatinine ratio before hyperthyroid treatment may be useful in predicting development of chronic renal disease in cats.53,54 Ultimately, a trial course of therapy with methimazole is the more prudent approach given that the effects of methimazole are reversible when therapy is discontinued. Serum biochemical and urinalysis values are monitored during methimazole treatment to determine which cats develop renal insufficiency and thus should not receive a permanent and irreversible therapy. In one study, hypokalemia was detected in 32% of cats with hyperthyroidism.30 Potential causes include loss from vomiting, diarrhea, anorexia, or polyuria.10 Also, thyrotoxicosis may increase the sensitivity of all body systems to catecholamines. Stress-induced release of catecholamines and increased plasma thyroid hormone concentration may stimulate movement of potassium from the extracellular space into the intracellular compartment in cats with hyperthyroidism.30 Presumptive diagnosis of feline hyperthyroidism is based on the history, signalment, and results of physical examination. More than 90% of cats have a palpable thyroid nodule in the neck, although the mass may be close to the thoracic inlet instead of the normal location.10 Measuring serum total T4 concentration is the screening test of choice. Serum total T4 concentration is increased in more than 90% of cats with hyperthyroidism.10 If a cat suspected to have hyperthyroidism has a normal serum total T4 concentration, the test should be repeated days to weeks later. If the results remain normal but the diagnosis is still suspected, further testing may include measurement of free T4 concentration, T3 suppression test, or TRH stimulation test.10 Scintigraphy can be a very useful tool to confirm the diagnosis and, critically for surgeons, anatomically localize hyperactive tissue. The most common radionuclide used to perform thyroid scintigraphy is technetium-99 m (99mTcO4 or pertechnetate). The pertechnetate ion is trapped by thyroidal iodide-concentrating mechanism but is not incorporated into organic thyroid hormone. Therefore, pertechnetate uptake reflects the trapping mechanism, but not function, of the gland. Antithyroid drugs (e.g., methimazole) do not affect the trapping mechanism of the thyroid pump; instead, they inhibit the organification of the iodine and coupling of iodotyrosyl groups. Therefore, pertechnetate still concentrates in the thyroid gland even after a cat has been made euthyroid with these drugs.10 Pertechnetate normally concentrates in three tissues: thyroid glands, salivary glands, and gastric mucosa. Normal cats have a 1 : 1 ratio in size, shape, and uptake intensity of salivary and thyroid glands. Most cats with bilateral hyperthyroid disease are asymmetric, with one gland larger than the other. Because of negative feedback of thyroid hormone on the pituitary gland, all normal thyroid tissue should be completely atrophied in a hyperthyroid cat. Therefore, any scintigraphic thyroid activity in a hyperthyroid cat should be interpreted as active adenomatous or cancerous tissue.10 Scintigraphy cannot be used reliably to differentiate adenomatous hyperplasia from carcinoma.23,25,30 Preoperative management is very important in cats with hyperthyroidism, considering the multisystemic nature of the disease and the severe consequences that the disease can have on other organs, particularly the heart and kidneys. For this reason, surgery must be approached as an elective procedure and not one that must be performed hastily.10 The way to achieve this goal is to treat the cat preoperatively to achieve a euthyroid state and control the adverse effects of the disease on the heart. Propylthiouracil and methimazole block synthesis of thyroid hormones by inhibiting organification of iodide and coupling of iodothyronines to form T4 and T3. Methimazole is the drug most commonly used in the United States. Carbimazole is metabolized to methimazole and therefore has equivalent effects. Because it may be better tolerated, carbimazole is recommended in countries where it is licensed.10 Methimazole should be administered orally or topically for 6 to 12 weeks before the surgery. The dose of the drug should be increased in a stepwise manner to gradually control the hyperthyroid state while minimizing the likelihood of side effects. A suggested protocol10 is to start with an oral dosage of 5 mg/d (2.5 mg twice a day) for 2 weeks. If there are no side effects noticed, the dosage can be increased to 7.5 mg/d for another 2 weeks. After this 4-week trial period, the cat is reevaluated, including serum T4 concentration (4 to 6 hours after the last administration of methimazole). If T4 concentration is normal, the dose is maintained for another 2 to 6 weeks. If the T4 concentration is too low, the dose is reduced. If the T4 concentration is too high, the dose is increased by another 2.5 mg/d every 2 weeks until the serum T4 concentration is in the normal range. More details regarding methimazole therapy can be found in the section on other therapies for feline hyperthyroidism (below). An additional benefit of methimazole therapy is that it will slow the heart rate.14 Among other deleterious effects, tachycardia prevents good coronary perfusion, which occurs during diastole. For cats with severe tachycardia or supraventricular tachyarrhythmias, a beta-antagonist drug such as propranolol or atenolol can be administered to slow the heart rate, improve cardiac stroke volume, and increase cardiac output.10,14 Anesthetic drugs to be avoided include those that stimulate or potentiate adrenergic activity or are capable of inducing tachycardia and arrhythmias,10 such as ketamine, halothane, and atropine. Most cats can be sedated with acepromazine and butorphanol or buprenorphine; acepromazine should not be used in hypovolemic or dehydrated cats.4 Anesthesia can be induced with intravenous propofol or etomidate; etomidate should only be used in well-sedated cats.4 Mask or box induction with sevoflurane or isoflurane is acceptable if animals are not struggling. Rapid metabolic rate can increase the absorption, distribution, tissue uptake, and inactivation of anesthetic agents. Cats with hypokalemia should receive potassium supplementation before anesthesia.30 This can be accomplished with potassium-supplemented parenteral fluid therapy.10 If possible, jugular sampling on the affected side should be avoided before surgery because hematoma formation can obscure dissection. The patient is placed in dorsal recumbency. A towel can be placed under the neck to slightly elevate it. The thoracic limbs should be pulled caudally and positioned along the chest for the surgery. Because this is a relatively delicate surgery, use of fine instruments, including iris or other fine scissors, can make surgery easier and more precise. Sterile cotton-tipped applicator swabs and bipolar cautery can be helpful during dissection. Careful hemostasis during the surgery to improve visualization while preserving the parathyroid gland and its blood supply is very important. Even small amounts of blood can dissect through the delicate fascia surrounding the thyroid gland and obscure the location of important anatomic structures such as blood vessels, parathyroid glands, and recurrent laryngeal nerve.6 The thyroid glands are identified and inspected. Whereas the normal thyroid lobe is pale tan, thyroid adenomatous hyperplasia or adenoma is typically brown to reddish brown.10 Unaffected glands should be atrophied and therefore small, thin, and pale;51 however, a distinction between normal and atrophied is not always obvious. If a gland looks normal, in all likelihood, it is abnormal and should be removed.6,10,34,51 Inspection and identification of the external parathyroid glands is also necessary. The external parathyroid gland is typically spherical, 1 to 3 mm in diameter, paler than thyroid tissue, and usually located on the ventral surface of the cranial pole of the thyroid gland.51 In some cats, the parathyroid gland is located at the middle or caudal pole of the thyroid gland.10,51 Parathyroid glands must be differentiated from fat deposits on the thyroid capsule. The parathyroid glands have a fine blood vessel that bifurcates and surrounds the gland;51 magnification is useful for identifying the vessel. If unilateral disease is determined on scintigraphy, surgery is an excellent treatment option because the risks are low compared with the risks for bilateral disease. Only one gland is resected when the disease is determined to be unilateral.10 The likelihood of the contralateral gland’s becoming hyperactive in the future appears low.30,47 Four techniques are used to perform a thyroidectomy: intracapsular, modified intracapsular, extracapsular, and modified extracapsular. The intracapsular technique involves incision into the thyroid capsule and blunt dissection of the parenchyma to separate it from the capsule and remove it, leaving the capsule in situ to preserve the external parathyroid. Rate of hyperthyroid recurrence is high with the intracapsular technique because of regrowth of microscopic or gross tissue that adheres and remains attached to the capsule. With the extracapsular technique, the intact thyroid gland is removed with its capsule, with no attempt to preserve the external parathyroid gland.6,14 This technique results in a high rate of hypoparathyroidism in animals with bilateral disease. Modified techniques are aimed at decreasing the respective complications and are preferred to the original intracapsular and extracapsular techniques. Modified Extracapsular Technique.: The thyroid capsule is incised with a #15 or #11 scalpel blade around the external parathyroid gland while preserving the parathyroid branch of the cranial thyroid artery (Figures 121-3 and 121-4). The external parathyroid gland and attached capsule are gently freed from the thyroid gland, being careful not to disturb the parathyroid artery. With fine hemoclips or 4-0 or 5-0 synthetic suture material, the thyroid artery is ligated downstream to the parathyroid artery bifurcation. Tissues downstream of the ligature are then transected. In rare cats with a caudal thyroid artery, this artery is ligated and transected as well. The thyroid gland can then be removed. Alternatively, the surgeon can begin with dissection and ligation at the caudal pole of the thyroid gland and gradually dissect cranially, leaving the dissection of the external parathyroid gland for the end.51 Modified Intracapsular Technique.: An incision is made in the ventral capsule of the thyroid gland in an avascular area using a scalpel blade or iris scissors. Blunt dissection of the thyroid parenchyma is performed with moistened cotton-tipped applicator swabs to tease the tissue from the capsule. Sharp dissection using iris scissors or similar delicate instruments can be required in areas where parenchyma is adhered to the inner surface of the capsule.6 The external parathyroid gland and associated blood vessels are preserved. Most of the capsule is then removed except for a small cuff of capsule adjacent to the external parathyroid gland and associated blood supply. A #15 or #11 scalpel blade or a pair of fine scissors is used to cut the capsule around the external parathyroid gland. If the external parathyroid gland is inadvertently removed or its blood supply damaged, autotransplantation of the parathyroid can be performed. The external parathyroid gland is freed from excised thyroid gland or the cervical area (in instances of vessel damage), and the parathyroid tissue is minced into approximately 1-mm cubes; alternatively, it can be left intact if small. A small incision in the sternohyoideus or sternothyroideus muscle is performed, and the parathyroid tissue is inserted into the muscle pocket. The muscle is closed with 3-0 or 4-0 absorbable suture.10,34,51 Transplantation of thyroid cells must be avoided during parathyroid autotransplantation. Parathyroid autotransplantation permits revascularization of the parathyroid tissue. The transplant will begin functioning within 7 to 21 days;10,34,51 therefore, acute hypocalcemia is not prevented. In cats undergoing bilateral thyroparathyroidectomy, a median duration of hypocalcemia is 71 days (range, 17 to 84 days).15 When autotransplantation of parathyroid tissue was performed after bilateral thyroparathyroidectomy, 87% of cats did not require postoperative calcium supplementation.35,51 Some authors suggest parathyroid autotransplantation in cats undergoing unilateral thyroidectomy,10 and others are concerned about the risk of transplantation of diseased thyroid along with parathyroid tissue.34 When a bilateral thyroidectomy is planned, some clinicians elect to stage the modified thyroidectomy procedures by 3 to 4 weeks.14,34,51 The waiting period allows time for the blood supply of the ipsilateral parathyroid gland, left in situ after unilateral thyroidectomy, to recover from surgical trauma. Staged bilateral thyroidectomy decreases the incidence of postoperative hypocalcemia34 but requires two anesthetic episodes. The risk of hypoparathyroidism must therefore be weighed against the risk of two anesthetic episodes. Removal of one adenomatous thyroid gland often resolves the clinical sign of hyperthyroidism for up to 6 months;14 the second thyroidectomy should therefore be performed within that time period. The overall prognosis for cats with hyperthyroidism treated by thyroidectomy is excellent.10,30 The perioperative mortality rate is very low when cats are properly managed preoperatively.30 Complications associated with thyroidectomy in cats include hemorrhage, dyspnea, laryngeal paralysis, Horner syndrome, hypothyroidism, hypoparathyroidism, and recurrence of hyperthyroidism.6 Hypoparathyroidism with resultant hypocalcemia is the most serious complication of bilateral thyroidectomy. Clinically significant hypocalcemia occurs in 6% or fewer of cats when bilateral thyroidectomy is performed by an experienced surgeon.30,51 A mild, transient decrease in serum calcium is common.10 Hypocalcemia is possible but rare after unilateral thyroidectomy.10 Even though 35% to 50% of cats have ectopic parathyroid tissue,15,24,31 all cats that have all parathyroid glands removed will become hypocalcemic, indicating that ectopic tissue is unable to initially maintain normocalcemia.15,35 Clinical signs of hypocalcemia include restlessness, facial or generalized muscle twitching, weakness, anorexia, panting, tetany, or convulsions and can occur 12 hours to 6 days postoperatively.15,51 Only 60% of cats with severe hypocalcemia (<6.5 mg/dL) demonstrate clinical signs.51 However, it has been proposed that all cats with severe hypocalcemia with or without clinical signs should be treated with oral vitamin D and calcium supplementation.10 Treatment of Acute Hypocalcemia.: In cats with acute signs of hypocalcemia, 10% calcium gluconate can be given at 0.25 to 1.5 mL/kg IV slowly (over 10 to 20 minutes) to effect.16,51 After the cat is stable, it is treated with a constant rate infusion (CRI) of calcium gluconate at 5 to 15 mg/kg/hr IV.51 Another option for intravenous maintenance is to place 10 mL of 10% calcium gluconate in a 250-mL bag of lactated Ringer’s solution and administer it at 60 mL/kg over 24 hours.6,16 It has been suggested that calcium salts should not be added to solutions containing lactate because they may precipitate.10 Ideally, an electrocardiographic (ECG) monitor should be used to monitor for bradycardia and arrhythmias during the intravenous administration of calcium; alternatively, the cat can be auscultated regularly to ensure safety of the infusion, particularly when the bolus is administered. If cardiac abnormalities arise, the infusion should be discontinued temporarily and eventually resumed at a slower rate. An alternative to the intravenous protocol for parenteral maintenance is to take the dose of calcium gluconate (60 to 90 mg of elemental calcium/kg/d), dilute it 1 : 1 to 1 : 4 with saline, and administer the solution subcutaneously in two to three sites at divided doses two to four times a day.10,51 Development of calcinosis cutis with skin necrosis is a reported risk of subcutaneous calcium gluconate.40,44 Calcium chloride should be avoided for the intravenous and subcutaneous routes.10,16 Parenteral therapy is continued for 24 to 96 hours, the time it takes for vitamin D to take effect.
Thyroid and Parathyroid Glands
Anatomy
Parathyroid Glands
Ectopic Tissue
Physiology
Parathyroid Hormone and Calcium Metabolism11,21
Feline Hyperthyroidism
Clinical Signs
Clinical Findings
Renal Disease
Hypokalemia
Diagnostics
Scintigraphy
Preoperative Management
Anesthesia
Surgery
Approach
Thyroidectomy
Parathyroid Autotransplantation
Staged Bilateral Thyroidectomy
Complications and Prognosis
Hypoparathyroidism
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Thyroid and Parathyroid Glands
Only gold members can continue reading. Log In or Register to continue