CHAPTER 4 Round Cells
The list of processes included in the discrete round cell neoplasm varies in the literature because of either including or excluding melanocytic neoplasms from the group. Since many of the cutaneous melanocytic neoplasms have a prominent round cytologic presentation, these are often included. A brief comment on the clinical and cytologic presentation of melanoma is included in this chapter. The entire group of discrete round cell neoplasms follows3,4,8,9,20,21:
TRANSMISSIBLE VENEREAL TUMORS
Presentation and Biological Behavior
Transmissible venereal tumors (TVTs) were the first described transplantable neoplastic process. They are more commonly seen in more tropic urban areas where there are large numbers of free-roaming dogs. Young sexually active dogs are most commonly affected because of direct contact and transplantation of neoplastic cells; external genitalia are most commonly affected. The typical social behavior of dogs licking and sniffing result in transplantation in other locations including the oral and nasal cavities. The TVT is considered mostly to be a benign neoplastic process that, with the aid of a competent immune system, will often spontaneously regress; however, metastatic disease, particularly to the intraocular space, may rarely occur.10,26
Cytologic Presentation
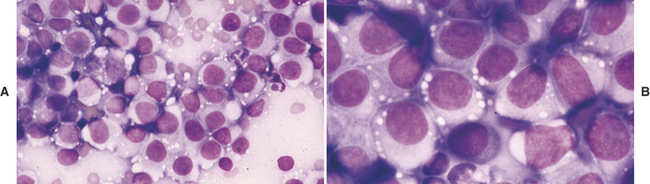
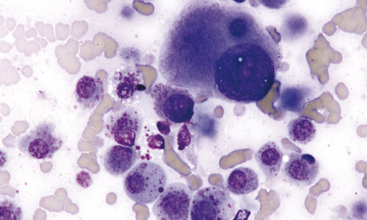
Aspirates and touch preparations of a TVT generally yield a large number of neoplastic cells. The typical TVT cell is the epitome of the discrete round cell neoplasm group. The distinctive discrete round cells have eccentric round nuclei with uniform granular chromatin patterns and, oftentimes, a single round prominent nucleolus. Mitotic figures are common and abnormal mitotic figures may be present, but these abnormal figures are not useful in predicting biological behavior. Neoplastic cells range from 12 to 24 μm in diameter and they have moderate amounts of granular and moderately blue staining cytoplasm. Distinguishing cytoplasmic granules are not present; however, low to moderate numbers of TVT cells have distinguishing, clear, distinct, punched-out cytoplasmic vacuoles. These vacuoles are commonly similar in size and arranged in a linear array along the inner surface of the cell membrane (Figure 4-1).20,22
In addition to the neoplastic cells, normal-appearing small lymphocytes, few well-differentiated plasma cells, and rarely seen histiocytes/macrophages are commonly present.20 These cells tend to increase in relative numbers during spontaneous regression of the neoplastic process; a localized immune response is the cause for the regression.
MAST CELL TUMORS
Presentation and Biological Behavior
Mast cell tumors are among the most common cutaneous neoplasms of dogs and cats; however, they may occur at any anatomic location both as primary and secondary neoplastic disease.26 They are most commonly seen in middle aged dogs and cats, but any age animal is susceptible. In the dog, mast cell tumors are considered malignant with potential of wide-spread dissemination. A histologic grading scheme has been used to help predict biological behavior; however, even well-differentiated Grade I mast cell tumors have the potential for dissemination. Cytologic identification of metastatic or disseminated mast cell tumor, regardless of degree of differentiation, is of great value. Recently, investigators have suggested that detection of agyrophilic nucleolar organizer regions (AgNORs), which are an indirect measurement of cellular proliferation, may prove more subjective in predicting biological behavior. This staining process can be applied to both cytologic specimens and formalin-fixed and paraffin-embedded tissue sections. High AgNOR counts correlate well with poor prognosis.
The general features of the histologic grading system with canine cutaneous mast cell tumors are outlined next:10,20
Feline mast cell tumor has multiple presentations. Isolated cutaneous mast cell tumor is generally considered a benign lesion in the cat and complete excision typically proves curative; however, dissemination may even be seen in cases with suspected isolated cutaneous mast cell tumor.6,12,25 A guarded prognosis with short median survival times is reported in cats with multiple cutaneous mast cell tumors, recurring mast cell tumors and primary splenic disease. Degree of differentiation is commonly considered a poor prognostic finding; however, the literature is contradictive related to the more poorly differentiated mast cell tumor in the cat. One study found no correlation between degree of cell differentiation and prognosis.3,13
Cytologic Presentation
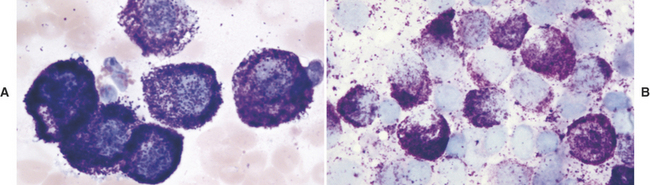
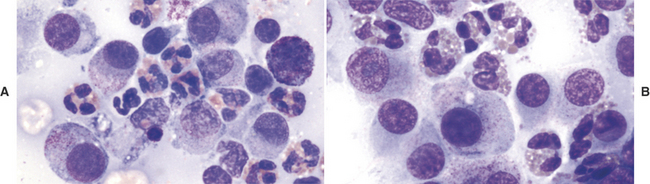
The diagnosis of mast cell tumor in the dog and cat is relatively straightforward in most cases. Cytologic specimens are commonly very cellular and peripheral blood contamination is common. In addition to the distinctive discrete round cell characteristics, mast cell tumors have distinguishing fine to coarse purple granules when stained with a Romanowsky-type stain (Figure 4-2).3 These granules allow a definitive diagnosis in most cases; however, it is reported that one of the most commonly used stains in practice, Diff-Quik, often fails in staining these granules making accurate identification of mast cell tumor from other discrete round cell tumors difficult or impossible. Anecdotal reports suggest that approximately 15% of canine mast cell tumors will not stain with Diff-Quik (Figure 4-3). In reality, if one examines the specimen very carefully, although staining may be significantly less with Diff-Quik, there are still identifiable purple granules in neoplastic cells. Additional anecdotal reports suggest that prolonged fixation of mast cell tumors in the first solution of the Diff-Quik stain improves staining of mast cell granules.
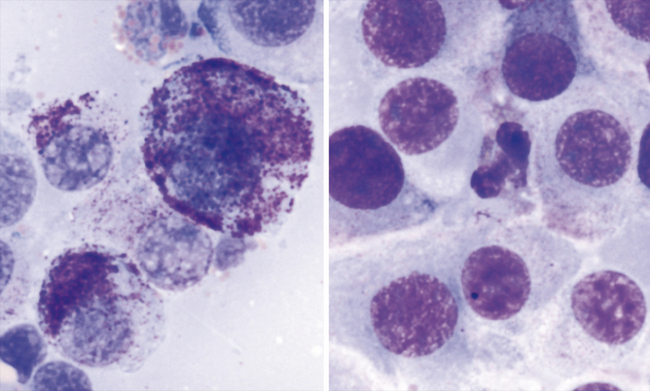
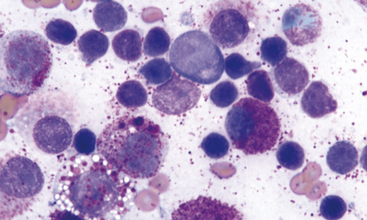
Although canine mast cell tumor grading is based upon histologic evaluation where comments on degree of tissue invasion becomes an important criterion, degree of differentiation of mast cell tumor cells is possible cytologically and correlation to histologic grading is good. The less differentiated cells are less granulated and have greater variability of cell size, nuclear size, and nuclear-to-cytoplasmic ratios. Prognosis should be considered guarded for the less differentiated canine neoplasms; however, it should be noted that well-differentiated mast cell tumors may disseminate also. Identification of metastasis to possible regional lymph nodes or parenchymal organs) proves most beneficial in characterizing the biological behavior of mast cell tumors in the dog and cat (Figure 4-4). With the feline mast cell tumor, the distribution of neoplasms (isolated or multiple or disseminated) and the degree of circumscription of the neoplasm is potentially more predictive on biological behavior than degree of differentiation (Figure 4-5).
Mast cell tumor cell nuclei are generally round to slightly oval and paracentral in location. If discernable, nuclear chromatin patterns are finely stippled and uniform; however, irregularly clumped chromatin patterns may be seen in less differentiated mast cell tumors. Nuclei are often obscured from view with heavily granulated mast cells or they are extremely pale staining due to the high affinity of the cytoplasmic granules to the stain. Eosinophils may be present in the background and these are more commonly seen in canine mast cell tumors than feline mast cell tumors (Figure 4-6). They are not predictive of biological behavior. Low numbers of normal appearing small lymphocytes may be seen in feline cutaneous mast cell tumors.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree