Chapter 32 Nutritional Strategies in Gastrointestinal Disease
Gastrointestinal Tract
Definition
The gastrointestinal tract (GIT) in dogs and cats is a dynamic organ that performs numerous functions (see Chapter 1) essential for the health and well-being of the animal. A critical function of the GIT is digestion and absorption of nutrients, as well as elimination of potentially harmful substances and waste products. In addition, the GIT is the largest immunologic organ in the body and it also functions as an endocrine organ. Nutrition is important for maintaining a healthy GIT, and it plays a key role in the management of many GIT problems.
Protein
Protein and amino acids are important for synthesis and repair of tissue, and also play a role in energy metabolism. Although all proteins are functional, protein is the second largest potential store of energy in the body after adipose tissue.1 Energy and protein needs are tied together, and amino acids from protein can be converted to glucose by gluconeogenesis. They serve as a continuing supply of glucose after glycogen is consumed during fasting. The GIT is the primary route by which protein enters the body, and protein balance in the body is dependent on both the availability of dietary protein and the ability of the GIT to digest and absorb protein. Disorders affecting the absorption of protein can rapidly deplete protein stores in the body and lead to protein malnutrition. Protein malnutrition has adverse effects on numerous functions in the body, including muscle strength, organ function, and immune function. Protein malnutrition can also result in mucosal atrophy in the GIT, further impairing protein absorption.
Dietary protein also serves as a source of food allergens. The GIT uses a variety of immunologic and nonimmunologic mechanisms to prevent foreign proteins from entering the body. Under normal circumstances, food antigen exposure via the GIT results in a local immunoglobulin (Ig) A response, activation of regulatory lymphocytes that reside in the gut-associated lymphoid tissue (oral tolerance), and systemic immune response.2,3 However, in some incidences there is an abnormal interaction between food allergens, the GIT, and the immune system that can result in some GIT disorders such as inflammatory bowel disease (IBD) and acute enteritis. (The reader is referred to Chapter 31 for additional information on adverse reactions to food.)
Glutamine
Specific amino acids play a key role in maintaining a healthy GIT.4 For example, the amino acid glutamine has traditionally been considered a nonessential amino acid; however, it is now recognized as a conditionally essential nutrient under stress conditions, such as starvation, infection, injury, and recovery from surgery. Glutamine is a fuel for enterocytes lining the small intestinal epithelium, providing as much as 40% of the energy needs for these cells. It is also used by white blood cells and contributes to normal immune system function, as well as being involved in critical processes such as nucleotide synthesis, protein synthesis, and gluconeogenesis. Studies show that dietary glutamine supplementation decreases susceptibility of enterocytes and lymphocytes to apoptosis while enhancing antioxidative function and cell proliferation in the small intestine. It was once believed that only luminal glutamine supported gut function and mucosal integrity. More recent studies show that supplementing total parenteral nutrition (TPN) with glutamine is also an effective route of administration for the gut.4a
Arginine
Arginine stimulates intestinal fluid secretion through a NO-mediated mechanism, and inhibition of NO synthase (NOS) leads to decreased intestinal secretion and intestinal ischemia. Arginine supplementation is also effective in improving intestinal barrier function and vascular development. Oral arginine decreases the mucosal injury caused by lipopolysaccharide endotoxemia in mice, and pretreatment with arginine enhances survival and intestinal mucosal barrier function after intestinal mesenteric ischemia. Arginine-enriched diets fed to rats also have been shown to protect the gut mucosa from injury caused by radiation-induced enteritis, accelerate healing ability, and prevent translocation of bacteria. However, it is important to keep in mind that the beneficial effects of arginine may be dose dependent. Lower levels of arginine supplementation (0.7%) have beneficial effects on microvascular development, but higher doses of dietary arginine (1.2%) can cause adverse effects, such as gut dysfunction.4b
Glycine and Lysine
Both glycine and lysine are directly utilized by the intestine for protein synthesis and other metabolic processes and may have protective effects on the gut.4 The small intestinal mucosa utilizes glycine to synthesize glutathione (GSH), and it may have powerful cytoprotective effects, such as osmoprotection, scavenging of oxygen free radicals, extracellular signaling, and modification of biologically active molecules. Although research using glycine and lysine supplementation in GIT disorders are just underway, one or both of these amino acids may be useful in the management of some GIT disorders.
Protein Digestibility
Modulation of dietary protein is often very effective in managing many types of GIT disorders. It is very important that protein used in intestinal diets be highly digestible (>87%).5 This is particularly important in conditions where protein maldigestion or malabsorption is present. Most dietary protein is absorbed in the small intestines, and unabsorbed protein that pass into the large intestines are metabolized by bacteria and produce undesirable metabolites such as gas.
Carbohydrates
Although dogs and cats do not have an absolute dietary requirement for carbohydrates, carbohydrates are a major component of most diets for dogs and cats. The predominant carbohydrate found in pet food is starch. Starch is the storage form of carbohydrates in plants, whereas glycogen is the storage form in animals. The digestibility of dietary starch is dependent upon the source and the degree and type of processing. If properly processed, corn, wheat, rice, and barley can be highly digestible (>90%).5 Other starches, such as potato and tapioca, are less digestible, especially if undercooked.
Lactose
Lactose is the primary carbohydrate found in milk, and it is broken down by the brush-border enzyme lactase. Puppies and kittens normally have adequate levels of intestinal lactase to permit digestion of lactose in the mother’s milk. However, lactase activity often decreases after weaning, and some adult dogs and cats are lactose intolerant. If these animals consume dairy products, such as milk, osmotic diarrhea often results. In humans with lactose intolerance, some studies have shown that the use of probiotics decreases or suppresses symptoms of lactose intolerance.6–8 Some lactic acid bacteria produce enzymes that hydrolyze lactose, thereby alleviating clinical signs. The majority of the research in this area has been done in humans, and it is unknown at this time whether similar beneficial effects occur in dogs and cats. Carbohydrate intolerance also develops secondary to enteritis, and inadequate disaccharidase activity may be one of the factors responsible for diarrhea associated with rapid changes in the diet.
Fiber
Introduction of Dietary Fiber
Dietary fiber was originally defined as “the remnants of plant cell walls not hydrolyzed by the alimentary enzymes of man,” but the definition was subsequently modified to include all plant polysaccharides and lignin, which are resistant to hydrolysis by digestive enzymes.9 More recently, the definition of fiber has been modified further and is now defined as the composite of all dietary constituents that are not digested by endogenous enzyme secretions in mammals. Although not digestible, dietary fiber is considered to have nutritional value because of its importance in maintenance of the functional integrity of the GIT.9a
Dietary fiber consists of material of diverse chemical and morphologic structure. Large differences exist in the physical form and the physiologic effect of various classes of dietary fiber in dogs and cats, and it is now recognized that specific fiber types can be utilized for specific effects on the GIT. Major components of dietary fiber include nonstarch polysaccharides, cellulose, hemicellulose, mixed-linkage β-glucans, pectins, gums, and mucilages. Lignins are also included in the estimates of total dietary fiber because they are plant cell wall constituents that can greatly affect the digestibility of plant-derived foods.10 Quantitatively, lignins do not make a significant contribution to total dietary fiber intake unless intact seeds are consumed.
The diverse nature of fiber has led to numerous ways of classifying fiber, including by solubility in water, rate of fermentation, digestible and indigestible fractions, water-holding capacity, viscosity, fecal-bulking ability, cation exchange capacity, bile acid–binding ability, and microbial fuel value.11–14
In the past, dietary fiber has been classified by its solubility (soluble vs. insoluble). This classification is based on how fiber reacts with water.11 All fibers hold water to some degree; however, the soluble fibers have a greater water-holding capacity than insoluble fibers, and they may form gels and viscous solutions in the GIT (Table 32-1). In more recent years, this classification of fiber has fallen into disfavor. Categorization of fiber types based on fermentability (Table 32-1) is a more meaningful way to describe certain fiber sources for dogs and cats because the fermentability or the capacity for fiber breakdown by intestinal bacteria more accurately assesses fiber’s potential beneficial effects in the GIT than does solubility.
Table 32-1 Dietary Fiber Fermentation in Dogs15
| Fiber Type | Solubility | Fermentability |
|---|---|---|
| Beet pulp | Low | Moderate |
| Cellulose | Low | Low |
| Rice bran | Low | Moderate |
| Gum arabic | High | Moderate |
| Pectin | Low | High |
| CM-cellulose | High | Low |
| Methylcellulose | High | Low |
| Cabbage fiber | Low | High |
| Guar gum | High | High |
| Locust bean gum | High | Low |
| Xanthan gum | High | Moderate |
| Inulin | High | Moderate |
| Psyllium | Both | Moderate |
Beneficial Effects of Fermentation of Fiber by Intestinal Bacteria
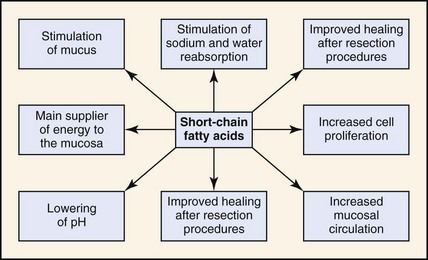
Fiber usually passes through the stomach and small intestines intact in dogs and cats because they do not endogenously produce the enzymes needed to digest fiber. Until recently, fiber fermentation was thought to be irrelevant in dogs and cats. However, once fiber reaches the large intestines, intestinal bacteria are able to ferment certain types of fiber, resulting in the production of short-chain fatty acids (SCFAs). As a result, fermentation of fiber is very important in dogs and cats. The major SCFAs produced from fermentation are acetate, propionate, and butyrate. In ruminants and herbivorous animals, SCFAs provide a significant source of energy (i.e., up to 75% of daily energy requirement [DER]) in these species. However, dogs and cats have a relatively short and simple structure of the large intestines, and as a result, SCFAs provide less than 5% of energy needs and have little effect on energy balance.16,17
Although production of SCFAs in the large intestines of dogs and cats provides very little energy, it does have a number of beneficial effects (Fig. 32-1). Some of the beneficial effects of SCFAs include the following:
1. An energy source for colonocytes.18 Colonocytes derive more than 70% of their energy needs from luminally derived SCFAs. As a result, fermentation of fiber provides a readily available source of energy for colonocytes that aids in maintaining the health and function of these cells. SCFAs are also important for cell renewal and repair. Epithelial cells of the GIT turnover rapidly and must be replaced on average once every 3 days. By providing energy for intestinal cells, SCFAs facilitate the replacement of cells that have been sloughed during the normal process of cell turnover.
2. Maintenance of normal intestinal electrolyte and fluid balance.19 SCFAs facilitate the absorption of sodium, chloride, and water in the colon. For example, a study in dogs showed that sodium and SCFA absorption could account for the entire osmotic absorption of water from the colon. As a result, providing fermentable fiber in the diet is essential in maintaining the normal homeostatic absorptive function of the intestine of dogs.
3. Maintenance of ileal and colonic motility.20 Normal ilead motility appears to be influenced by the presence of SCFA. Kamath et al. infused bolus doses of physiologic concentrations of SCFA into the ileum of dogs. As the dose of SCFA increased, ileal motility increased. Therefore providing fermentable fiber in the diet may be important in maintaining normal ileal and colonic motility.
4. Amelioration and prevention of pathogenic bacterial overgrowth.21 Harmful bacteria (e.g., Clostridium, Salmonella, enterobacteria) can produce (a) toxins, (b) carcinogens, and (c) putrefactive substances.22 Beneficial bacteria (e.g., bifidobacteria, lactobacilli) (a) inhibit the presence of harmful bacteria, (b) stimulate immune function, (c) aid in digestion and/or absorption of food, and (d) synthesize vitamins. Maintenance of beneficial indigenous bacterial populations is important in prevention of pathogenic bacterial overgrowth in the intestine. Indigenous bacterial populations in the dog or cat GIT ferment certain fiber sources that result in the production of SCFA.17,23 The presence of SCFA inhibits the growth of pathogenic bacteria.24,25 As a result, not only are indigenous bacterial populations necessary for the production of SCFA, but they can also directly inhibit pathogenic bacterial overgrowth in the gut. A growing body of evidence supports the theory that certain dietary fiber sources can modify the composition of the intestinal microflora.
5. Maintenance of optimal colonic morphology.26,27 Dogs fed a fermentable source of fiber had increased colon weight, increased mucosal surface area, and mucosal hypertrophy compared with dogs fed a nonfermentable fiber source (cellulose). These effects on the colon also aid recovery after intestinal surgery.28
6. Amelioration of intestinal inflammation.29 Diets containing nonfermentable fiber (cellulose) as the sole source of dietary fiber fed to dogs resulted in a higher incidence of mucus distention and cryptitis when compared with similar diets that contained a fermentable fiber source.
All Fermentable Fibers Are Not Created Equal
Fiber sources can vary in their level of fermentability (see Table 32-1). As fermentation rate of fiber increases, gastrointestinal transit time decreases, fecal bulk decreases, and fecal bile acid excretion increases. Fibers with low fermentability (e.g., cellulose, methylcellulose, oat fiber, peanut hulls, xanthan gum, locust bean gum) are not metabolized well by intestinal bacteria to produce SCFAs. Rather they retain their structure while passing through the GIT intact and act as bulking agents. Highly fermentable fibers (e.g., pectin, guar gum) are rapidly metabolized by intestinal bacteria. One of the products from bacterial fermentation of fiber is SCFAs. However, less-desirable substances are also produced from bacterial fermentation, including carbon dioxide, hydrogen, and methane. If a fiber source is rapidly fermentable, large amounts of gases will be rapidly produced in the colon, resulting in diarrhea, flatulence, and cramping. Moderately fermentable fiber sources (e.g., beet pulp, rice bran, gum arabic, xanthan gum) produce SCFAs without resulting in rapid production of gases and associated diarrhea. Therefore, moderately fermentable fiber produces the beneficial effects associated with production of SCFAs without the undesirable effects seen with rapidly fermentable fiber. An ideal fiber source for dogs and cats should contain a moderately fermentable portion to facilitate SCFA generation, as well as a nonfermentable portion to provide bulk and enhance peristalsis.29a
Nomenclature of Fat
Fatty acids are classified as short-, medium-, or long-chain fatty acids. SCFAs contain fewer than six carbons, medium-chain fatty acids (MCFAs) contain six to 12 carbons, and long-chain fatty acids (LCFAs) contain more than 12 carbons. LCFAs are the predominant type of fat consumed in the diet by dogs and cats. When LCFAs are absorbed from the GIT, they first enter the lymphatics before eventually making their way to the blood via the thoracic duct. Generally, once SCFAs and MCFAs are absorbed in the GIT, they bypass the lymphatics and instead directly enter the portal vein.30 However, it has been shown that when dogs are fed low-fat diets (5.35%, as fed) containing 3.2% (as fed) eight-carbon and 1.5% (as fed) 10-carbon length medium-chain triglycerides (MCTs), that some MCT is absorbed into the intestinal lymphatics.31 Nonetheless, the ability of MCFAs to bypass the lymphatics in most situations make them useful in managing some types of GIT disorders, especially those involving the lymphatics (lymphangiectasia). However, the diet must always contain LCFAs because these provide the essential fatty acids required in the diet of dogs and cats. Diets containing MCTs usually restrict the amount of MCTs to 25% to 30% of the fat in the diet because MCTs are not very palatable, and animals will often refuse to eat diets containing higher quantities of MCTs. Because MCTs are ketogenic, they should be avoided in patients with acidosis or ketosis.
Both dogs and cats have a dietary requirement for linoleic acid (LA), which is an n-6 fatty acid.32 Cats also require arachidonic acid (AA), which is also an n-6 fatty acid. Dogs, however, are able to synthesize adequate amounts of AA from LA and therefore do not have a dietary requirement for AA. Although definitive studies have not been done in dogs and cats, most nutritionists believe that the n-3 fatty acid, α-linolenic acid (ALA), is also necessary in the diet of dogs and cats. Although both dogs and cats have the ability to synthesize eicosapentaenoic acid (EPA) from ALA, their ability to synthesize docosahexaenoic acid (DHA) from ALA is very limited or not present at all. Because DHA is very important for retinal and brain development, DHA is considered a conditionally essential fatty acid. Including DHA in the diet of puppies and pregnant bitches improves learning abilities of the puppy (data on file, P & G Pet Care). Dietary sources of EPA and DHA include fish oils, such as menhaden oil.
Eicosanoid Production from Fatty Acids
When PUFAs are metabolized in the body, n-6 fatty acids produce eicosanoids that are different from those produced by metabolism of n-3 fatty acids. These differences can be useful when managing certain types of GIT disorders, especially those associated with inflammation. Eicosanoid synthesis begins with metabolism of AA or EPA by one of two enzyme systems. The first enzyme system is cyclooxygenase, which yield prostaglandins (PGs) and thromboxanes (TXs). The lipoxygenase system yields leukotrienes (LTs), as well lipoxins (LXs), hydroperoxyeicosatetraenoic acids (HEPEs), and hydroxyeicosatetraenoic acids (HETEs). The amount and types of eicosanoids synthesized is dependent upon the availability of the fatty acid precursors, and the activity of the cyclooxygenase and lipoxygenase pathways. The eicosanoids produced from the n-6 fatty acid, AA, are proinflammatory, whereas those produced from n-3 fatty acid, EPA, promote minimal to no inflammatory activity. Because many of the GIT disorders are associated with inflammation, feeding a diet supplemented with n-3 fatty acids may prove useful in reducing the level of inflammation in the GIT. For example, diets supplemented with n-3 fatty acids have been shown to reduce the degree of inflammation in experimental models of colitis.33
Fat Malassimilation
Dietary fat is one of the most complex nutrients to process in the GIT. Many steps are involved in the digestion and absorption of lipids (see Chapter 1). In the small intestine, large dietary fat droplets must first be emulsified by bile salts from the liver. This dramatically increases the surface area available for enzyme action. The resulting smaller fat droplets (triglycerides) are broken down into free fatty acids and monoglycerides by the actions of pancreatic lipase and then incorporated into micelles before absorption through the epithelial membrane. Once both the free fatty acids and monoglycerides are inside the enterocytes, they are repackaged into triglycerides, coated with a lipoprotein to form chylomicrons before entering the lymphatic vessels. Because this process involves multiple organs and multiple steps, disruption of fat digestion and absorption may take place in several gastroenterologic disorders. The resulting fat malassimilation not only interferes with fat absorption, but also interferes with fat-soluble vitamin, mineral, and bile salt absorption. Malabsorbed fat and bile acids are hydroxylated and deconjugated by colonic bacteria to produce potent secretagogues. The deconjugated bile acids and hydroxylated fatty acids are toxic to the colonic mucosa, causing increased mucosal permeability, altered motility, and secretory diarrhea. Dogs and cats with this type of diarrhea can become rapidly dehydrated, develop electrolyte abnormalities, and experience rapid weight loss. The best way to manage these patients is to address the underlying cause of the problem if possible, such as exocrine pancreatic insufficiency, biliary obstruction, and IBD. These animals may also benefit from a lower-fat diet until the underlying cause is addressed. However, it is very important to keep in mind that fat has 2.25 times more energy density per gram than protein or carbohydrate, and if dietary fat intake is reduced, it may be more difficult to meet the energy needs of the patient.
Vitamins and Minerals
Numerous electrolyte and vitamin abnormalities can be associated with GIT disorders, including perturbations in folate and cobalamin (vitamin B12) metabolism. A decrease in serum folate concentrations can be associated with proximal small intestinal disorders, while a decrease in serum cobalamin concentrations can be associated with distal small intestinal disorders and exocrine pancreatic disease. Both serum folate and cobalamin may be decreased with diffuse small intestinal disease. It is particularly important to keep these two vitamin abnormalities in mind for patients with chronic diarrhea and an unthrifty, unkempt appearance, especially cats. Dietary therapy alone is unlikely to resolve these problems. The reader is referred to Chapter 53 for further discussion of folate and cobalamin metabolism.
Folate
Most folate found in pet foods is in the form of folylpolyglutamate and very little is found in the free folate (folylmonoglutamate) form. Liver, animal by-products, and oil-seed meal, such as soybean meal, are the most common sources of folate in pet foods.34 Dogs reportedly are able to absorb folylpolyglutamates across the small intestines and therefore are much less likely to develop folate deficiency than are cats. However, only the folylmonoglutamate form of folate is actively transported across the small intestines in cats. Folylpolyglutamate in food needs to be deconjugated by the brush-border enzyme, folate deconjugase, to produce folylmonoglutamate. Deficiencies of folate result in impaired biosynthesis of DNA and RNA, and, thus, reduced cell division. The GIT is lined by a rapidly dividing population of cells, and folate deficiency can interfere with this process. Decreased DNA synthesis caused by folate deficiency can also result in megaloblastosis. Treatment of folate deficiency involves identifying and treating the underlying intestinal problem, as well as supplementing the diet with folic acid (200 µg of folic acid per day for 1 month).35
Cobalamin (Vitamin B12)
Synthesis of vitamin B12 is limited almost exclusively to bacteria. As a result, it is found only in foods that have been fermented and those derived from the tissues of animals that have obtained it from their intestinal microflora. Vitamin B12 is absorbed in bound form (to intrinsic factor), and it is absorbed only in the ileum. A recent study showed that 61% of 80 cats with clinical signs of chronic gastrointestinal disease had decreased serum cobalamin concentration,36 and almost all cats diagnosed with exocrine pancreatic insufficiency (EPI) had cobalamin deficiency.37 Some cats with EPI will respond poorly to pancreatic enzyme therapy unless cobalamin deficiency is concurrently treated. Unfortunately, supplementation to correct cobalamin deficiency requires that it be given parentally and not enterally because cobalamin deficiency causes cobalamin malabsorption. The dose of cobalamin in cats is 150 to 250 µg of cobalamin (cyanocobalamin injection, Elkins-Sinn, Goldline, or others) given subcutaneously once a week.35 The serum concentration of cobalamin should be rechecked before the fifth and sixth injection. If serum cobalamin concentration has normalized, the dosing schedule can be modified to one injection every 2 to 4 weeks. These animals generally will require lifelong therapy with cobalamin injections.
Prebiotics, Probiotics, and Synbiotics
Companion animals have extensive gastrointestinal bacterial ecosystems. For example, the mammalian digestive system contains more than 500 different species of bacteria.38 The balance between beneficial and pathogenic bacteria has an effect on the overall health of the animal. As a result, the microbial population in the GIT is recognized to play a substantial role in the health of animals, and its role appears to extend beyond the GIT. Many of the extragastrointestinal effects appear to be related to changes in the immune system. Enteric bacteria also contribute significantly to the host’s resistance to infectious disease. It is well recognized that a healthy gut bacterial flora is essential for overall health, and this flora is often disrupted during illnesses and may, in some cases, be a precipitating factor.
Establishment of Intestinal Bacterial Flora
Intestinal microflora are established at birth as the neonate passes through the birth canal and after birth through suckling and environmental exposure. Optimal maturation of the gut-associated immune system during the first months of life depends on the development and composition of this native microflora. The most common bacteria with potential to enhance health in pets are the bifidobacteria and lactobacilli. With advancing age, the level and diversity of these beneficial bacteria begin to decline, while less-favorable Clostridium spp. bacteria increase.41 In addition, the bacterial flora composition in the GIT is influenced by many factors including host species, breed, age, dietary history, environmental conditions, geographic locale, intestinal motility patterns, disease, and medication history.
Definitions of Pre-, Pro-, and Synbiotics
Prebiotics are defined as nondigestible food ingredients that beneficially affect the host by selectively stimulating the growth and/or activity of one or a limited number of beneficial bacteria in the colon that improve host health. The most common prebiotic found in diets of dogs and cats is dietary fiber.22,42
Probiotics are defined as living beneficial bacteria, which upon ingestion in sufficient numbers exert health benefits to the host.43 It is very important that both parts of the definition are met in order to call a product a probiotic.
Fructooligosaccharides
Certain fermentable fiber sources, such as FOS, are good sources of prebiotics for dogs and cats, and the proportion of different bacterial species is related to the type of fermentable substrate available. Fructooligosaccharides are found naturally in many different foods, including plants like beet root (after pulp processing), soy (in the hulls), psyllium, chicory (after hydrolysis), and numerous other fruits, vegetables, and grains.44 They can also be synthesized commercially. Beneficial intestinal bacteria (e.g., lactobacilli and bifidobacteria) use fermentable fiber as a metabolic fuel, whereas pathogenic bacteria (e.g., Salmonella, Escherichia coli, Clostridium perfringens) cannot metabolize FOS for energy. Production of SCFAs from fiber by beneficial bacteria also lowers colonic pH, further impeding the growth of bacterial pathogens.45 As a result, in the presence of FOS, beneficial bacteria thrive, multiply, and crowd out pathogenic bacteria. In addition, a study by Willard et al. showed that supplementing the diet of dogs with FOS resulted in a significant decrease in the number of aerobic and anaerobic bacteria in the small intestine of dogs with small intestinal bacterial overgrowth (SIBO).46 Similarly, FOS supplementation increased numbers of beneficial bacterial and decreased numbers of potential pathogens in the large intestine of healthy cats.47
Dried beet pulp is another fermentable fiber source in the diet of dogs that beneficially enhance the growth and survival of good bacteria in the gut. Beet pulp is the fiber material that remains after sugar is extracted from sugar beets.47a It has been widely used in the livestock industry for many years, and in the last 10 years, the use of beet pulp has expanded into the pet food industry as a source of fiber. It is a safe fiber source and contains no known toxins.
Mannanoligosaccharides
MOS are unique fiber sources similar to FOS. The difference between FOS and MOS is that fructose is the predominant sugar molecule in FOS, whereas mannose is the predominant sugar molecule in MOS. MOS are natural fibers found in yeast cells, and they prevent the growth of harmful bacteria in the GIT through a different mechanism. Pathogenic bacteria establish themselves in the GIT by attaching to the intestinal wall and colonizing the GIT. Pathogenic bacteria are able to attach to the intestinal wall because they have finger-like projections, called fimbriae, that allow them to bind to specific residues (e.g., mannose) on intestinal cells. Because MOS contains mannose, fimbriated mannose-specific pathogens can bind to MOS instead of the intestinal wall.48 By preventing these bacteria from adhering to the intestinal wall, MOS can inhibit the growth of pathogenic organisms, reduce their effects in the GIT and aid in the excretion of these harmful bacteria. They are very effective in preventing diarrhea and contribute to the prevention of digestion-related infectious diseases.
Fructooligosaccharides and Mannanoligosaccharides Enhance the Effectiveness of the Gastrointestinal Immune System
The gut contains both nonimmunologic barrier defenses and immunologic barrier defenses. Nonimmunologic barriers include (a) the anatomy of the gut (intact microvilli and tight junctions between cells); (b) peristalsis and mucus, which makes it difficult for pathogens to attach and enter cells; (c) low pH of gastric secretions; and (d) digestive and bactericidal enzymes secreted by the stomach, pancreas, and epithelial cells to inhibit the attachment and growth of bacteria.49
The gut is also the largest immunologic organ in the body (see Chapter 3). As a result, immunologic barriers play a critical role in maintaining normal health and function both locally and systemically. Gut-associated lymphoid tissue (GALT) is composed of cells residing in the lamina propria, intraepithelial lymphocytes interspersed between epithelial cells, and immune cells located in organized lymphatic tissue (Peyer’s patches and mesenteric lymph nodes).
Fermentable prebiotic fibers, such as FOS, can have a major impact on the gut immune function. In a study by Field et al., dogs were fed isonitrogenous, isoenergetic meat-based diets supplemented with either a combination of fermentable fibers (beet pulp, gum arabic, and FOS) or with nonfermentable fiber (cellulose).50 Each diet contained similar amounts of fiber but differed in fermentability. The diet supplemented with fermentable fiber significantly (P <0.05) decreased the proportion of Ig+ cells and increased the CD4/CD8 (i.e., T-helper cell-to-cytotoxic T cell ratio) in peripheral blood. Therefore, adding fermentable fiber to the diet of dogs changes the composition and function of immune cells in GALT.
Probiotics
The most common type of probiotic bacteria are the lactic acid bacteria, such as Bifidobacterium and Lactobacillus. Probiotics have been used for many years in human medicine. Studies evaluating their potential use in dogs and cats are only in their infancy, but so far they have demonstrated their effectiveness at improving gastrointestinal health in both humans and animals.50a The appealing properties of probiotics include their ability to reduce antibiotic use, the apparent high indicator of safety, and the public’s continuing positive perception about “natural” therapies.
Clinical Benefits of Probiotics in Patients with Gastrointestinal Tract Disorders
Numerous clinical trials have validated the use of probiotics for specific clinical conditions in humans. Although research on probiotic use in dogs and cats lags behind that in humans, it is becoming increasingly clear that probiotics confer many of the same health benefits to dogs and cats. New research has now demonstrated that probiotics can positively affect gastrointestinal conditions in dogs. It is known that the microbial flora of the gut play a role in the normal function and maintenance of health of the GIT. Recent studies have shown that feeding of probiotics to dogs improves stool quality (P&G Pet Care, unpublished research). In one study, young dogs (14 to 16 months old) were fed a probiotic (canine-derived Bifidobacterium animalis AHC7; Iams, Prostora Max) or placebo prior to traveling to a training kennel. Fewer of the dogs that received the probiotic experienced loose stools during the transition to the kennel compared with dogs that received placebo. Another study examined the effects of feeding this same probiotic product to dogs with acute severe watery diarrhea.51 The time to resolution of diarrhea was approximately 40% less (2.5 days faster) in dogs that received the probiotic than in dogs that received the placebo. Another study examined the effects of this same probiotic in service dogs. Service dogs typically spend the first 14 to 16 months of their lives in a foster home before returning to the service organization for training. It is not uncommon for dogs to develop diarrhea during this transition, which interferes with their training. When compared with dogs that received the placebo, there was a statistically significant reduction in the number of dogs with less-than-ideal fecal scores when probiotics were started 5 weeks prior to this transition. In addition, serum C-reactive protein (CRP) concentrations, which reflect inflammation, were evaluated in both groups. The dogs that received the placebo had elevated levels of CRP above the reference range for at least the first 10 days at the training facility, whereas levels of CRP remained within the reference range the entire time in dogs that received the probiotic. This study illustrates how probiotics not only have beneficial effects in the GIT but systemically as well. These studies indicate that feeding this organism may promote GIT health in dogs, even in the setting of acute diarrhea.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree