and Daniele Tomassoni1
(1)
Department of Experimental Medicine and Public Health, University of Camerino, Camerino, Italy
Abstract
Arterial hypertension is a well-known risk factor for stroke and cognitive deterioration of vascular origin, including vascular cognitive impairment (VCI) and vascular dementia (VaD). Patients with ischemic VaD have a significantly greater incidence of hypertension than patients with dementia of the Alzheimer’s type. Areas of white matter (WM) hyperintensity and ventricular enlargement are more common in hypertensive subjects compared with that in normotensive individuals. An association between hypertension and reduced cerebral blood flow (CBF) and VCI is documented. Hypertension in midlife is associated with a higher probability of cognitive impairment than hypertension developed later in life. These findings collectively indicate that arterial hypertension is a main cause of vascular brain disorder (VBD).
In this chapter, the main characteristics of cerebrovascular and brain damage in spontaneously hypertensive rats (SHR) and stroke-prone (SP) SHR are reviewed. SHR and SHR-SP are two widely used animal models of hypertension and hypertension plus stroke, respectively. They are normotensive at birth and gradually develop severe hypertension in the first 2–4 months of life. At 6 months, they have developed a sustained hypertension. SHR were the model most extensively investigated for assessing hypertensive brain damage and treatment of it. The time-dependent rise of arterial blood pressure, the occurrence of brain atrophy, loss of nerve cells, and glial reaction are phenomena shared to some extent with hypertensive brain damage in humans. SHR, therefore, can represent a reasonable model of morphological damage induced by hypertension and possibly reversed by appropriate antihypertensive treatment.
From a neurotransmitter point of view, SHR present changes of some specific neurotransmitter systems that may have functional and behavioral relevance. An impaired cholinergic neurotransmission characterizes both SHR and SHR-SP, similarly as reported in patients affected by VaD. The possibility that cholinergic neurotransmission mechanisms contribute to central nervous system plasticity or may improve brain circulation in this animal model is suggested by some studies. SHR are also characterized by a dopaminergic hypofunction and noradrenergic hyperactivity. This causes an imbalance between noradrenergic hyperfunction and dopaminergic hypofunction similarly as occurs in attention-deficit with hyperactivity disorder (ADHD).
The majority of microanatomical, behavioral, and neurochemical data on SHR are in favor of the hypothesis that this strain is a suitable model of VBD. Changes in catecholaminergic transmission put forward SHR as a possible model of ADHD as well. Hence, SHR could represent a multifaced model of two important groups of pathologies, VBD and ADHD. As for most models, researchers should always consider that SHR offer some similarities with corresponding human pathologies, but they do not suffer from the same disease.
Key words
HypertensionHypertension plus strokeRat modelVascular dementia1 Introduction
For most of the last century, adult-onset dementia disorders were generally attributed to arteriosclerosis and consequently chronic cerebral ischemia. The increasing capabilities of diagnosing Alzheimer’s disease and the demonstration that infarcts more likely than chronic ischemia were the basis of dementia of vascular origin brought about the introduction of the concept of multiinfarct dementia. The definition of multiinfarct dementia was replaced by the term “vascular dementia” (VaD) as a consequence of the recognition that different etiologies apart from multiple infarcts, such as single infarct, episodes of hypotension, leukoaraiosis, incomplete infarction, and hemorrhage in specific brain areas, elaborating cognitive functions can be involved (1, 2). However, in the currently accepted definition of dementia, the presence of memory disturbances is considered as the sine qua non condition for a diagnosis of dementia, whereas at least in the early phases of cognitive impairment associated with cerebrovascular disease, executive control dysfunction occurs earlier than the development of other symptoms. This observation led to the development of the concept of Vascular cognitive impairment (VCI), as a part of the broader concept of vascular cognitive disorder (VCD) (1, 2).
Arterial hypertension represents a well-known risk factor not only for stroke but also for VCI (3–6). Several studies have suggested a positive correlation between hypertension and cognitive deterioration (7, 8). Patients with ischemic VaD have a significantly greater incidence of hypertension than patients with dementia of the Alzheimer’s type (9). Neuropathological and neuropsychological investigations have shown that areas of white matter (WM) hyperintensity and ventricular enlargement are more common in hypertensive subjects compared with that in normotensive individuals. An association between hypertension and reduced Cerebral blood flow (CBF) and VCD is documented (3–5). Longitudinal studies have also suggested that hypertension in midlife is most associated with a higher probability of cognitive impairment than hypertension developed later in life (10).
Arterial hypertension is a multifactorial and polygenic disease involving complex interactions between genetically determined homeostatic control mechanism and environmental factors (11, 12). Exploration of such a complex syndrome requires the availability of animal models. Essential hypertension is the most common form of arterial hypertension in human beings. Multiple genes contribute to the individual phenotype, each expressing diverse alleles and degree of penetration.
The ideal animal model for hypertension research should have human-like cardiovascular anatomy, hemodynamics, and physiology and should develop a disease course and complications in a timely or even accelerated fashion (13). The phenotype-driven experimental approach takes advantage of a natural variation among inbred strains and crosses to find quantitative traits and determine which genes are responsible (14). The phenotype-driven rat experimental model is the most abundant for hypertension research and has particular potentials for exploration of polygenic hypertension. In general, development of homozygous hypertensive rat strains is achieved by selective breeding of animals displaying the desired phenotype over several generations. Once a trait is fixed, sib mating is maintained for about 20 generations to achieve genetic homogeneity. This approach has been useful to develop fundamental models such as spontaneously hypertensive rat (SHR) (15) or stroke-prone SHR (SHR-SP) derived from the Wistar strain (16, 17).
Much progress in experimental research was made with the development of rat strains with characteristics of human disease to evaluate the pathophysiology and the pharmacological treatment of a given disease. Although an animal model for VBD does not exist, SHR currently represents the best model of essential hypertension, allowing investigation of causes, mechanisms, and pathology of hypertension (18).
In this chapter, the main characteristics of cerebrovascular and brain damage in SHR are reviewed, and the validity of this rat strain as an animal model of VBD is discussed.
2 Development and Main Characteristics of Spontaneously Hypertensive Rats
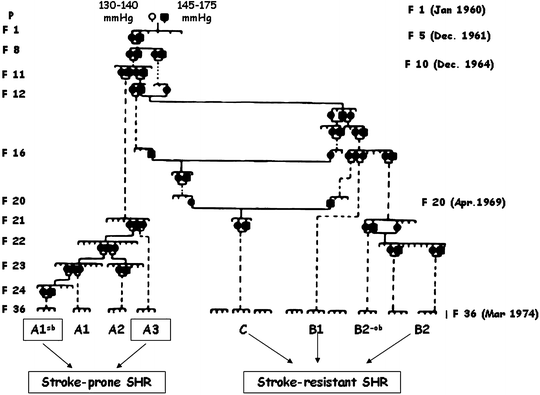
SHR and SHR-SP are two widely used animal models of hypertension and hypertension plus stroke, respectively (19–22). They are normotensive at birth and gradually develop severe hypertension in the first 2–4 months of life. At 6 months, they show a sustained hypertension (23, 24). The development of the SHR strain began in 1950s by Okamoto and coworkers. The selection of hypertensive rats started by mating male Wistar Kyoto rats (WKY) with mild hypertension (systolic pressure 145–175 mmHg) and females with relatively high blood pressure (systolic pressure 130–140 mmHg). Through their efforts to select hypertensive offspring by repeatedly checking the blood pressure, they finally were able to establish a colony of rats developing hypertension without exception and, in 1963, reported them as SHR (15–17) (Fig. 1).
To develop a suitable model of hypertension-related human cardiovascular pathology, a new substrain of SHR, namely the SHR-SP strain, was established through selective inbreeding of the SHR offspring of animals that had died of stroke. In SHR-SP, more than 80% of the population develops stroke (25). SHR and SHR-SP spontaneously develop moderate-to-severe hypertension and are different from the Dahl salt-sensitive or the Sabra-hypertensive strain, which require salt loading or deoxycorticosterone acetate (DOCA) salt administration for the development of a severe hypertension (26, 27). SHR generally develop hypertension with a systolic blood pressure plateau of about 200 mmHg. SHR-SP show a more rapid increase in blood pressure from young age and reach blood pressure values of approximately 240 mmHg. SHR, and to a greater extent SHR-SP, have lower body weight compared with WKY rats. SHR-SP, when compared with SHR, show various symptoms of cerebrovascular involvement such as irritability, hyperkinesis, hypokinesis, and paralysis (17).
The genetic mechanisms of hypertension in SHR have been attributed to both neural and vascular alterations. Individual variations in the genetic background of SHR may influence evolution of hypertension and end-organ damage resulting from elevated arterial pressure (28). Three major genes are involved in early development of hypertension, whereas an additional gene identified on chromosome 10 contributes to development and maintenance of hypertension during aging in SHR (29). Androgens probably modulate hypertension and age-related renal injury in this model (30) by altering natriuretic mechanisms and tubuloglomerular feedback (31) in a manner potentially resembling human hypertension. This prominent role of androgens in SHR suggests that the model could imitate postmenopausal hypertension as well (32).
The possibility to develop a 100% hypertensive strain of rats by selective inbreeding demonstrates that the heredity itself is an extremely important factor. Fingerprint analyses in SHR-SP and WKY disclosed various DNA polymorphisms. Linkage of these polymorphisms has allowed the identification of a Bp1 gene on rat chromosome 10 with a LOD score of 5.10. This gene is linked to the gene encoding angiotensin-converting enzyme (ACE), an enzyme playing a major role in blood pressure homeostasis and which is an important target of antihypertensive drugs (33–35). Analysis using molecular biology techniques and cDNA microarray has identified genes differentially expressed between SHR and WKY rats (36). Twenty-four genes were found to be upregulated and 20 were downregulated in SHR, even though the identification of differentially expressed gene may not be an efficient method for selecting candidate genes for hypertension in the SHR-WKY system (36).
In general, in most phenotype-driven models, hypertension is associated with cardiac hypertrophy, endothelial dysfunction, and renal functional impairment (proteinuria and decreased creatinine clearance). Other complications of hypertension such as cardiac insufficiency and end-stage renal disease are not constantly observed (37). The outcomes seem to depend on the underlying origin, genetic background, and possibly species differences, as well as on the degree of hypertension.
In SHR, cardiac function is preserved at 1 year of age but tends to fail between 18 and 24 months. At this age, 57% of SHR have a myocardial decompensation that can be identified by clinical signs. On echocardiography, left ventricular diameters are increased together with a reduced left ventricular fractional shortening and left ventricular filling pressure (38). SHR with heart failure are characterized by increased myocardial apoptosis and neurohumoral changes in comparison with SHR without heart failure (39, 40).
SHR with heart failure appear to be the ideal model to mimic hypertension-induced heart failure in humans due to the expression both in the animal model and in human beings of relevant cardiac, hemodynamic, and neurohumoral changes during the transition from compensated hypertrophy to failure. Another advantage of this model is that it does not require technical interventions and is not complicated by the mortality rate caused by surgical or pharmacological manipulations. A main limitation of this model is the long time required until the development of heart failure over a period of approximately 2 years.
3 Cerebrovascular Anatomy and Physiology in SHR
SHR were the model most extensively investigated for assessing hypertensive brain damage and treatment of it. The time-dependent rise of arterial blood pressure, the occurrence of brain atrophy, loss of nerve cells, and glial reaction are phenomena shared to some extent with hypertensive brain damage in humans. SHR, therefore, can represent a reasonable model of morphological damage induced by hypertension and possibly reversed by appropriate antihypertensive treatment (23).
In hypertensive humans, CBF is comparable to normotensive individuals. The constancy in CBF is achieved through a rise of cerebrovascular resistance, because the median blood pressure is increased. In chronic hypertension, cerebral blood vessels show hypertrophy and hyperplasia, limiting their capacity to dilate. Hence, chronic hypertension increases resistance of large cerebral arteries, leading to protection of microcirculation from elevated blood pressure (41). While these changes protect the brain from high perfusion pressures, they impair dilation of the vessels at low blood pressures and therefore impair adaptation mechanisms of brain circulation (42, 43).
Cerebrovascular alterations typical of hypertension are closely related to the degree of blood pressure elevation and consist in hypertrophy or remodeling. Hypertrophy consists in thickening of arterial wall with decrease of internal diameter, increase of external diameter and consequently reduction of lumen size. Remodeling consists of smooth muscle rearrangement with decrease of both external and internal diameters and lumen size reduction (44–47). These changes collectively increase the wall-to-lumen ratio and consequently arterial resistance.
In terms of possible anatomical selectivity of cerebrovascular involvement in SHR, frontal cortex of 24-week-old SHR displayed wall hypertrophy and luminal narrowing (Fig. 2) (48 49). In the striatum, the increase of wall area was not accompanied by luminal narrowing, indicating the occurrence of arterial hypertrophy without vasoconstriction (49). In hippocampal arteries of SHR, luminal narrowing was not accompanied by changes of wall area (49). In line with the hypothesis of a prevalent smooth muscle hypertrophy, cerebral arteries of SHR aged 24 weeks display prominent lumen diameter reduction compared with normotensive WKY rats (Fig. 2) (49). An important parameter in the analysis of vascular changes is the values of wall-to-lumen ratio, which directly relates to vascular resistance (50). It has been emphasized that the increase of the wall-to-lumen ratio is an important factor in the pathogenesis and maintenance of hypertension (51). Assessment of wall-to-lumen ratio, plotted versus lumen diameter of vessels in 24-week-old SHR (Fig. 2), revealed an increased sensitivity of intracerebral vessels to hypertension parallel with the decrease of luminal diameter (49). In general, smallest arterioles were the vessels most sensible to hypertension and may represent candidates to degenerative phenomena leading to brain hypertensive damage. These findings are compatible with data of a significant increase of smooth muscle actin-immunoreactive areas in pial arteries of SHR, without increase in the collagen content (52). Parenchymal small arteries of SHR-SP undergo fibrinoid necrosis with a decrease of smooth muscle actin, whereas small arteries perfusing WM do not show these changes (53).
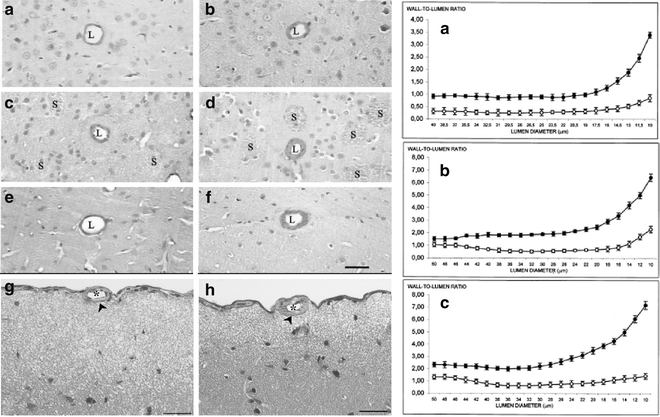

Fig. 2.
Left–hand panel – Micrographs of arterioles of frontal cortex (pictures a and b), striatum (pictures c and d), and hippocampus (pictures e and f) in Wistar-Kyoto (WKY) rats (a, c, and e) and spontaneously hypertensive rats (SHR) (b, d, and f) aged 6 months. Micrographs of pial artery of 6-month-old WKY rats (g) and age-matched SHR rats (h). Hematoxylin and eosin. In intracerebral arterioles of frontal cortex, striatum, and hippocampus of SHR, an increased thickness of the wall and luminal narrowing were found in comparison with WKY rats. L, arterial lumen; s, striosomes. Calibration bar: 30 µm. Right-hand panel – Plotting of wall area values versus lumen diameter in arteries (lumen diameter <46 µm) and arterioles (lumen diameter between 46 and 10 µm) of frontal cortex (panel a), striatum (panel b), and hippocampus (panel c) in Wistar-Kyoto (WKY) rats (○) and SHR (●). Data are the mean ± S.E. In frontal cortex and striatal vessels, differences were statistically significant (P < 0.01 versus WKY rats) in all different-sized vessels examined. In hippocampus, differences were statistically significant (P < 0.01 versus WKY rats) since vessels with a lumen diameter of <40 µm (Adapted from (49)).
A moderate increase in arterial blood pressure induces cerebral blood vessel autoregulation and consequently constriction (54). With severe increase in pressure, the autoregulatory capacity of cerebral vessels is exaggerated and large cerebral arteries dilate (55). This dilatation may lead to increased stress of the vessel wall, with consequent probable blood–brain barrier (BBB) damage. In chronic hypertension, vascular changes might influence the susceptibility of BBB. Adult SHR cerebral vessels are less susceptible to disruption of the BBB induced by rise of blood pressure than WKY rats (56). This does not occur in young SHR, which are as susceptible as WKY rats to cerebral complications of abrupt elevation of blood pressure (57).
It has been suggested that chronic hypertension in SHR may cause more pronounced defects in the integrity of the blood–cerebrospinal fluid (CSF) barrier than in the BBB (58). An increased permeability of the blood–CSF barrier toward sucrose was found in 4-month-old SHR, whereas BBB was found to be resistant to passage of sucrose and lanthanum in both SHR and WKY rats. This indicates that BBB functionality is maintained (58).
The expression of aquaporin 4 (AQP4 ), a membrane channel protein serving as selective pores through which water crosses the plasma membranes, was assessed by one group to verify the integrity of the BBB and the characteristic of cerebral water content in SHR. AQP4 is expressed in astrocyte foot process near blood vessels in rat (59, 60) and human brain (61), as well as in ependymal and pial surface in contact with CSF (59). This localization suggests that AQP4 plays a critical role in brain water balance. Different studies indicate that the expression of brain aquaporins is sensitive to brain injury, swelling, and other experimental procedures. In rodents, AQP4 is upregulated in astrocytes in response to cerebral edema caused by brain injury (62, 63), focal brain ischemia (64), and hyponatremia (65). In rat models of brain injury, AQP4 is overexpressed in astrocytes at the site where BBB is disrupted (62). Figure 3 shows AQP4 immunohistochemistry in frontal cortex of 6-month-old SHR and age-matched WKY rats. An intense immunoreaction is noticeable in cerebral vessels (Fig. 3), with a remarkable increase of immune reaction in cerebral vessels of SHR (Fig. 3b). These findings suggest that an increased expression of AQP4 in the brain of SHR may be implicated in impaired autoregulation of CBF and water content in this animal model. Based on these data, the occurrence of a cytotoxic edema in SHR cannot be excluded. This may contribute to the development of brain lesions documented in SHR.
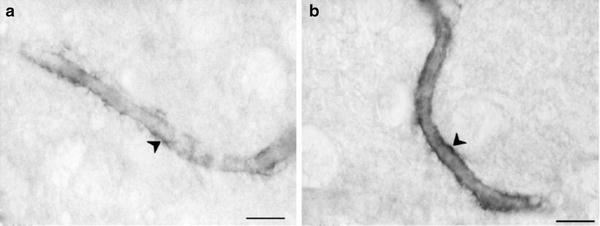

Fig. 3.
Micrographs of arterioles of cerebrocortical white matter (WM) in a Wistar-Kyoto (WKY) rat (a) and spontaneously hypertensive rat (SHR) (b) of 6 months. Aquaporin 4 (AQP4) immunohistochemistry. Note in SHR the increased expression of AQP4 immunoreactivity indicating enhanced blood–brain barrier permeability. Calibration bar: 15 µm.
4 Gross and Microanatomy of SHR Brain
Ventricular enlargement accompanied by loss of brain tissue, and decrease of brain weight and volume of grey matter were reported in 8-week-old SHR (66–68). Altered anatomical organization of hypothalamic regions (69) and gross morphological brain differences between SHR and age-matched normotensive WKY rats were reported (70). At 14 weeks of age, SHR display little differences in brain morphology compared with age-matched WKY rats (71). Around the age of 26 weeks, brain microanatomical changes become obvious in SHR (68, 71, 72). In 6–7-month-old SHR, ventricular volume is twofold greater in SHR than in WKY rats; the volume of the entire brain and of different gray matter areas as well as the thickness of cerebral cortex were 11–25% less in SHR (Fig. 4). Some investigators reported the same neuronal density in SHR and WKY rats at 6–7 months of age (67). However, the presence of less neurons in different brain areas of SHR is obvious if density/number of nerve cell profiles is corrected by the volume, which is decreased in SHR compared with their normotensive cohorts (68, 71). A more recent study employing magnetic resonance imaging (MRI) has reported significantly smaller caudate-putamen and cerebellar vermis in 4–6-week-old SHR compared with age-matched normotensive WKY rats (Fig. 5) (73).
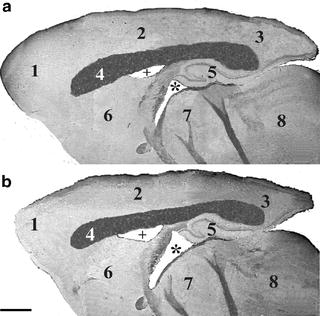
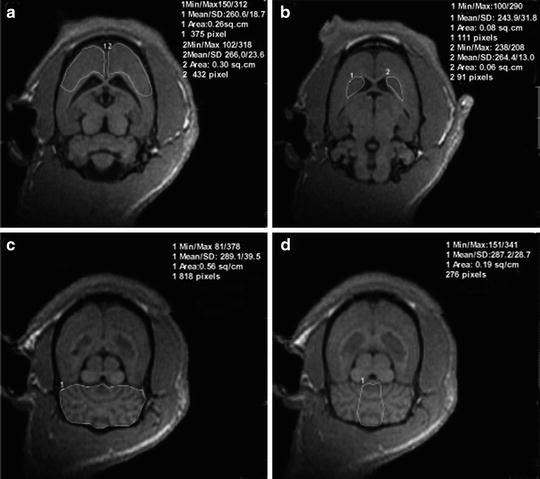

Fig. 4.
Parasagittal section of brain of a 6-month-old normotensive male Wistar-Kyoto (WKY) rats (a) and age-matched spontaneously hypertensive rats (SHR) stained with Luxol fast blue. Note in SHR the enlargement of lateral ventricle (+) and of the third ventricle (asterisk), a decreased thickness of cerebral cortex (2) and a reduced size of corpus callosum (4). 1: frontal cortex; 3: occipital cortex; 5: hippocampus; 6: striatum; 7: dienchephalon; 8: midbrain. Calibration bar: 1.5 mm (Adapted from (72) with permission from Wiley-Blackwell).

Fig. 5.
Magnetic resonance imaging (MRI) data from coronal sections of the brains of spontaneously hypertensive (SHR) and Wistar-Kyoto (WKY) rats. Image a shows the prefrontal area; b shows caudate nucleus; c shows the cerebellum; d shows the vermis cerebelli (Adapted from (71) with permission of publication from Elsevier).
The hippocampus, a key area for learning and memory, is smaller in 6-month-old SHR compared with that in normotensive cohorts (66, 74–76), as well as the density of nuclei in the CA1 subfield in SHR aged 6–7 months (67) (Fig. 6). Data of local cerebral glucose utilization in most gray matter areas of SHR (77, 78) suggest depressed neuronal activity in SHR around age 6 months. A greater susceptibility of SHR to brain ischemia is documented. Brain infarct size and behavioral abnormalities caused by middle cerebral artery (MCA) occlusion were greater in SHR than in WKY rats 3 weeks after occlusion of the right MCA (79).
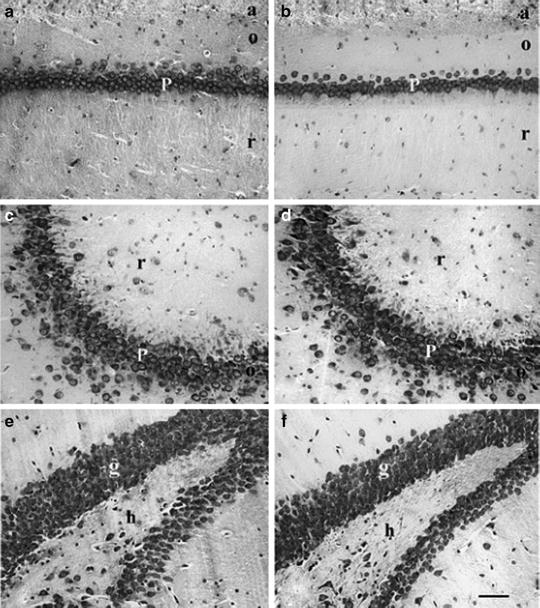

Fig. 6.
Sections of the CA1 subfield (a, b), CA3 subfield (c, d) and dentate gyrus (DG) (e, f) of the hippocampus of 6-month-old normotensive Wistar-Kyoto (WKY) rats (a, c and e) and age-matched spontaneously hypertensive rats (SHR) (b, d and f). Cresyl Violet staining. Note in SHR the decreased density of nerve cell profiles in the CA1 subfield and to a lesser extent in the DG. No changes in the number of nerve cell profiles can be seen in the CA3 subfield between WKY rats and SHR. P: pyramidal neurons; a: alveus; o: stratum oriens; r: stratum radiatum; g: granule neurons; h: hilum. Scale bar: 50 µm (Adapted from (75)).
4.1 White Matter Changes
WM changes are often associated with risk factors for stroke such as age and arterial hypertension (80). In aging and hypertension, the occurrence of WM lesions increases as documented by neuroimaging techniques, such as computerized tomography or magnetic resonances imaging (81, 82). WM lesions are closely associated with cognitive impairment and gait disorders in the elderly, and, therefore, may represent the anatomical correlate of complications of cerebrovascular disease and a predictive factor for the development of cognitive impairment and stroke (83). Data on WM in SHR are sparse. Smaller volumes of anterior commissure and genu of the corpus callosum were reported in 6–7-month-old SHR in comparison with WKY (67). A more systematic microanatomical study revealed a remarkable decrease in volume of internal capsule and striosomes, a moderate reduction in volume of the corpus callosum, and no changes in volume of the external capsule and of WM of hippocampus (68). The loss of forebrain WM in SHR is more pronounced in areas bearing fibers originating or ending in the cerebral cortex. These changes probably reflect the reduction of afferent or efferent cerebrocortical fibers to cerebral cortex (68). WM lesions characterized by disarrangement of nerve fibers, occurrence of vacuoles and/or disappearance of myelinated fibers were reported only in caudate-putamen of 20-week-old SHR. The same group also reported a marked WM increase in different areas (corpus callosum, anterior commissure, internal capsule, and caudate-putamen) of 20-week-old SHR-SP (52).
Recent studies of our group enclosed evidence of a microglial reaction in brain WM of 6-month-old SHR. Histochemistry of Bandeira simplicifolia isolectin BSI-B4 revealed an increase of positive reaction in corpus callosum and fimbria fornix (Fig. 7) and no obvious changes in the striosomes. Quantitatively, this phenomenon was more pronounced in brain areas undergoing volume reduction (84).
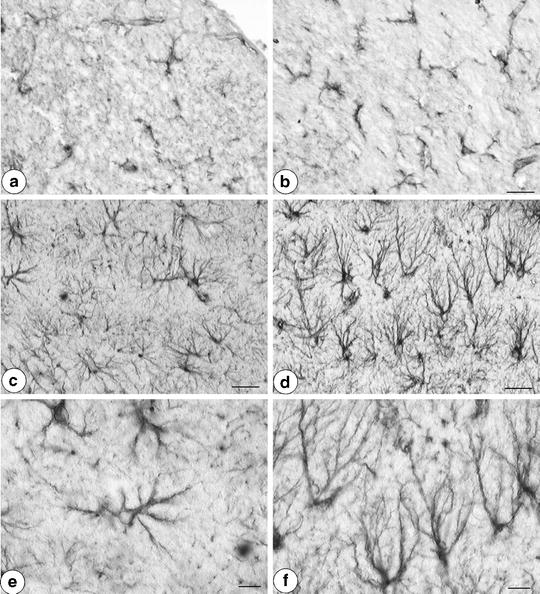

Fig. 7.
(a and b) Microglial reaction in Fimbria Fornix from Bandeira simplicifolia isolectin BSI-B4 binding. (a) 6-month-old Wistar-Kyoto (WKY) rat; (b) 6-month-old spontaneously hypertensive rat (SHR). Note in SHR the activation of microglia. Calibration bar: 25 µm. (c–f) Sections of rat dentate gyrus processed for glial fibrillary acidic protein (GFAP) immunohistochemistry. Pictures show parenchymal astrocytes. (c, e) 6-month-old WKY; (d, f) 6-month-old SHR. Note in SHR, the increase of dark-brown GFAP immunoreactive astrocytes in comparison with WKY rats. Calibration bar c, d: 25 µm. Calibration bar E, F: 10 µm.
Major histocompatibility complex (MHC) class I antigen-immunoreactive microglia were found primarily in the WM of SHR-SP and to a lesser extent in that of SHR but not in normotensive WKY rats (52). These findings, which are consistent with human data reported, suggest an involvement of parenchymal microglia in hypertension. Hyperactivation of microglia may cause neuronal degeneration, due to release of proinflammatory cytokines such as interleukin-1, interleukin-6, and tumor necrosis factor-α (85). On the other hand, microglial activation results from the engagement of degenerative and phagocytic processes following the formation of necrotic or apoptotic tissue after neuronal injury (86).
Spontaneous hypertension after bilateral common carotid artery ligation (BCAL) in SHR may accelerate WM lesions with vacuolation of nerve fibers, microglial activation, and oligodendroglial DNA fragmentation (87). It has been suggested that apoptosis-like cell death of oligodendrocytes was induced in the WM of hypoperfused SHR brain. The development of oligodendroglial DNA fragmentation in the WM might be mainly due to cerebral hypoperfusion. Increase of infarct size and delayed recovery of local CBF have been reported in SHR after BCAL (88). Besides hypertension, acute reduction and delayed recovery of local CBF in SHR might affect oligodendroglial vulnerability (87).
4.2 Astroglia
Astroglia perform many functions, including cellular support during central nervous system development, ion homeostasis, and constitution of the BBB. They also provide neuronal nutrition, take up neurotransmitters, and contribute to neuroimmune function (89–91). The cytoskeletal protein glial fibrillary acidic protein (GFAP) represents a selective marker for mature astrocytes (92). Different studies have assessed the functional significance of this astrocyte protein using genetic knockout animals (93–95). Astrocytes in GFAP knockout animals show disturbed neuronal plasticity, loss of long-term depression (94), late-onset dysmyelination (93), and increased susceptibility to ischemia (96). Astrocytes and microglia respond to a great variety of lesions in the nervous tissue. In this sense, although astroglial cells react in a relatively constant manner, the intensity and time course of these changes largely depend on the type of lesion involved. Transformation of quiescent into reactive astrocytes is termed astrogliosis or reactive gliosis and may result in the formation of a glial scar. This phenomenon occurs primarily in the immediate vicinity of a lesion (89). Astrocytes distal from an injured area do not undergo extensive and permanent changes (97).
Several different central nervous system injuries are accompanied by proliferation and/or hypertrophy of astrocytes. An increased expression of brain GFAP and of GFAP mRNA was reported in association with aging or disease (98–101) and also specifically in brain neurodegenerative disorders (102, 103). A significant increase of GFAP mRNA and an increase of GFAP immunoreactivity were noticeable in various brain areas of SHR compared with those in normotensive WKY rats at 6 months, but not at 2 or 4 months of age (104) (Fig. 7). Immunohistochemistry revealed a numerical (hyperplasia) and size (hypertrophy) increase of GFAP-immunoreactive astrocytes in frontal cortex, striatum, and hippocampus of SHR (Fig. 7) (75, 76, 104, 105). The occurrence of astrogliosis in the brain of SHR with established hypertension suggests that hypertension induces a condition of brain suffering enough to increase biosynthesis and expression of GFAP, similarly as reported in several neurodegenerative disorders and in brain ischemia.
5 Neurochemistry and Chemical Neuroanatomy of SHR
Besides the gross and microanatomical changes described earlier, SHR present changes of some specific neurotransmitter systems that may have functional and behavioral relevance. Cholinergic, dopaminergic, and noradrenergic systems are most affected and extensively investigated.
5.1 Cholinergic System
Acetylcholine (ACh) plays an important role in central, peripheral, autonomic, and enteric nervous system neurotransmission. ACh is also involved in the endothelium-dependent vasodilatation in several vascular beds and is present in different nonneuronal cells (106). Brain cholinergic neurons form a global network characterized by nerve cell bodies located in selected areas from which ascending and, to a lesser extent, descending projections originate (Fig. 8). Ascending cholinergic pathways supplying cerebral cortex are involved in important higher brain functions such as attention, arousal, motivation, memory, and consciousness (106). The cholinergic system is altered in age-related neurodegenerative diseases, such as Alzheimer’s disease, and is vulnerable to the effects of aging (106). A decline in the integrity of the cholinergic system characterized by a decrease of the ACh biosynthetic enzyme choline acetyltransferase (ChAT) and by a reduction of CSF ACh levels was also reported in patients with VaD (107). This, together with the observation of microanatomical changes typical of hypertensive brain damage in SHR, as well as of deficits in central nicotinic cholinergic receptors, has contributed to the validity of SHR as a possible model of age-related human neurological disorders characterized by impaired cholinergic function (108).
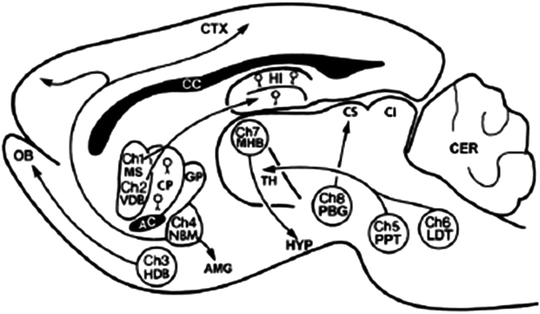

Fig. 8.
Main cholinergic nuclei and their projections represented in rat brain. Ch1-Ch8: main cholinergic brain nuclei; AC: nucleus accumbens; AMG: amygdala; CC: corpus callosum; CTX: cerebral cortex; CER: cerebellum; CS: superior colliculus; CI: inferior colliculus; CP: caudate-putamen; GP: globus pallidus; HDB: horizontal diagonal band; HI: hippocampus; HYP: hypothalamus; MS: medial septum; NBM: nucleus basalis magnocellularis; OB: olfactory bulb; PBG: parabigeminal nucleus; TH: thalamus; VDB: ventral diagonal band; MHB: mesopontine tegmental nuclei; PPT: pedunculopontine tegmental nuclei; LTD: laterodorsal tegmental nuclei (Adapted from (106) with permission of publication from Bentham Science Publishers Ltd).
Cholinergic neurons localized in various brain regions are implicated in blood pressure regulation, and central ACh may contribute to the pathogenesis of experimental hypertension. Brain regions expressing a cholinergic system related to hypertension are the rostral ventrolateral medulla (RVL), the posterior hypothalamus, and the lateral septal area (109, 110). The RVL is the major source of efferent sympathetic activity (111), and its cholinergic mechanisms are involved in the regulation of the cardiovascular system (110). Cholinergic function in RVL is enhanced in SHR, which could contribute to the development and/or maintenance of hypertension in this strain (110).
ChAT activity was reported to be enhanced in the brain stem of SHR (112). Intravenous administration of the ACh degradation inhibitor physostigmine, with or without coadministration of the peripheral muscarinic receptor antagonist methylatropine, evokes an exaggerated pressor response in unanesthetized SHR compared with normotensive WKY rats. In contrast, there is no difference in the hypertensive response to the cholinergic muscarinic receptor agonist oxotremorine in SHR or WKY rats pretreated with methylatropine (113).
The occurrence of an impaired cerebral cholinergic system, both in SHR and in SHR-SP, is documented. In SHR-SP, alterations of the cholinergic cerebral system consisting of a decrease of cerebrocortical and hippocampal levels of choline and ACh, as well as in cerebral spinal fluid, were found (114–116). Changes of ACh levels in cerebral cortex and hippocampus are related to impaired learning and memory functions (116, 117).
More recent studies have assessed the expression of vesicular ACh transporters (VAChT) in SHR (108, 118, 119). In nerve terminals, vesicular transporters pack neurotransmitters into synaptic vesicles, essential for neurotransmitter release. VAChT was investigated by immunochemical (108), and both immunochemical and immunohistochemical (118, 119) techniques in 4 and 15- (108) and 6-month-old SHR (118, 119). The studies reported a slight increase of the expression of the transporter in the hippocampus of the older age group investigated (108), and similarly to observed in frontal cortex, hippocampus and striatum of 6 months-old SHR (119). Treatment with the acetylcholinesterase (AChE) inhibitor galantamine countered the changes of VAChT expression in SHR (119). This suggests a greater sensitivity of the cholinergic system to pharmacological manipulation in this hypertensive strain. In humans, higher levels of VAChT expression were reported in early stages of Alzheimer’s disease (120), as well in the hippocampus of individuals with mild cognitive impairment (MCI) (121, 122). These changes were considered as an up-regulation mechanism to counter cholinergic hypofunction in early phases of adult-onset cognitive dysfunctions. The same is probably true in 15-month-old SHR (108) in which the increased expression of VAChT may represent an attempt to compensate cholinergic impairment at this stage of hypertensive brain damage. Focal ischemia did not affect the expression of VAChT in SHR (123).
In SHR, learning and memory abilities explored with behavioral tests (spatial memory, water maze, conditioning tests, and radial arm maze) revealed some impairments (108, 124–127). These findings, combined with observations of deficits in nicotinic ACh receptors in various brain regions, including cerebral cortex, hippocampus, thalamus, and striatum in SHR compared with those in WKY rats (128–130), further support the view of impaired cholinergic neurotransmission in the brain of SHR. The reduced density of central nicotinic ACh receptors in prefrontal cortex, caudate-putamen, and enthorinal cortex but not in the hippocampus of 15-month-old SHR could, in part, explain the modest spatial learning decrements by SHR compared with that in WKY (130). Analysis of central muscarinic cholinergic receptors in the same animal model demonstrated a significant increase of M1 receptors in cingulate cortex, neostriatum, CA1 and CA3 subfields of hippocampus, posterior hypothalamic area, and substantia nigra (108, 126). An increased M2 receptor density was reported in olfactory tubercle, nucleus accumbens, and the basolateral amygdaloid nucleus in 12-week-old SHR (127) and in the neostriatum of 15-month-old SHR compared with age-matched WKY rats (108). The increased expression of muscarinic cholinergic receptors in SHR might represent a postsynaptic up-regulation consequent to reduced cholinergic cerebrocortical projections with age and/or a compensatory phenomenon following central nicotinic cholinergic receptor deficiency in older SHR (108).
In summary, similarly as reported in VaD, an impaired cholinergic neurotransmission characterizes both SHR and SHR-SP. The possibility that cholinergic neurotransmission mechanisms contribute to central nervous system plasticity or may improve brain circulation in this animal model is suggested by our recent studies detailed in the section 6.4. In these experiments, we have observed that treatment of SHR with the AChE inhibitor galantamine or with the cholinergic precursor choline alphoscerate (alpha-glyceryl-phosphoryl-choline), alone or in association, although not affecting blood pressure levels, exerts a neuroprotective effect countering typical hypertension-dependent microanatomical changes in SHR (131).
5.2 Dopaminergic System
SHR exhibit behavioral abnormalities, such as hyperactivity, lower attention, inhibition deficit, and hyperreactivity to stress, that resemble behavioral abnormalities of human attention-deficit with hyperactivity disorder (ADHD). Because dopamine has been implicated in the pathogenesis of ADHD, possible abnormalities of this system were extensively investigated in SHR (132).
In SHR, terminals of mesocortical, mesolimbic, and nigrostriatal dopaminergic neurons release less dopamine in response to electrical stimulation and/or depolarization as a result of exposure to high extracellular K+ concentrations, than WKY (132, 133). Dopamine transporter (DAT) and vesicular storage of dopamine is impaired in SHR. DAT gene expression is transiently reduced in SHR midbrain during the first postnatal month and increased in adult SHR compared with controls (134, 135). Alterations in DAT gene expression can affect dopamine uptake and reutilization. This causes leakage of dopamine into the cytoplasm and increases d-amphetamine-induced transporter-mediated release. Release of dopamine evoked by electrical stimulation is reduced in prefrontal cortex of SHR, suggesting dopaminergic hypofunction in this cerebrocortical area. This hypofunction alters autoreceptor-mediated inhibition of noradrenaline release in SHR (136). From a receptor point of view, postsynaptic dopamine D1-like/D2-like receptors in the SHR are hyposensitive, while presynaptic dopamine D2-like receptors located particularly in the nucleus accumbens are hypersensitive compared with normotensive WKY rats (137). More recent studies have further characterized the dopaminergic system of SHR by analyzing genes coding for the five major subtypes of dopamine receptor (D1, D2, D3, D4, and D5), for enzymes responsible of catecholamine synthesis tyrosine hydroxylase and dopamine-beta-hydroxylase, and for DAT. Significantly lower expression of dopamine D4 receptor gene and protein synthesis was observed in the prefrontal cortex of SHR. No differences in the same genes were observed in the midbrain, prefrontal cortex, temporal cortex, striatum, or amygdala of SHR and WKY. These data, consistent with the situation seen in patients with ADHD, suggest a downregulation of dopamine D4 receptors, rather than a general downregulation of catecholamine synthesis in prefrontal cortex of SHR (73). In conclusion, the dopaminergic system is characterized by a hypofunction affecting primarily prefrontal cortex. DAT and dopamine D4 receptor genes are most probably involved in the dysfunctions of the dopaminergic system in specific brain areas, which resemble those identified in patients with ADHD.
5.3 Noradrenergic System
Analysis of SHR as a model of ADHD has also shown that, similarly as reported in the prefrontal cortex of children with ADHD, noradrenergic neurons of this cerebrocortical area are hyperfunctional in this animal model too.
Locus coeruleus is one of the main brain noradrenergic nuclei. Noradrenergic projections originating from this nucleus supply the entire cerebral cortex, various subcortical areas, cerebellum, and spinal cord. These nerve pathways are involved in attention, arousal, orienting, and vigilance (138). Noradrenergic projections to the prefrontal cortex modulate the transfer of information through neuronal circuits responsible for selective and sustained attention (138). In SHR, compared with normotensive WKY rats, noradrenaline levels are increased in several brain areas, including locus coeruleus, substantia nigra, and prefrontal cortex (139). This increase is probably because of an enhanced expression of the catecholamine biosynthetic enzyme tyrosine hydroxylase in the ventrolateral medulla oblongata (140, 141). Parallel to the increased noradrenaline levels, a downregulation of cerebrocortical alpha2-adrenoceptors, with decreased mRNA levels for the receptor and less-efficient alpha2-adrenoceptor-mediated inhibition of noradrenaline release, were found (132). In contrast, stimulus-evoked release of noradrenaline from prefrontal cortex slices of SHR was unchanged compared with that of WKY rats (142, 143). High firing of locus coeruleus neurons, overproduction of noradrenaline, and decreased alpha2-adrenoceptor-mediated regulation of the neurotransmitter, with its consequent spill over into the extracellular space are a trait of SHR. This may cause repeatedly increased release of noradrenaline from sympathetic nerve terminals and to consequent stress-dependent development of hypertension in SHR (144). In conclusion, the noradrenergic system is hyperactive in SHR, particularly in response to stress, thereby inducing an imbalance between noradrenergic hyperfunction and dopaminergic hypofunction similarly as occurs in ADHD.
5.4 Other Neurotransmitter Systems
Other neurotransmitter systems were investigated more sparsely. Some studies focused on the serotonergic system. Injection of serotonin into the nucleus tractus solitarius (NTS) elicits hypotension and bradycardia in rats. In SHR, this depressor response was markedly larger than in WKY rats. These results were considered suggestive that serotonergic system is altered in SHR rats, apparently acting tonically to reduce blood pressure, and that stimulation of 5HT-2 receptors in the NTS contributes to cardiovascular regulation independent of the baroreceptor reflex (145). In SHR-SP, repeated exposure to stress dramatically exacerbates neuronal death by transient ischemia. The 5-HT-4 receptor was suggested to be involved in this stress-induced exacerbating mechanism of the neuronal damage (146).
Amino acid neurotransmitter systems were investigated in SHR primarily in terms of their contribution to cardiovascular and blood pressure regulation but not for their possible interference with cognition and behavioural mechanisms; therefore, further investigations are required.
6 Preclinical Pharmacology and Pharmacotherapeutics
Risks of primary stroke and of secondary (recurrent) stroke are directly related to the blood pressure level. Clinical studies have clearly demonstrated that lowering blood pressure markedly reduces the incidence of stroke. Both SHR and SHR-SP were used in pharmacotherapy research to assess the effect of different neuroprotective treatments in case of stroke or hypertensive brain damage. Administration of antihypertensive doses of the α-adrenoceptor antagonist phentolamine or of the Ca2+ antagonist nicardipine decreased mean blood pressure during brain ischemia and suppressed the anomalous rise of arterial pressure occurring in ischemia. Although controlling blood pressure during an ischemic insult attenuates ischemia-induced damage of muscarinic cholinergic M1 receptors in the brain of SHR (147), lowering of blood pressure during stroke may exacerbate damage through reduction of blood supply to hypoperfused areas (steal effect).
A previous paper by our group has reviewed the influence of antihypertensive treatment on brain structure and function in animal models of hypertension with particular emphasis to SHR (22). Here we analyze the contribution of SHR and SHR-SP preclinical research in developing possible therapeutic strategies for protecting brain from arterial hypertension.
6.1 Calcium Antagonists
Arterial hypertension is characterized by altered Ca2+ metabolism not only in brain but also in other tissues, including vascular smooth muscle. In SHR, increased intracellular Ca2+ concentrations were attributed to genetic abnormalities in Ca2+ATPase (132). Increased intracellular Ca2+ levels have several negative consequences on neurons, including decreased neurotransmitter release, altered Ca2+ signaling, impaired mitochondrial ATP synthesis and increased levels of reactive oxygen species. A better knowledge of altered Ca2+ metabolism in arterial hypertension has brought to the proposal of Ca2+ antagonists (Ca2+ channel blockers) as antihypertensive agents.
The dihydropyridine derivative nimodipine was investigated primarily in the treatment of brain ischemia. Protocols in general included intravenous administration of the drug or local application in the hippocampus. The compound was given before, during, and after ischemia in rabbit. Although nimodipine-sensitive Ca2+ channels (of the L-type) play a minor role in regulating Ca2+ influx of hippocampal neurons, the compound possesses a vasotropic action and may also protect brain neurons from neurotoxicity caused by Ca2+ overload (148). Studies on the effects of association between nimodipine and mannitol on infarct size and on the amount of apoptosis after transient focal cerebral ischemia have demonstrated that the two compounds provide neuroprotection by preventing both necrotic and apoptotic components of cell death (149). Another study has assessed the influence of a 12-week treatment with a dose of 15 mg kg-1 day-1 of nimodipine on hypertensive brain damage (71) and cerebrovascular changes (150) in SHR. Although the compound only partially lowered systolic blood pressure, it displayed cerebrovascular activity and was able to provide neuroprotection (71, 150).
Another series of studies have investigated the effects of isradipine on permanent and transient focal cerebral ischemia in SHR. Isradipine administered subcutaneously 30 min after permanent MCA occlusion significantly reduced the volume of ischemic brain damage in the cerebral hemisphere, cerebral cortex, and caudate nucleus when assessed at 24 h post-MCA occlusion. In SHR undergoing transient (2 h) MCA occlusion, isradipine administered 30-min post-MCA occlusion significantly reduced hemispheric infarct volume, whereas isradipine administered at the onset of reperfusion did not induce neuroprotection and did not counter functional deficit (measured using a subjective neurological scoring system) induced by MCA occlusion (151). The activity of AE0047, Ca2+ antagonist, in countering brain damage of SHR-SP with signs of stroke was compared with that of nicardipine and hydralazine. The drug (1 and 3 mg kg-1 day-1) given daily to diseased SHR-SP prevented mortality and improved neurological symptoms. Nicardipine (10 mg kg-1 day-1) and hydralazine (10 mg kg-1 day-1) were less effective than AE0047 in a dosage equal to or higher than the hypotensive dose, respectively. AE0047 may be beneficial for treating the acute stage of stroke in humans because of its long-lasting hypotensive action and undefined direct actions on the cerebral vasculature (152).
< div class='tao-gold-member'>
Only gold members can continue reading. Log In or Register to continue
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree