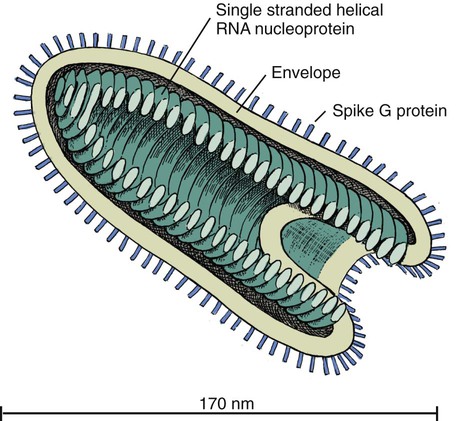
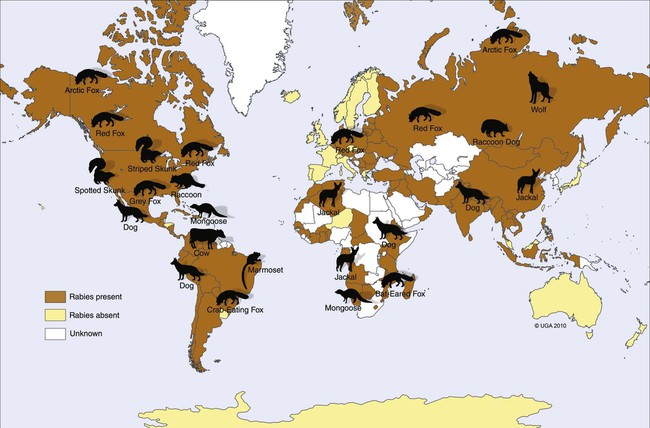
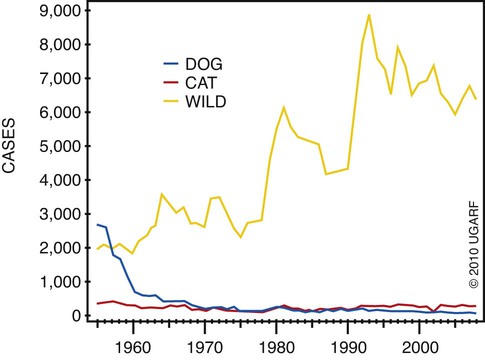
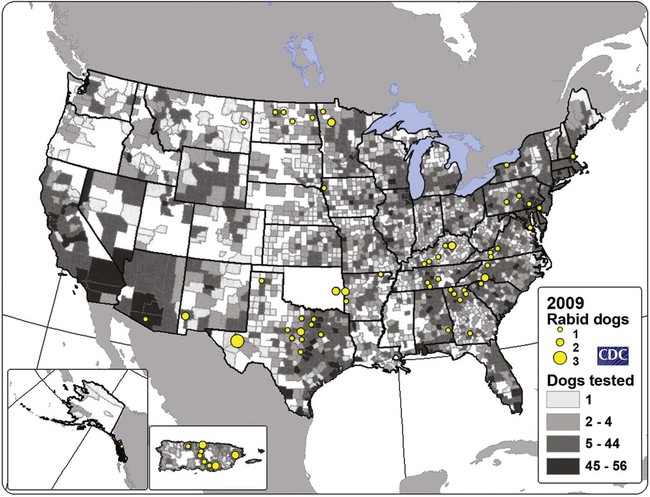
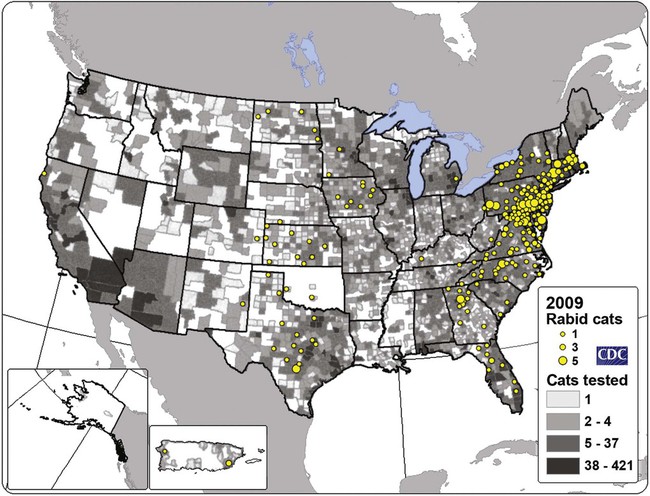
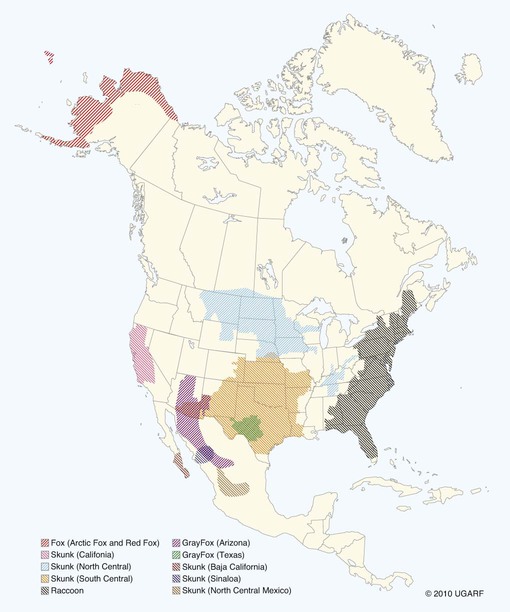
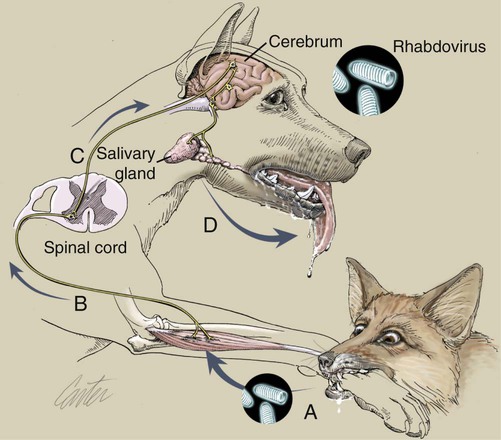
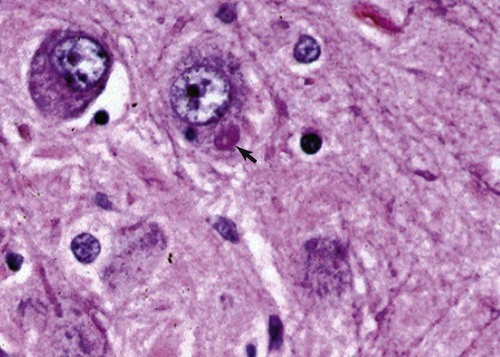
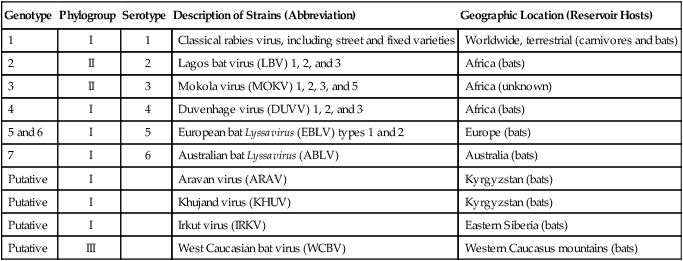
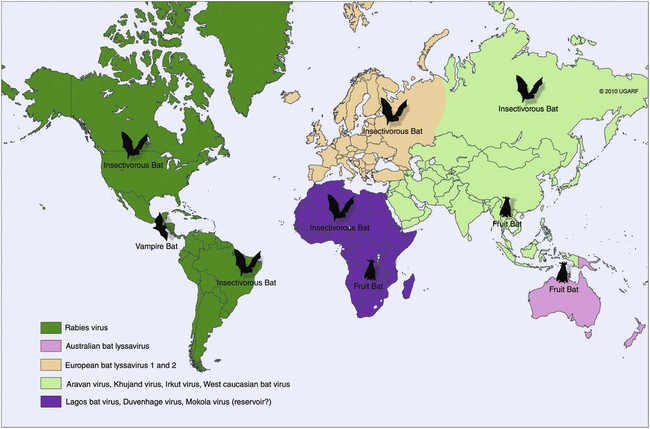
Rabies virus is the prototype of the genus Lyssavirus in the family Rhabdoviridae.89 These agents are enveloped, bullet-shaped RNA viruses that usually measure 75 × 180 nm (Fig. 20-1). The single strand of nonsegmented RNA (negative-sense) encodes five structural proteins: a nucleocapsid (N) protein, a phosphoprotein (P), a matrix (M) protein, a glycoprotein (G), and an RNA-dependent RNA polymerase (L). These viruses have been isolated worldwide and were originally considered to belong to one common antigenic type. However, techniques using monoclonal antibodies (MABs) produced against viral proteins and gene-sequencing techniques have provided evidence for antigenic and genetic differences (variants) among various isolates from major wildlife and domestic animal hosts within a given geographic region. At least seven distinct genotypes of rabies virus exist. Analysis of sequences from the nucleoprotein gene of worldwide virus isolates supports studies of MAB typing, which suggest that currently circulating terrestrial-mammal rabies virus strains may have originated in domestic dogs from south of the Indian subcontinent 1500 years ago.45 Subsequent European colonization was followed by spread to areas outside this region, such as the Americas, Asia, and Africa.314,316 For example, dog rabies virus began circulating within the past 200 years in Africa, associated with European colonial influence; it spread from east to west over the next 100 years.323 Because it has become well established, eradication will be difficult.215 Infection in imported domestic dogs may have contributed to spread to sylvatic species, with establishment of enzootic foci in wildlife within these areas.216 The regional distribution of rabies virus strains by host species and geographic location suggests that once introduced, virus spreads gradually within cohabitating animal populations. Rabies virus can remain viable in a carcass for several days at 20° C, although it may survive much longer when the body of the victim is refrigerated.221 Immunofluorescent or genetic testing, commonly used for rabies diagnosis, does not depend on the presence of viable viral particles. Viral antigen or nucleic acid may be detected at times beyond the presence of viable virus. Virus survival for isolation by mouse inoculation can be greatly increased in unrefrigerated tissue by storing it in 50% glycerol in phosphate-buffered saline at room temperature (20° C). Preservation can also be enhanced if a 20% suspension of infected tissue or virus culture is made with a solution that is high in protein or amino acids. Storage at ultralow temperatures (−30° C to −80° C) prolongs viral activity for years in untreated fresh-frozen tissue. However, freezing samples in a household-type freezer with subsequent defrosting cycles will damage the tissue and destroy the virus for subsequent detection. Besides rabies virus, at least six other Lyssavirus genotypes have been described (Table 20-1); reports of more types are expected in the future as surveillance is enhanced, particularly among bats.16 (See Other Lyssavirus Infections, under Epidemiology.) TABLE 20-1 Classification of the Genus Lyssavirus The disease is nearly always caused by the bite of an infected animal that has rabies virus in its saliva. Other modes of transmission are infrequently involved in infections of the dog and cat but may serve to maintain infection in wildlife. Transmission from exhaled or excreted virus has been suggested for spread between animals in extremely large colonies of cave-dwelling bats and by infections after laboratory exposures.124,161 Such airborne infections probably involve large quantities of aerosolized virus under conditions of poor ventilation and a susceptible exposed host. Rabies can occasionally result from ingesting infected tissue or secretions.132 Suspected transplacental rabies infections in skunks, bats, and a cow have been reported.93 Environmental transmission by fomites is rarely, if ever, involved. Human rabies is usually caused by a bite, but it has been acquired by corneal transplantation. The disturbing number of human cryptic rabies cases in which no obvious source of exposure can be determined argues against complacency with this disease. Infections with salivary shedding of virus before obvious clinical signs have been observed, and thus the absence of dramatic neurologic abnormalities cannot be used to rule out absolutely the possibility of rabies infection. More than 27,000 cases of animal rabies are reported yearly in the world. The estimated actual number of cases is many times greater. Rabid dogs are the main source of infection in people. Rabies virus transmission from dogs to people is intensified as the density of susceptible dogs exceeds 4.5 dogs/km.2,31 Combined measures of immunocontraception and rabies vaccination have been proposed to help alleviate this zoonotic risk.31,368 The World Health Organization estimates that 55,000 to 100,000 human rabies cases occur annually, most in tropical countries of Asia and Africa.* This number is equal to one fatality from rabies every 10 minutes. As a result of potential exposure to rabies virus, approximately 10 million people annually receive postexposure prophylaxis (PEP). Most affected victims in these regions are children under 15 years of age who did not receive appropriate PEP after being bitten by rabid dogs. In contrast, a major reduction in the disease incidence in the Americas has been achieved by coordinated vaccination programs for dogs. For example, in the United States, where rabies vaccination for dogs is mandatory, three or fewer humans die of rabies annually, and approximately 45,000 more receive PEP. The incidence of indigenous canine rabies has become so low that the United States was officially declared canine–rabies free in 2007.60 Canine cases that have been observed are in imported dogs. The low incidence of dog infections in North America has allowed it to be a part of the Pet Travel Scheme (PETS), similar to European Union countries, for dogs entering Great Britain.53,180 There have been similar reconsiderations of the quarantine restrictions on dogs and cats entering Japan from the United States.182 In Latin America, vaccination programs for dogs have intensified in the past 20 years and have resulted in a reduction in the annual dog-rabies virus fatalities in people from more than 150 to approximately 20.52,289a,367 Many island or peninsular nations, such as Antarctica, New Zealand, Taiwan, some of the Caribbean islands, Ireland, Norway, Finland, Sweden, Iceland, Hawaii, and Japan, are reportedly free of rabies. In western Europe, countries such as Spain, Portugal, Italy, and Greece have become free of terrestrial rabies at considerable cost through oral vaccination of wildlife, especially the red fox (Vulpes vulpes). However, there is a continual risk of reintroduction of rabies through importation of infected animals.357 Most rabid animals imported in the European Union have been entering from Morocco.252a Compulsory parenteral vaccination and serologic testing for imported domestic animals have also been instituted in some areas to help maintain this status. Although all mammals are susceptible to rabies virus, members of Canidae, Viverridae, and chiropteran species are the most capable vectors of the disease. Throughout the world, in most of the Northern Hemisphere, rabies is predominately a sylvatic disease of wildlife, whereas in the Southern Hemisphere, the feral dog in urban areas is the primary species involved in the transmission of the disease (Fig. 20-2). Turkey is the only country in Europe where dog rabies exceeds that in wildlife species and where urban dog-mediated rabies persists.177a,225 Rabies virus in a given enzootic area is usually a distinct variant that adapts itself to a single dominant reservoir host. Therefore, independent host-specific enzootic cycles of infection exist among individual host species. For example, wildlife reservoir species in various geographic areas of the United States are raccoons, skunks, foxes, coyotes, and insectivorous bats (see sections on Wild Carnivores and Bats). In Europe and parts of Asia, the primary wildlife species are foxes and raccoon dogs (Nyctereutes procyonoides), whereas in South Africa, jackals (Canis adustus and Canis mesomelas) and bat-eared foxes (Otocyon megalotis) are the predominant reservoir hosts.34,130a,260,283 In neighboring Botswana, spillover isolates in domestic animals are from jackals and the yellow mongoose (Cynictis penicillata).176 Reservoirs for rabies in Mongolia include wolves (Canis lupus), foxes (V. vulpes), and manuls (Felis manul).260 In certain Caribbean nations, mongooses (Suricata suricata) are the important reservoir host. Although bats can transmit their infection to terrestrial carnivores, the genotypes affecting bats or carnivores also remain distinct. As with rabies virus, other lyssaviruses are maintained in wildlife reservoir hosts while existing in independent manner from rabies virus. Eleven genotypes of Lyssavirus are known to exist worldwide (Fig. 20-3, and see Table 20-1): rabies virus, Lagos bat virus, Mokola virus, Duvenhage virus, two European bat Lyssavirus (EBLV) strains, and one Australian bat Lyssavirus (ABLV) type.332 Four novel lyssaviruses, Aravan, Khujand, Irkut, and West Caucasian, have also been identified.207–209 Although boasting a rabies-free status, Great Britain and Australia do have these Lyssavirus strains in bats.113,177,177 EBLV strains occasionally infect terrestrial mammals as a “spill-over.” No evidence has been found that these infections have resulted in adaptation to these inadvertent terrestrial hosts. Infected domestic animals suffer terminal illness, so the infection is not transmitted. Antibodies to EBLV-1 were detected in a domestic cat in Denmark.335 EBLV-1 infection was documented in two cats in France; bat-to-cat transmission was suspected.82 Since 1987, EBLV type 2 (EBLV-2) has been identified from Daubenton’s bats (Myotis daubentonii) in England.113,116,143,177,360 A serosurvey conducted on Daubenton’s bats in Scotland indicated between 6% and 19% of the bat sera had positive test results for antibodies to Lyssavirus,15 indicating an endemic reservoir. Foxes were experimentally susceptible to EBLV-1 and EBLV-2 by intracranial but not intramuscular inoculation, suggesting resistance to natural infection.76 Only the foxes infected intracranially with ELBV-1 developed neurologic disease. The role of these rabies-like bat Lyssavirus strains in the spread of infection to people is thought by some researchers to be marginal.365 Infections of humans with EBLV-1 have been rare, with one in Russia in 1985.178 Subsequently, a fatal human rabies caused by EBLV-2a was reported in Finland in 1985, and later another case was described in the United Kingdom in an unvaccinated bat handler.253 This latter incident was the first indigenous case of a Lyssavirus infection in a person within the United Kingdom in over 100 years. The remainder of human infections in that country have been rabies, which were acquired by foreign travel.174 Other references should be consulted for a review of worldwide Lyssavirus infections in humans.178 ABLV, or Ballina virus, has been identified in fruit bats (“flying foxes”) in Australia,120 and in addition, it has been identified in all species of wild and some captive Australian bats.111,353 Fatal infections in two people have been reported.6 Serologic evidence suggests that a closely related virus exists in the Philippines.18 Because of the close antigenic relationship of ABLV with rabies virus, cross-protection from vaccination is presumed effective.332 Experimental inoculation of dogs and cats with bat-derived Lyssavirus strains has shown a relative insusceptibility to inoculation in comparison with terrestrial rabies virus strains.232 Although outcomes have varied depending on the strains used and age of animals studied, transient or mild clinical signs with prolonged incubation periods have been observed. Seroconversion has been observed after inoculation; however, lesions within nervous tissue have been mild, and cultivable virus, viral antigen, or nucleic acid has been difficult to detect. These findings, which have also been observed in other carnivores and naturally infected humans, suggest differences in mammalian infections with bat-derived Lyssavirus compared to terrestrial strains. The highest incidence of dog and cat rabies in the United States generally occurs in areas where wildlife rabies is endemic. Most dogs and cats are infected with the predicted terrestrial rabies virus variant associated with the dominant wildlife reservoir host in their respective geographic region.236 Although the prevalence of wildlife rabies has been on the increase, cases of rabies in dogs and farm animals have been decreasing (Fig. 20-4). Vaccination of dogs and animal control programs have been the main factors responsible for this decline. Although cases of dog rabies have declined, dogs account for the majority of reported animal bites in the United States (see Bite Wound Infections, Chapter 51). Many of these bites result in people seeking antirabies prophylaxis. Despite the general elimination of rabies in dogs, the dog-virus variants have been identified as having established some foci in wild terrestrial carnivores.340 As described earlier, dog-virus variants spilled over into jackals and mongoose in Africa. In the United States, Texas had a transient spike in the occurrence of dog rabies because of infection with a particular domestic dog–coyote strain of rabies virus in coyotes (Canis latrans) beginning in the late 1980s (Fig. 20-5).205 An oral vaccination program was initiated to thwart the spread of this variant; there have been no reports of its occurrence since 2004, indicating its elimination from the United States.* Worldwide, domestic and feral dogs account for most of human rabies deaths and PEP.64 In less developed nations, where dog rabies has not been controlled, the prevalence of canine and human rabies is quite high.72,382 Adequate vaccination of at least 50% to 70% of dogs in a given population may be necessary to block the occurrence of rabies epidemics.78 Most rabies cases in dogs in dog rabies-free countries, such as the United States or in the European Union, have been in imported pups destined for commercial sale or for humane rescue.59,116,121,235,317 Because of the 4- to 8-week incubation period, clinically healthy dogs from dog rabies-endemic countries may be incubating infection. United States federal importation laws require vaccination of dogs 12 weeks of age or older from rabies-endemic areas; however, unvaccinated puppies must be admitted with owner confinement of 30 days. Vaccination restrictions are waived for dogs from rabies-free countries. Unfortunately, laxity in these importation policies has resulted in lack of compliance and an increasing number of documented imported rabies in young dogs within the United States.57,58,227,235 An increase in cases of rabies in cats usually is related to spillover of infection from wildlife because no specific virus variant attributed to cats has been reported. The relative importance of rabies in cats as a source for human exposure in a given geographic area depends on whether canine rabies is being controlled by vaccination. In the United States, since 1979, rabies in cats has shown a slight increase over the previous 7-year period. Beginning in 1981, more cases of rabies in cats than in dogs have been reported annually. This increase probably reflects the low number of cats vaccinated for rabies, community tolerance of feral cats, and the epidemics of wildlife rabies in the mid-Atlantic and Northeast regions of the United States (Fig. 20-6). Numerically, cats have been the most important domestic animal affected since 1992, with 200 to 300 cases annually.42,286,286 The frequency of human rabies exposures attributed to rabid cats is now increasing at a greater rate than that related to dogs. Rabid cats, which usually are reclusive, often become aggressive and may attack humans and other animals when disturbed.100 The striped skunk (Mephitis mephitis), the most common skunk in the United States, is one of the most important species in perpetuating wildlife rabies in the central regions of the United States (Fig. 20-7). Studies based on antigenic typing have demonstrated the existence of at least three distinct variants in skunks: one in the south central states, another in the north central states extending into Canada, and another in California. Striped skunks are commonly found to range in urban-suburban-wildland interfaces and interact with many other carnivorous species, making them prominent risks for becoming infected and transmitting rabies to other animals and humans.356 Most frequent behaviors reported in rabid striped skunks were appearing outside during the day, entering dog pens, and attacking pets.261 Although the spotted skunk (Spilogale sp.) presented a serious rabies threat in the western United States during the 1800s, the involvement of this small, secretive animal is relatively minor today. Foxes are important reservoirs in the ecology of wildlife rabies throughout the Northern Hemisphere, although they account for relatively few human exposures. In North America, fox rabies occurs throughout the range of the red fox (V. vulpes), the gray fox (Urocyon cinereoargenteus), and the Arctic fox (Alopex lagopus). An outbreak occurring among red foxes in 1990 in upstate New York was probably an extension of a Canadian fox epidemic. Coyotes have a role in the maintenance of the disease in southern Texas. A focus of another variant of rabies virus in gray foxes has been found in Arizona and Texas. The prevalence of the disease seems to decline when fox populations are reduced to levels in balance with available resources through either natural mortality or fox population-reduction programs. Oral vaccination programs, using modified live-virus (MLV) or recombinant vaccine placed in bait, had considerable success in control of red fox rabies in Europe and Ontario, Canada, and gray fox and coyote rabies in Texas.318 Despite wildlife vaccination in Europe, the nidus of infected foxes from Eastern Europe maintains the infection cycle.273 Emergence of dog rabies in northern Israel has been attributed to acquisition of the virus from rabid foxes.85 In the mid-Atlantic coast states, a major epidemic began in the late 1970s caused by the apparent translocation of infected animals from the Southeast. The mid-Atlantic outbreak spread throughout the northeastern states and southward into North Carolina by the mid-1990s. Foci from this and the Florida–Georgia–Alabama epidemic have now merged. The epidemic had spread northward along the Atlantic coastal states up into Canada.10,42,42 Raccoons have adapted well to suburban and semiurban environments; thus the number of rabies cases in cats and dogs and other domestic animals in the Northeast has dramatically increased.46 Rabid raccoons have even been identified in urban centers.218 The danger of human exposure to rabid raccoons has also increased, and at least one human death directly associated with rabid raccoons has been confirmed in Virginia. Exposure of people to rabid domestic animals such as the cat, which commonly become infected by raccoon attacks, has resulted in the greatest public health risk. Oral bait vaccination of raccoons in these regions has been shown to be effective in reducing the spread of rabies within the zone of immunization.281 Raccoons may be occasionally co-infected with canine distemper virus, which may alter the immune response within the nervous system and confuse the diagnosis as to the cause of neurologic dysfunction.134 Lyssaviruses have been identified in bats in the Americas, Africa, Australia, and Eurasia. Because of their mobility and the opportunity for bats to infect new areas, no geographic region can be truly considered free of lyssaviruses.44 Rabies in North American insectivorous bats was first recognized in the early 1950s, but studies suggest that rabid bats were in this region much earlier. The range of bat rabies is widespread throughout North America. In the United States, less than 1% of randomly caught bats are infected; however, 5% to 15% of dead or ill bats submitted for examination may have rabies.23 Bats that interact with humans are more likely to have rabies than those avoiding people; bats that bite people have the highest prevalence.270 Rabid bats may develop paresis or paralysis. They may be disoriented and fly into obstacles. Aggression has been observed in some cases. In the United States, most wild bats are insectivorous. Subclinical infection has been suggested in fruit bats in zoologic gardens in Europe, but additional investigation is needed.285,304 Although multiple cocirculating variants of rabies virus can be found in insectivorous bats, most submissions and rabid bats are from only a few of the common species (primarily Eptesicus fuscus, Myotis lucifugus, Lasiurus borealis, and Tadarida brasiliensis). Phylogenetic studies have shown that bat rabies virus variants found in terrestrial animals are distinct, and variants identified in different bat species are also quite distinct. Uncommonly, bat rabies virus variants have crossed over into terrestrial mammals, with secondary transmission.220 For example, since 2001 a bat rabies virus variant has spread into skunks and foxes in northern Arizona.15a,66 From 1980 until 2007, 61 of the 178 total cases of human rabies in the United States and Canada were caused by the bat rabies virus variant.90 Bat variant rabies cases have become more predominant than terrestrial animal variant cases with each successive year.90 Brain tissues from most of them have been examined by MAB or gene-sequencing techniques, or both. Variants associated with insectivorous bats were identified most frequently and usually were associated with the silver-haired bat (Lasionycteris noctivagans) or eastern pipistrelle (Pipistrellus subflavus), but victims had no history of bite. Bats are small and cause tiny lesions, and people may not realize that exposure has occurred.124,240,241,313 The number of humans who develop rabies from nonbite exposure is higher for contact with bats as compared with terrestrial carnivores; however, public education on nonbite transmission is needed because of the perceived greater concern and prophylaxis associated with bite lesions.33 Despite these reports in people, few authenticated cases of rabies transmission have been reported from insectivorous bats (E. fuscus) to cats and even fewer cases of transmission to dogs.268 Rabid bats seldom attack; bat bites usually occur from bats found paralyzed or semiparalyzed or from normal-appearing bats found in buildings. Vampire bats, which feed exclusively on blood, are a major rabies threat to people and animals in Mexico, Central America, and parts of South America. More than 100,000 cases of rabies in cattle attributed to vampire bats occur annually in Latin America. The routine nightly feeding of vampires makes them extremely effective in transmitting rabies virus, and the presence of rabies in vampire bats parallels that seen in insectivorous bats and terrestrial animals. The vampire bat is not found in North America, except for Mexico. Use of the same cave by vampires and other species of bat may be a source of transmission between species. Genetic characterization of rabies isolates from vampire bats and dogs in Brazil indicated two independently maintained cycles of infection.162 A focus in Colombia in domestic dogs and humans was found to be caused by virus variants found in insectivorous bats.268 In addition, an independent focus in marmosets (Callithrix jacchus jacchus) has been associated with human infections.106 The prevalence of clinical rabies among rats, mice, squirrels, and rabbits and hares is very low.99 Rodents and rabbits account for a high percentage of animal bites to people, but no cases of human rabies have ever been associated with these species, probably because they are extremely susceptible to infection and generally will not survive the attack by a rabid carnivore. For such reasons, these species are not routinely examined for rabies in public health laboratories (see Exposure Incident under Postexposure Prophylaxis for People). In the United States, rabies has been reported among large rodents such as woodchucks (Marmota monax) and beavers (Castor canadensis); most cases have been in the eastern states where raccoon epizootics exist.14 Therefore, unprovoked encounters with large aggressive rodents should be considered as a possible source of rabies exposure. The incubation period is influenced by the age of the bitten individual, the degree of innervation of the bite site, the distance from the point of inoculation to the spinal cord or brain, the variant and amount of virus introduced, PEP, and other factors. The virus may be undetectable in local tissues after the bite, and it does not enter the blood. Rabies is unique in that the incubation period, which is relatively prolonged compared with that of other infectious diseases, is primarily a result of the route of virus entry into and spread within the central nervous system (CNS) (Fig. 20-8).305 After IM inoculation, virus may enter peripheral nerves directly or replicate locally in nonnervous tissue. Virus may enter neuromuscular junctions and neurotendinal spindles after a variable period of days, weeks, or months.61 Rabies virus glycoprotein has homology to certain neurotoxins and attach to axon terminals through lipoprotein receptors, including those for acetylcholine. Virus spreads passively by intra-axonal flow in peripheral nerves at a rate of up to 100 (range, 10 to 400) mm per day. Both motor and sensory fibers may transport virus. The greater the degree of innervation at the site of the bite, the shorter the incubation period. In naturally occurring cases of rabies, ranges of incubation periods before CNS signs have been reported to be 3 to 24 weeks (average, 3 to 8 weeks) in dogs, 2 to 24 weeks (average, 4 to 6 weeks) in cats, and 3 weeks to 1 year or more (average, 3 to 6 weeks) in humans. Rabies virus variants may vary in their ability to be transmitted by nonbite means. In comparing the dog and coyote strains with silver-haired bat viruses, the latter was better able to infect epithelial cells, replicate at lower temperatures, and have neuroinvasive tendencies.122,144,144 Interneuronal spread of virus corresponds to the progression of clinical signs that are noted. The virus enters the spinal cord or brainstem ipsilateral to the site of initial virus inoculation by retrograde axoplasmic flow. Once in the CNS, virus spreads by intra-axonal means to involve the contralateral neurons and ascends rapidly and bilaterally in the spinal cord or brainstem to the forebrain. In naturally infected dogs, virus in the brain preferentially localizes in the limbic areas, thalamic nuclei, reticular formation, and trigeminal and vagal nuclei.319 Damage to the motor neurons causes progressive lower motor neuron (LMN) disease, which, in turn, produces the typical ascending flaccid (hyporeflexic) paralysis of rabies. Damage to the CNS caused by rabies virus has mainly been attributed to direct viral invasion of the nervous system. Damage to neural tissue is visibly limited in comparison to the severe degree of paralysis; inhibition of function or synthesis of neural transmitters is suspected. Apoptosis, or genetically induced premature cell death, may be important in the limited amount of observed neuronal necrosis.165 Host immune responses to rabies virus may accentuate the inflammation and degeneration of nervous tissue. Interference with cardiorespiratory control results in death. Recovery from rabies has been exceedingly rare.166 In many instances, demonstrating the presence of virus may be difficult. During the early incubation period, rabies virus may be sequestered at the site of inoculation while replicating in myocytes and nerves. The long period between exposure and clinical signs may be the result of local replication and has been associated with high titers of anti–rabies virus antibody in the cerebrospinal fluid (CSF) and CNS tissue. Adequate serum titers of anti–rabies virus antibody, acquired by active or passive immunization, have been correlated with protection against infection and restricted viral replication.175 Effective cell-mediated immunity is essential to the eventual elimination of rabies virus. Recovery should be regarded as of extremely minor importance in the epidemiology of the disease and not relevant in public health considerations. With appropriate intensive medical management and passive immunotherapy humans have recovered (see Exposure Incident, under Postexposure Prophylaxis for People). The furious or psychotic type of the disease in dogs usually lasts for 1 to 7 days and is associated with forebrain involvement. Animals become restless and irritable and have increased responses to auditory and visual stimuli. They frequently become excitable, photophobic, and hyperesthetic and bark or snap at imaginary objects. As they become more restless, they begin to roam, usually becoming more irritable and vicious. Dogs may eat unusual objects (pica), especially wood, that become gastrointestinal foreign bodies. They may avoid contact with people and prefer to hide in dark or quiet places. When caged or confined, dogs often try to bite or attack their enclosure. They usually develop muscular incoordination, disorientation, or generalized grand mal seizures during this phase. If they do not die during a seizure, they may experience a short paralytic stage and then die (Fig. 20-9). Cats can develop more consistently the furious phase of the disease, showing erratic and unusual behavior. These cats are described as having anxious, staring, wild, spooky, or blank looks in their eyes.112 When confined in cages, they may make vicious, striking movements and attempt to bite or scratch at moving objects. In addition, they may have muscular tremors and weakness or incoordination. Some cats may run continuously until they seem to die of exhaustion. The paralytic or dumb type of rabies usually develops within 2 to 4 days (range, 1 to 10 days) after the first clinical signs are noted. LMN paralysis usually progresses from the site of injury until the entire CNS is involved. Cranial nerve paralysis may be the first recognizable clinical syndrome if the bite occurs on the face. When the brainstem becomes affected, a change in the tone of the bark, resulting from laryngeal paralysis, may be observed. Dogs, which more commonly show this type of disease, may begin to salivate or froth excessively as a result of the inability to swallow and the deep labored respiration that occurs. A “dropped jaw” develops as a result of paralysis of the masticatory muscles (Fig. 20-10). Dogs may make a choking sound, which convinces an owner that something is caught in the animal’s throat. Owners or veterinarians may then become exposed to the virus in the saliva while attempting to remove a suspected foreign object. The course of the paralytic phase usually lasts 2 to 4 days. The animal often goes into a coma and dies of respiratory failure. The paralytic disease in cats often follows the furious form of the disease and begins around day 5 of clinical illness. Although the total course of illness may last 10 days, rabid cats often die after 3 to 4 days.295 As in dogs, initial paralysis of the bitten extremity can progress to paraparesis, incoordination, and ascending or generalized paralysis, terminating in coma and death. Mandibular and laryngeal paralysis is less common in cats. Increased frequency of vocalization is a commonly reported sign in cats, and owners often recognize a change in the pitch of the cat’s voice.112 Cats occasionally develop the paralytic form directly after the prodromal phase with few or no signs of excitement. Atypical, abortive forms of rabies virus infection with recovery may occur but are considered very rare phenomena. Clinically healthy carriers have not been found in domestic dogs within enzootic areas.380 Experimentally infected dogs that developed acute progressive LMN paralysis have shown clinical improvement a few days to months later. Survival with chronic infection has been reported to rarely occur after experimental rabies infection in cats, but clinical recovery from paralysis has not been observed. The clinical syndrome of rabies in people is similar in duration and variability to that in dogs and cats. Genetic studies of viral isolates from human rabies indicate that clinical manifestations such as encephalitic or paralytic rabies are not determined by different virus variants but by other factors such as the site of the inoculation and spread of virus within the nervous system.150 Fever, headache, anxiety, nervousness, and hyperesthesia at the bite site have been reported. As the syndrome progresses to the excitable phase, clinical signs consist of excitability, restlessness, hyperkinesis, and violent behavior. Humans salivate incessantly and may refuse to drink water. They experience painful pharyngeal spasms when attempting to swallow fluids, which gives rise to the term “hydrophobia.” As disorientation and excitability continue, some patients die in convulsive episodes, whereas others develop generalized LMN paralysis and respiratory arrest. Although the rare instance of recovery has been reported after extraordinary efforts, the disease is considered to be invariably fatal after the onset of clinical signs. For a review of the diagnosis of rabies, also see the Centers for Disease Control and Prevention (CDC) website at www.cdc.gov/rabies/. The definitive diagnostic test is the demonstration of rabies virus antigen by the direct fluorescent antibody (FA) test in suitable brain tissue.30 Direct immunochemical methods using light microscopy have given comparable accuracy.217 No premortem diagnostic tests are sensitive enough to be consistently reliable for rabies diagnosis in animals. Nevertheless, there may be some limited indications for testing serum, CSF, or biopsy specimens before the death of the animal. No hematologic or serum biochemical changes are characteristic or specific for rabies. Biochemical changes in CSF have been minimal in experimentally infected dogs and have been rarely reported in natural infections. Increased CSF protein (110 to 150 mg/dL) and leukocytes (120 to 1140 cells/µL), with small lymphocytes predominating, have been reported in dogs with postvaccinal rabies encephalomyelitis.29 Cats with postvaccinal rabies also have had increased CSF protein (55 to 80 mg/dL) and increased CSF lymphocyte count (5 to 17 cells/µL). This method has been predominantly used to diagnose rabies antemortem in people. Because virus enters extraneural tissues via outward spread from the CNS, it arrives at nerve endings in the skin and at the salivary glands simultaneously. Because of the heavy sensory innervation, the skin at the nape of the neck (in humans) and the sensory vibrissae on the maxillary areas (in animals) may be selected for direct FA testing. Direct FA testing of a skin biopsy has a 25% to 50% probability of being positive about the time that clinical rabies develops; accuracy is increased as the disease progresses. Of all dogs and cats tested that were confirmed to be rabid by positive results of brain immunofluorescence, neurologic signs developed within 10 days of the biting incident.328 Inactivated or recombinant rabies vaccines commonly used in dogs and cats do not give false-positive results. The skin biopsy technique should never be substituted for brain examination of an unvaccinated animal with suspect neurologic signs. This test appears to be accurate if the virus is present, but a negative test result does not rule out the possibility that the animal is infected. In humans, polymerase chain reaction (PCR) testing of skin has shown much higher degrees of sensitivity and specificity compared to direct FA methods.83 Use of these methods is restricted at present to human diagnostics and has not been approved for routine laboratory diagnosis of rabies in animals. Salivary tests for rabies virus have been used as one test to diagnose rabies in people. Rabies virus has been detected in dog saliva by slide agglutination using latex particles coated with polyclonal immunoglobulin.184 The load of viral particles in saliva is lower than it is in brain tissue, and negative results must be confirmed by more reliable means such as the direct FA procedure on brain. Increased sensitivity and specificity of saliva testing can be achieved by using virus isolation or genetic detection methods (see later discussion). Serologic tests are rarely used for epidemiologic surveys or for diagnosis because of the low percentage of surviving animals that have time to develop postinoculation antibody. Serologic tests are used to determine vaccine immunogenicity. Some countries require a positive antibody titer for importation.9 Mouse inoculation was performed historically for serologic testing but has been replaced by cell culture methods. The rapid fluorescent-focus inhibition test (RFFIT), can quantify concentrations of specific rabies virus antibody in serum. Other tests for rabies virus antibodies, based on enzyme-linked immunosorbent assay (ELISA, or fluorescent antibody virus neutralization [FAVN] test), have been proposed to augment the RFFIT for serodiagnosis or support of immunization. Comparative testing under field conditions in dogs indicates that ELISA test results are higher than those with RFFIT, making ELISA methods less reliable for estimating adequate seroconversion after vaccination.27,190a Improvements in ELISA methods, which overcome some inaccuracies, have been described.379 During the incubation period of rabies, antibody responses are not usually observed, and the virus may be hidden from the immune system. After neurologic signs develop, antibodies appear in serum and later in the CSF.276 Testing dogs or cats for serum antibodies to rabies virus to determine recent exposure to rabies virus can be ambiguous because elevated titers can result from vaccination or from past or recent exposure to virus. Therefore, a serologic response can in no way be definitively differentiated from vaccination or infection. Testing for rabies virus antibody in CSF is a possible means of documenting rabies infection because antibody is locally produced, and CSF titers may increase 2 to 3 weeks or more after the onset of clinical rabies. Because of this delay, a negative titer result does not eliminate rabies infection as a possibility. The World Health Organization and the Office International des Épizooties have developed minimum guidelines as supportive evidence of rabies immunization in animals. This measurement has been adopted as one requirement for importation of animals into rabies quarantine areas (see later discussion, Quarantine and Shipment of Animals). In an update of these guidelines, the RFFIT test has been augmented by the FAVN test.8 This latter test has become another international standard for shipment of animals. No difference has been observed in sensitivity or specificity for either method in comparing sera of vaccinated or unvaccinated animals.49,309 Modification of the FAVN using MABs and peroxidase conjugates has allowed automation of the procedure for dogs.158,262 In following antibody titers after vaccination, titer levels may vary based on the degree of similarity between the rabies viral strain used in the test system and that in the vaccine.247 Many other factors may influence the increase in antibody titer in individual dogs or cats.231 For example, the timing of serum collection after vaccination; the breed, size, and age of the animal; the number of prior boosters; whether a monovalent versus polyvalent vaccine is used; whether a dog or cat is vaccinated; and the choice of vaccine may be important determinants of the level of titer measured.187,242,242 A titer of 0.5 IU/mL has been used as the standard level expected for an adequate titer in people and animals. Although parenteral vaccination may more readily achieve this level, oral vaccination with the SAG2 strain did not always induce this level of seroconversion.297 Nevertheless, dogs showing any measurable titer after vaccination were protected. A titer of 0.5 IU/mL is required for animals exported to most rabies-free areas. No “protective” titer has been found in animals. Individual interpretation is the responsibility of the veterinarian submitting the specimen and the agency requesting the test be done. See Web Appendix 5 for laboratories that perform these assays. Selection and submission of proper specimens are critical for accurate rabies diagnosis. Handling live, suspected rabid animals must be done with extreme care. Heavy protective gloves must be worn, and catchpoles, cages, and other equipment often facilitate capture and transport of such animals. The animal must be euthanized by a humane method, and the brain must be protected from damage. The use of an ax or power saw should be discouraged when opening the skull, because these may create hazardous aerosols. A procedure to remove the brain of a suspected rabid animal has been published.333 Small specimens such as mice and kittens may be submitted whole. A technique for retroorbital removal of brain specimens for collection of material for epidemiologic studies has been described.244 Complete brain removal is still indicated when human exposure has occurred. When brain tissue has been inadvertently damaged or destroyed, the spinal cord is an alternative but less desirable substitute. A method of submitting brain tissue, dried on filter paper, has been described that permits submission of specimens without refrigeration.247 Brains from rabid and control dogs were spotted on filter paper that was dried and stored for up to 222 days. Analysis was performed by nucleic acid sequence-based amplification and reverse transcriptase-PCR (RT-PCR). This postmortem test is both rapid and sensitive and is currently the most widely used and preferred and reliable method of diagnosing potential rabies infection in animals.142 False-negative results are rare with this test when compared with those of mouse inoculation.327 Thin touch impressions of the medulla, cerebellum, or hippocampus are used for this test. In decomposing canine brains at room temperature of 25° C to 29° C, the direct FA test result remains positive in most cases for up to 96 hours; the corresponding mouse inoculation test usually becomes negative by 48 hours.5 Unless it is completely decomposed, the head should be submitted because specific fluorescence may still be detected. Rabies virus has also been detected by immunoperoxidase techniques on formalin-fixed, partially autolyzed and paraffin-embedded tissue.20,133,212,351 Animals need not show neurologic signs at the time of examination, and all animals excreting virus in the saliva will have detectable virus in the CNS by immunohistochemical examination. In natural infections of dogs virus is usually localized to the neuronal perikaryon, extending along into the dendritic regions.319 Rabies virus RNA has been detected in nervous tissue by RT-PCR.131,147,163,183,351 Amplified sequences have included the phosphoprotein, nucleoprotein, and glycoprotein genes. PCR has been used as a confirmatory test in direct FA-negative samples or in decomposed brain tissue that is difficult to evaluate with direct FA methods or virus culture,86,146,146 but the predictive ability of a negative result has not been discussed. In situ hybridization can detect rabies virus genomic RNA in paraffin-embedded brain tissues.167,351 For detection of rabies in living human patients, RT-PCR has been used on saliva, CSF, and urine,81,257,344,345 and in dogs, quantitative PCR has been used on saliva and CSF300; however, it is not considered a desirable alternative in detecting potentially rabid animals exposed to humans, in which the brain tissue is preferred for confirmation. In humans, RT-PCR analysis of saliva has been the most accurate means of antemortem diagnosis of rabies when compared with other detection methods and is as accurate as brain biopsy.257 Quantitative (real-time) PCR methods have been developed for this purpose.252 Direct sequencing of PCR products along with automated gene sequencing can also help distinguish among virus variants to determine the most probable wildlife reservoir for an isolate from human or animal infection. A quantitative PCR assay has been developed to detect rabies virus in human and animal brain specimens.346,348 Quantitative PCR has also been more accurate than other PCR assays used to detect lyssaviruses in brains from bats.117 The classic test for the presence of rabies is to examine the brain for the presence of intracytoplasmic inclusions, known as Negri bodies, in larger neurons (Fig. 20-11).206 They are most commonly found in the thalamus, hypothalamus, pons, cerebral cortex, and dorsal horns of the spinal cord. Negri bodies are most common in neurons of the hippocampus in carnivores and in Purkinje’s cells of herbivores. Negri bodies in tissue sections or impressions of brain tissue are best demonstrated with Seller’s or Van Gieson’s stains, in which they stain magenta. Unfortunately, Negri bodies take time to develop and cannot be found during all stages of infection and in all infected cats. They usually cannot be detected until neurologic signs are apparent; therefore premature killing of the animal may reduce the chances of finding these inclusions. Negri bodies may be found in approximately 50% of the samples that test positive by direct FA. In some cases, nonrabid tissues have displayed inclusions that resemble Negri bodies. Cytoplasmic inclusions that are confused with Negri bodies can be found in the brains of healthy cats.50,51 They occur in the pyramidal cells of the hippocampus and in neurons of the dorsal part of the lateral geniculate nucleus. Detection of Negri bodies is no longer used in most developed nations for routine diagnostic confirmation. Intracerebral inoculation of laboratory mice with fresh or fresh-frozen homogenized tissue is a confirmatory test for rabies but is not widely conducted for routine diagnosis of suspected rabies cases in the United States. Specific neutralizing antibody is incubated with extracted tissue before its inoculation to confirm that rabies virus is responsible for the observed neurologic signs. Brain tissues from infected mice are examined for virus by direct FA. This test does not distinguish between virulent and vaccine viruses because, regardless of attenuation, many of the virus strains produce a similar illness in mice. Replacement of mouse inoculation by viral inoculation into tissue culture is now feasible because virulent virus can now be grown in various cell lines.288
Rabies and Other Lyssavirus Infections
Etiology
Rabies
Other Lyssavirus Infections
Genotype
Phylogroup
Serotype
Description of Strains (Abbreviation)
Geographic Location (Reservoir Hosts)
1
I
1
Classical rabies virus, including street and fixed varieties
Worldwide, terrestrial (carnivores and bats)
2
II
2
Lagos bat virus (LBV) 1, 2, and 3
Africa (bats)
3
II
3
Mokola virus (MOKV) 1, 2, 3, and 5
Africa (unknown)
4
I
4
Duvenhage virus (DUVV) 1, 2, and 3
Africa (bats)
5 and 6
I
5
European bat Lyssavirus (EBLV) types 1 and 2
Europe (bats)
7
I
6
Australian bat Lyssavirus (ABLV)
Australia (bats)
Putative
I
Aravan virus (ARAV)
Kyrgyzstan (bats)
Putative
I
Khujand virus (KHUV)
Kyrgyzstan (bats)
Putative
I
Irkut virus (IRKV)
Eastern Siberia (bats)
Putative
III
West Caucasian bat virus (WCBV)
Western Caucasus mountains (bats)
Epidemiology
Susceptibility
Transmission
Hosts and Range
Other Lyssavirus Infections
Dogs and Cats
Wild Carnivores
Bats
Rodents and Lagomorphs
Pathogenesis
Entry of Virus
Spread in the Central Nervous System
Recovery
Clinical Findings
Dogs and Cats

People
Diagnosis
Detection of Virus in Dermal Tissues
Testing of Saliva for Virus
Serologic Testing
Documentation of Rabies Immunization with Antibody Titers
Pathologic Findings
Submission of Specimens
Direct Fluorescent Antibody and Immunohistochemical Testing of Nervous Tissue
Genetic Detection of Virus and Genetic Sequencing
Intracellular Inclusions
Mouse Inoculation and Virus Isolation
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Veterian Key
Fastest Veterinary Medicine Insight Engine