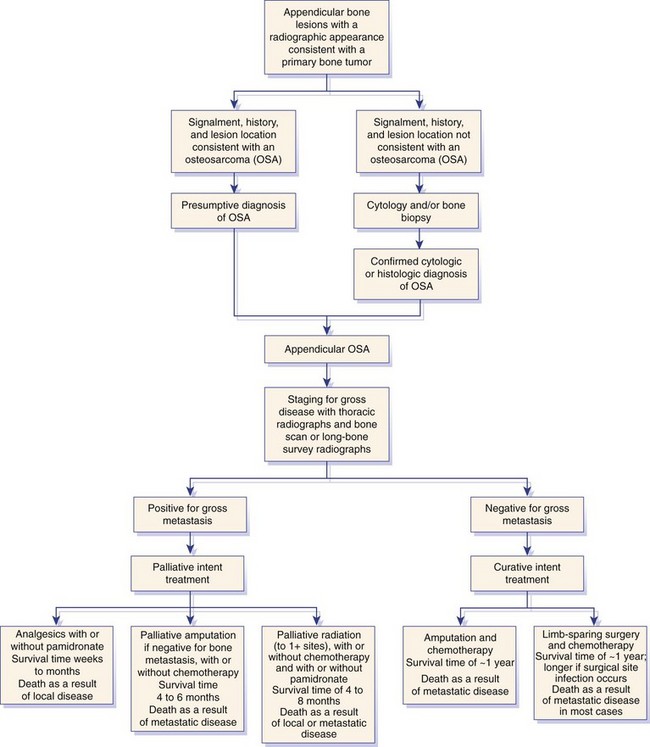
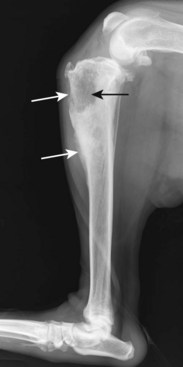
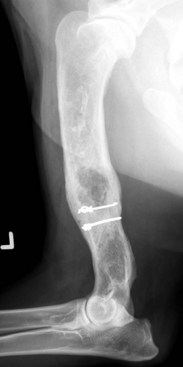
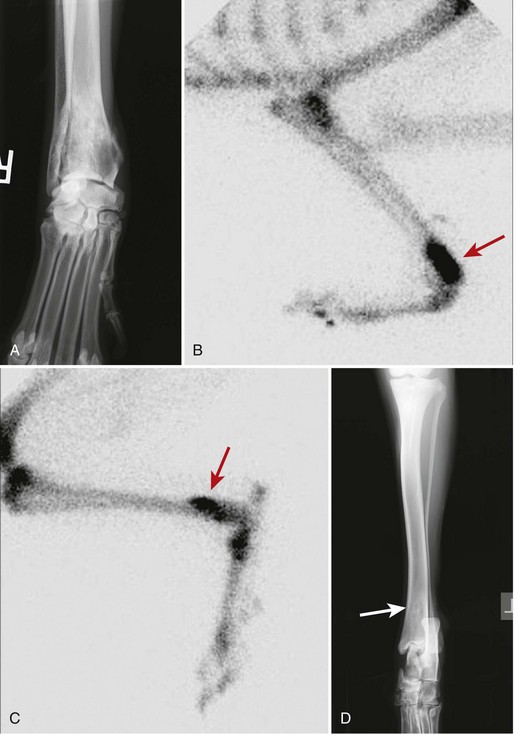
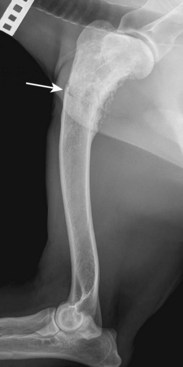
Chapter 72 Osteosarcoma is the most common primary bone tumor in dogs, accounting for approximately 85% of tumors of the skeleton.21 Appendicular osteosarcoma is primarily a disease of large- and giant-breed dogs.21,80 Increasing height and weight have been associated with increased risk of developing osteosarcoma.80 Commonly affected breeds include St. Bernard, Great Dane, Greyhound, Rottweiler, Irish Setter, Doberman Pinscher, Golden Retriever, Irish Wolfhound, Scottish Deerhound, Borzoi, and German Shepherd Dog.21,67,78 Breed-associated cytogenetic aberrations have been identified in the Golden Retriever and the Rottweiler, suggesting that there may be a correlation between breed and tumor karyotype in canine osteosarcoma.92 Older dogs are more commonly affected, with a median age of 7 years.21,80 However, two peaks in the age of onset have been noted: a small peak at 18 to 24 months, and a larger one at 10 years; therefore, young age does not preclude the diagnosis of osteosarcoma.21 An inverse relationship has been documented between the length of gonadal hormone exposure and the risk of developing osteosarcoma in Rottweilers, suggesting that early ovariohysterectomy or castration may result in increased risk of development of osteosarcoma.15 Axial skeletal osteosarcoma is less common than appendicular osteosarcoma.21 The biologic behavior of axial osteosarcoma is similar to that of appendicular osteosarcoma.22,39,42 With the exception of mandibular osteosarcoma,39,89 there does not appear to be a survival benefit for axial osteosarcoma compared with appendicular osteosarcoma.22,39,42 The reported pulmonary metastatic rate at presentation for dogs with axial osteosarcoma is 11% to 18%, with a metastatic rate at presentation of 40% for dogs with skull osteosarcoma in one study.22,39,42 One significant difference between axial and appendicular osteosarcoma is that death is often due to local disease rather than metastatic disease with axial osteosarcoma. Death rates due to local disease have been reported in three retrospective studies on canine axial osteosarcoma as 55%, 64%, and 79.6%.22,39,42 This is primarily because complete excision of the tumor in a patient with an axial osteosarcoma may not be possible, or if possible is more challenging than complete excision of the tumor in a patient with appendicular osteosarcoma. The focus of the following discussion of osteosarcoma will be on patients with appendicular osteosarcoma. Osteosarcoma has a predilection for the metaphyseal regions of long bones, but it can occur at any bone site. The most common sites of osteosarcoma include the metaphyseal regions of the distal radius, proximal humerus, distal femur, and distal and proximal tibia. The distal radius and the proximal humerus are the two most common sites in dogs.21 The decision-making process associated with the diagnosis and treatment of canine osteosarcoma can be challenging. An algorithm, including general prognosis for survival, is presented in Figure 72-1. A presumptive diagnosis of osteosarcoma is based on signalment, tumor location, and radiographic signs. Common radiographic signs of osteosarcoma include cortical lysis, periosteal reaction, extension of osteogenesis into adjacent soft tissue, loss of the fine trabecular pattern of the metaphysis, areas of fine punctuate lysis, and lack of a distinct border between normal and abnormal bone (Figure 72-2).21 A sharp demarcation between normal and abnormal areas of bone is more suggestive of an infectious process or a bone cyst than of a neoplastic process. The most common type of osteosarcoma is endosteal, which means that it arises within the medullary canal. Periosteal and parosteal osteosarcomas arise from the periosteum and are much less common. Periosteal osteosarcoma can invade the medullary cavity from the periphery, cause bone lysis and reactive bone formation, and produce neoplastic bone. Parosteal osteosarcoma also arises from the periosteum but differs from periosteal osteosarcoma in that it forms an expansive mass that surrounds but does not invade the underlying cortex. Both periosteal and parosteal osteosarcomas are relatively rare.102 The cortical lysis seen in early cases of osteosarcoma can be very mild and may involve only the endosteum in early lesions. Histopathology is necessary for a definitive diagnosis. Other differential diagnoses for a primary bone tumor include sarcomas (chondrosarcoma, fibrosarcoma, and hemangiosarcoma), which originate from the other mesenchymal cells present in bone, metastatic neoplasia, multiple myeloma, lymphoma, bacterial or fungal osteomyelitis, and bone cyst. Bone biopsy is performed using a Jamshidi needle or a Michele trephine and a closed technique in most cases.76 The accuracy rate of bone biopsy is 80% to 90%.76 Because of the larger size of the core sample removed (and therefore the larger defect in the bone), the risk of pathologic fracture is increased with a Michele trephine compared with a Jamshidi needle. For this reason, a single core is taken when a Michele trephine is used, compared with multiple cores with use of a Jamshidi needle. Core samples can be rolled onto a slide for cytologic evaluation. A portion of the sample should be reserved for bacterial and fungal culture. When planning the site of bone biopsy, it is important to take options for definitive therapy into consideration. The biopsy tract is considered contaminated with tumor cells and must be removed during definitive therapy. This is particularly important if limb-sparing surgery is an option. Only one small skin incision should be made in an area that is amenable to resection. A Jamshidi needle can be used to obtain multiple samples, but these should always be obtained through a single incision. Ideally, the skin incision is planned in an area of minimal soft tissue coverage over the bone. When obtaining the biopsy, care is taken to not penetrate the opposite cortex of the bone, as this may seed tumor cells that are not removed with local resection or may further weaken the bone. Tumors of the distal region of the radius should be biopsied from the cranial surface, slightly lateral or medial, depending on the preferred approach to the radius for limb-sparing surgery. If limb-sparing surgery is an option that the owner wishes to pursue, it is recommended that the case be discussed with the surgeon who will perform the limb-sparing procedure before the bone biopsy is performed. An emerging concern is that if stereotactic radiosurgery is a consideration for limb-sparing surgery, a bone biopsy may increase the risk of pathologic fracture at bone biopsy tract sites.29a An alternative to bone biopsy and histopathologic evaluation is cytologic evaluation of a sample obtained via a fine needle aspirate. The diagnostic accuracy of cytology was found to be 71% overall and was 92% when a neoplastic process was identified.8 This technique has been used in concert with ultrasound in an attempt to improve the diagnostic yield. Ultrasound can be used to identify a break in the cortex through which a 20 gauge hypodermic needle can be inserted. A diagnostic sample was obtained in a recent study in 32 of 36 cases, indicating that this technique may be a good alternative to surgical biopsy or reliance on a presumptive diagnosis.12 Staging is performed when osteosarcoma is suspected or has been confirmed, to determine whether there is gross metastasis of disease. Metastasis has occurred in nearly all patients by the time a diagnosis of osteosarcoma is made. However, this is micrometastatic disease in most cases, as gross metastasis is identified in ≈15% of patients at the time appendicular osteosarcoma is diagnosed.21 The two most common sites of metastasis are the lungs and another bone site. Three-view thoracic radiographs are an essential part of tumor staging, and lung lesions that are 7 to 9 mm in diameter should be visible on high-quality radiographs.69 Less than 10% of patients with osteosarcoma will be positive for gross disease on thoracic radiography at the time of initial staging.21 Computed tomography (CT) scan is the modality of choice for identifying lung metastasis in human beings with osteosarcoma.74,97 This test has been shown to be highly sensitive but not specific for diagnosing gross metastasis to the lung in human beings.74,97 The threshold size of the lesion for diagnosis of pulmonary metastatic nodules in dogs in one study was 1 mm.69 Detection of metastasis to bone before definitive therapy is initiated is important not only for prognostic purposes but because it will preclude amputation in most cases and may make other treatment options more practical. The probability of diagnosing gross bone metastases at the time of diagnosis of osteosarcoma is similar to the probability of finding gross pulmonary metastases.44 Detection of metastasis to other bones involves a complete orthopedic examination and long bone survey radiography or nuclear scintigraphy. A complete orthopedic examination, including palpation of the spine, is important in determining whether the dog is a good candidate for an amputation and in evaluating for possible metastatic disease. Focused radiographic evaluation should be performed on areas that are identified as being associated with a pain response. Radiography of all of the long bones and the pelvis can be performed to survey for evidence of metastatic disease.88 If available, nuclear scintigraphy using technetium-99m is a more sensitive way to evaluate for metastatic disease. Technetium is administered by an intravenous injection and is incorporated into areas of bone with active metabolism. Relative uptake in different areas of the skeleton is then evaluated using a gamma camera. This is a sensitive but not specific method of evaluating for bone metastases because it does not discriminate between bone tumor, infection, fracture, and other orthopedic conditions. However, an experienced radiologist can generally determine the likelihood of metastatic disease when evaluating areas of increased uptake. Areas that appear abnormal on bone scan should be radiographed to determine the cause of increased uptake of radioisotope (Figure 72-3). Jankowski et al. evaluated 399 dogs with osteosarcoma that were staged with a bone scan and found that 7.8% of those dogs were highly suspected as having metastatic disease based on follow-up radiographs of suspicious areas.44 The incidence of metastasis to regional lymph nodes is reported to be 4.4%.43 Dogs with osteosarcoma treated with limb amputation and chemotherapy had a much shorter median survival time when evidence of lymph node metastasis was found (median survival time = 59 days) compared with dogs in which such evidence was not found (median survival time = 318 days).43 The regional lymph nodes should be palpated and aspirated if enlarged. If the diagnosis of lymph node metastasis is not made before amputation, histologic examination of the regional lymph node removed during amputation may provide valuable prognostic information. Regional lymph nodes are considered to be the axillary and prescapular lymph nodes for front limb amputation and the popliteal and inguinal lymph nodes for hindlimb amputation. Complete blood count (CBC), serum biochemistry, and urinalysis are important parts of the overall evaluation of the patient prior to surgery and treatment. Renal status should be determined before initiation of chemotherapy, as renal toxicity is a potential side effect of some chemotherapeutic agents. Total alkaline phosphatase (ALP) should be assessed preoperatively because bone alkaline phosphatase (BALP) is an isoenzyme of alkaline phosphatase. Although bone alkaline phosphatase is not routinely evaluated separately, an increase in both total alkaline phosphatase and bone alkaline phosphatase has been identified as an indicator of poor prognosis in dogs with osteosarcoma.26 Failure of bone alkaline phosphatase activity to decrease after amputation was associated with a shorter survival and disease-free interval.26 Local staging to determine extent of disease is an important part of surgical planning in the limb-sparing surgery candidate. Generally, this additional staging occurs in patients with distal radial osteosarcoma because they are the most likely candidates for the limb-sparing procedure. Local staging can be performed using radiographs of the entire antebrachium, CT scan, magnetic resonance imaging (MRI), or scintigraphy. A study by Davis et al. compared radiographs, CT, and MRI versus histopathology using macroslides in cases of appendicular osteosarcoma of 10 amputated limbs. In that study, radiographs and CT were accurate, but each underestimated the extent of tumor in one case. MRI was the least accurate method but did not underestimate tumor length, suggesting that it may be a superior method of local staging for limb-sparing surgery candidates.18 Another study compared radiographs, nuclear scintigraphy, and histopathology for distal radial lesions and found that both radiographs and scintigraphy overestimated the extent of the tumor, but that scintigraphy did this to a greater degree. The authors concluded that the use of scintigraphy for margin planning in limb-sparing surgery cases will overestimate the extent of disease, and although this will provide a greater margin of safety, it may overestimate the disease to the point of excluding patients as limb-sparing surgery candidates.54 Wallack et al. evaluated MRI as a technique to assess the extent of osteosarcoma compared with macroslide histopathology, radiographs, CT, and bone scan when available. All modalities appeared to overestimate tumor extent, with MRI considered the most accurate method.99 Based on these studies, there does not appear to be a clear answer as to which diagnostic modality will provide the best information to ensure accurate planning for resection in cases of limb-sparing surgery. High-quality radiographs of the entire antebrachium are necessary to plan surgery and implant selection. The addition of at least one other imaging modality will help with surgical planning. CT or MRI has the added advantage of allowing visualization of the distal radius and ulna in three dimensions to determine whether there is extension of the tumor from the distal radius to the ulna. Advanced imaging should also be considered for surgical planning for bone tumors of the pelvis and scapula, as advanced methods are more accurate than radiographs in determining the extent of disease and the degree of soft tissue involvement. A small percentage of patients with appendicular osteosarcoma will present with a pathologic fracture (Figure 72-4). The pathologic nature of the fracture may or may not be apparent radiographically, and a high index of suspicion for a neoplastic process should be maintained in large- or giant-breed dogs that present for a fracture without a history of trauma. Appropriate radiographic evaluation and tumor staging are recommended if pathologic fracture is suspected. Dogs with pathologic fracture present a unique set of challenges. Recommended treatment for a patient with a pathologic fracture and a presumptive diagnosis of osteosarcoma or a primary bone tumor is limb amputation. If the patient is not a suitable candidate, or the owner does not consent to amputation, fracture repair should be considered. A recent Veterinary Society of Surgical Oncology retrospective study evaluated 16 canine patients with an appendicular pathologic fracture due to osteosarcoma or sarcoma that were treated with fracture repair. In most cases, bone plating was used as the method of fracture repair. However, interlocking nail, plate-rod, or external skeletal fixation was employed in some cases. In that study, the median survival time was 166 days, with a range of 18 to >897 days. The dog that lived for only 18 days had gross pulmonary metastasis at the time of presentation. A trend toward increased survival time was noted with adjunctive treatment.10a Fracture-associated osteosarcoma has been reported in dogs; the condition has been described in one cat.35,86 Fracture-associated osteosarcoma occurs most frequently in dogs that have had a comminuted fracture with a history of complicated healing, implant loosening, and/or infection, and it tends to occur after a lag period of 5 years or longer following fracture repair (Figure 72-5).86 Consequently, this condition is diagnosed most frequently in older dogs that suffer a fracture when relatively young.86 Typically, these are tumors of the diaphysis (most frequently, femur, humerus, or tibia) of large-breed dogs that are older than 7 years.86 When osteosarcoma develops at the site of a metaphyseal fracture within a short time (months) following fracture repair, it is most likely the original fracture was a pathologic fracture that was not diagnosed at the time of fracture repair instead of the tumor being a true fracture-associated osteosarcoma. The cause of a fracture-associated sarcoma is not known. Hypotheses include implant corrosion at the fracture site or inflammation at the fracture site associated with complicated healing, inflammation, and infection.86 Histopathologically, fracture-associated sarcomas are often characterized by inflammation that is not recognized with spontaneously occurring osteosarcoma.86 Implant corrosion has been suggested as a cause of implant- or fracture-associated osteosarcoma and was recognized when Jonas pins were used in veterinary medicine. Jonas pins are of historical interest because these spring-loaded pins were used for intramedullary pin fixation of fractures. Jonas pins are no longer commercially available. These implants were known to corrode because they were constructed of multiple types of metal.83,84 A more contemporary example of implant corrosion as the cause of bone sarcoma is the development of osteosarcoma at a tibial plateau leveling osteotomy site, which has been described in two case reports.11,87 It was hypothesized that these tibial plateau leveling osteotomy -related osteosarcomas occurred secondary to corrosion of the implants.11,14 Investigations into explanted, early-generation tibial plateau leveling osteotomy plates have corroborated this theory, as early generation tibial plateau leveling osteotomy plates have been shown to have irregularities in the manufacturing process and increased potential for corrosion.11,14 Staging and treatment of fracture-associated osteosarcoma should occur in a manner similar to that used for spontaneous osteosarcoma because the biologic behavior is similar. Standard therapy includes limb amputation and chemotherapy. Bone tumors that commonly affect the scapula include chondrosarcoma and osteosarcoma. Tumors of the scapula can be treated with forequarter amputation or, in some cases, with partial or subtotal scapulectomy and limb preservation. A partial scapulectomy involves the removal of a section of the proximal scapula, with preservation of the acromion, the acromial head of the deltoideus muscle, and the distal portion of the infraspinatus and supraspinatus muscles. This allows for greater stability of the shoulder joint compared with a subtotal scapulectomy.95 A subtotal scapulectomy involves removing most of the scapula (as far distal as the scapular notch) while preserving the glenoid and the glenohumeral joint. This procedure is sometimes referred to as a total scapulectomy; however, the glenoid is preserved in all cases. Loss of muscular support that occurs with subtotal scapulectomy will result in a greater dorsoventral excursion during weight bearing.95 The amount of scapula removed is dependent on the extent of disease. Advanced imaging of the scapula using MRI or CT is recommended prior to scapulectomy to aid with surgical planning. Candidates for scapulectomy include patients in which the proximal scapula is affected, the tumor has not extended into surrounding soft tissues, the scapula can be removed with a 2 to 3 cm tumor-free margin distal to the tumor, and the glenohumeral joint is preserved. If biopsy was performed, the biopsy tract must be removed with the scapula. The surgical technique is summarized in the following paragraph.95 A skin incision is made that encompasses the biopsy tract. For primary bone tumors, the skin directly over the scapula is preserved. Muscle attachments of the omotransversarius, trapezius, and deltoideus muscles are identified and excised 2 to 3 cm from their attachment to the scapula to allow an adequate soft tissue margin. The lateral surface of the distal part of the scapula is exposed by separating the supraspinatus and infraspinatus muscles. The teres major muscle is identified on the caudal aspect of the scapula and is transected. The rhomboideus, serratus ventralis, and subscapularis muscles are identified on the medial aspect of the scapula and are transected. The teres minor muscle and the long head of the triceps brachii muscle are cut along the caudal edge of the scapula. An oscillating saw is used to transect the scapula at the appropriate level.95 Depending on the level of the scapulectomy, holes can be drilled in the remaining scapula, and muscles can be sutured to their approximate origins or insertions using bone tunnels for suture placement. If a subtotal scapulectomy has been performed, the muscle edges are reapposed across the defect. A closed suction drain is placed to minimize dead space. Seroma formation is a commonly reported complication of scapulectomy.70,95 In three case series reporting on scapulectomy, limb use was determined to be good to excellent in 6 of 6, 4 of 5, and 3 of 4 cases, based on postoperative clinical assessment.48,70,95 In the author’s opinion, limb use after partial and especially subtotal scapulectomy is not completely normal. However, pain-free ambulation with functional lameness is possible. Some dogs will use the limb for support, much like a crutch or a cane. More normal ambulation may be achieved in dogs with a partial scapulectomy than in those with a subtotal scapulectomy. In a cases series reporting four dogs that underwent scapulectomy, the three dogs that underwent a partial scapulectomy were found to have excellent limb function, whereas the one dog that underwent a total scapulectomy had fair limb function. That dog had mechanical lameness but was thought to be pain-free.48 Hemipelvectomy is a treatment option for some patients with primary bone tumors of the pelvis. Although a closed approach may not be possible for biopsy of a tumor of the pelvis, if a biopsy is performed, a minimal approach should be used, with care taken to biopsy at a location that can be removed easily with definitive resection. Hemipelvectomy was first reported in veterinary patients in 1992.91 In that case series of 13 dogs and 2 cats, the tumor types encountered included osteosarcoma (8), fibrosarcoma (3), and chondrosarcoma (2), with fibrosarcoma (1) and rhabdomyosarcoma (1) identified in the 2 cats.91 Vaccine-associated fibrosarcoma may now be a common diagnosis and indication for cats undergoing hemipelvectomy.82 Hemipelvectomy can be divided into four categories: total hemipelvectomy, which includes the entire hemipelvis from the pubic symphysis to the sacroiliac joint; mid-to-caudal hemipelvectomy, which includes the hemipelvis from the pubic symphysis to mid-ilium, including the acetabulum (Figure 72-6); mid-to-cranial hemipelvectomy, which extends from the sacroiliac joint to just caudal to the acetabulum; and caudal hemipelvectomy, which includes the pubic symphysis to just caudal to the acetabulum.50 Caudal hemipelvectomy allows limb preservation.50 In general, the limb is resected with the acetabulum, but anecdotal reports have described preservation of the limb with a partial hemipelvectomy that involves the acetabulum. The amount of the hemipelvis that is removed is dependent on the degree of tumor involvement. Potential candidacy for a hemipelvectomy can be determined with the use of plain radiographs. However, CT scan is recommended for surgical planning. If an osteosarcoma has been definitively or presumptively diagnosed, scintigraphy is recommended for local and distant staging. Figure 72-6 Ventrodorsal radiograph of a canine pelvis after mid-to-caudal hemipelvectomy for a soft tissue sarcoma. When the procedure is planned and performed, 2 to 3 cm margins should be achieved beyond the extent of the tumor. It is possible to extend the resection across the contralateral pubis, but this will complicate reconstruction of the caudal body wall. Usually the abdominal wall defect can be reconstructed using local tissue. However, in some cases, a prosthetic mesh is used for reconstruction. Although it is conventional to disarticulate the ilium at the sacroiliac joint, a partial sacrectomy may be necessary to achieve appropriate margins in tumors that cross the sacroiliac joint. Resection of up to one third of the sacrum can be performed with normal function in the remaining contralateral limb.102a Significant blood loss is possible with hemipelvectomy, and preparations should be made so blood transfusion can be performed without delay if necessary. Postoperative pain management is an essential part of this procedure. A closed suction drain or a pain diffusion (soaker) catheter, or both, can be employed postoperatively to limit dead space and manage pain, respectively. This procedure lends itself to preemptive analgesia with an epidural,71 and an indwelling epidural catheter can be placed for postoperative management. A multimodal approach to analgesia should be used, with opioids, NSAIDs, and other analgesics administered as needed. The urinary catheter can remain in place for the first 8 to 24 hours to limit ambulation in the immediate postoperative period. The surgical procedure and recovery are more complicated for patients undergoing hemipelvectomy than for patients undergoing amputation by disarticulation or at the mid-femur level. However, for all intents and purposes, patients treated by hemipelvectomy are very similar functionally and cosmetically to other hindlimb amputees. Many limb-sparing techniques are currently used by surgical oncologists to treat canine distal radial osteosarcoma. These include cortical allograft, pasteurized autograft, intraoperative radiation (autograft), endoprosthesis, vascularized ulnar transposition, stereotactic radiosurgery, and bone transport osteogenesis. With the exception of intraoperative radiation and stereotactic radiosurgery, these techniques involve resection of the distal part of the radius with appropriate margins and replacement of the defect that is created with autograft, allograft, prosthesis, or regenerated bone. As with many surgical procedures in which multiple techniques are available, there is not one perfect technique that is without its own set of complications. Currently, the endoprosthesis technique appears to be used most commonly by surgical oncologists.98 Limb-sparing surgery is most commonly performed to treat osteosarcoma of the distal radius. However, procedures to treat tumors involving other sites are continually being developed. Full staging should be performed before limb-sparing surgery to ensure that gross disease at another site is not evident. For distal radial sites, ideal candidates are dogs that do not have a large amount of soft tissue involvement or evidence of pathologic fracture. Either of these situations greatly increases the probability of local recurrence. The tumor should involve less than 50% of the radial length to facilitate implant placement and decrease the risk of implant failure.20 The approach to the antebrachium may be cranial, craniolateral, or craniomedial and extends from just distal to the elbow joint to the level of the metacarpophalangeal joints, over metacarpal III or IV. The decision to make the approach from the lateral or medial aspect is based on preferred position and the surgeon’s preference. A more medial approach allows better access to the distal radius during dissection between the radius and the ulna. A more lateral approach allows better access to the proximal part of the radius; this is important in smaller dogs or when a large portion of the radius is removed. If possible, the cephalic vein is preserved. If a previous biopsy was performed, the biopsy tract is resected with the distal radius. The skin incision is continued through the subcutaneous tissue and the antebrachial fascia to the level of the extensor tendons and the tumor and its capsule. No attempt is made to maintain the function of the extensor muscles and tendons, and soft tissues that are involved in the tumor capsule are resected en bloc with the tumor, with a minimum 2 to 3 cm margin from the tumor. The supinator and pronator muscles are elevated from the proximal radius. Care is taken to preserve the radial nerve. The distal radius is osteotomized a minimum of 3 cm from the proximal extent of the tumor. In cases where an endoprosthesis is used, endoprosthesis length dictates the osteotomy site because the resulting defect must match the endoprosthesis length (see section on endoprosthesis). A sample of the bone marrow from the proximal osteotomy site is taken for histopathology to assess this margin. The joint capsule and extensor tendons at the level of the antebrachiocarpal joint are transected. This dissection is continued medially and laterally. Antebrachiocarpal joint flexion will facilitate disarticulation. Dissection continues on the caudal aspect of the radius by rotating the radius cranially from the osteotomy site around the radiocarpal joint, or by working caudal to the radius. Large vessels associated with the tumor are ligated with suture or metal clips. Attention is paid to preservation of the blood supply as much as possible. The distal radius is examined for evidence of fracture or tumor breakthrough. If tumor fracture or breakthrough is encountered at any point during resection of the distal radius, conversion to an amputation should be considered because the risk of local recurrence is much greater. If conversion to amputation is not an option, the site must be copiously lavaged, and the surgeon’s gloves and instruments should be changed once the tumor segment is removed. Local slow-release cisplatin therapy may also be employed in this situation. The ulna is sectioned sagitally using an osteotome. The medial ulnar cortex is removed with the distal radius. Alternatively, if the tumor extends into the ulna, the ulna is cut with a bone saw proximal to the level of tumor extension. Liptak et al., in a study of cadaver limbs, demonstrated that there is no biomechanical advantage to preserving the ulna when an allograft or an endoprosthesis is used for limb-sparing surgery.60 This finding has led to the practice by some surgeons of removing the ulna en bloc at the level of or distal to the radial osteotomy. However, further study is required to assess this practice, as it may have implications in vivo with regard to the blood supply. Limb-sparing surgery with a cortical allograft (Figure 72-7) was first reported in dogs in 1989.52 This technique initially involved preoperative radiation to downstage the tumor. Preoperative radiation is not currently administered prior to limb-sparing surgery because of the high complication rate associated with this technique. Because this was the first limb-sparing technique developed in dogs, it is often referred to as traditional limb-sparing surgery. Figure 72-7 Mediolateral radiograph of the antebrachium and manus after allograft limb-sparing surgery. Limb-sparing technique20,55 uses a fresh frozen cortical allograft from an in-house or commercial bone bank. The allograft is selected on the basis of side and size of the donor radius matching those of the recipient. During resection, the allograft is thawed in a 1 L solution of sterile saline, 1000 mg of neomycin, 3000 mg of ampicillin, and 500,000 units of polymyxin B at room temperature. After resection of the distal radius and the tumor capsule, the allograft is prepared. The articular cartilage is removed by performing an osteotomy, and any remaining soft tissue remnants are removed. The medullary canal is reamed using a bone curette to remove any fat or cellular debris. The graft is cut to fit the defect and is secured to the bone plate using two to three screws (of appropriate type to match the plate). The allograft should fit into the defect without a gap at the distal or proximal osteotomy site. Revision of the osteotomy sites may be necessary to ensure good bone-to-bone contact. A long (18 to 24 hole) 3.5 broad dynamic compression plate, or locking plate, or a limb-sparing plate (which has 24 holes) is used. The limb-sparing plate has a distal taper and can accommodate 2.7 or 3.5 cortical screws in the 7 distal screw holes. The plate should extend a minimum of 50% of the length of the metacarpal bone. The remaining 10 screw holes can accommodate 3.5 or 4.5 cortical screws, or 4.0 cancellous screws. The most distal 3.5/4.5 hole should be positioned over the radial carpal bone. The bone plate may need to be shortened from the proximal end, depending on the length of the radius. The plate should be shortened before surgery, if possible. The bone plate can be bent at the level of the carpus to 10 to 15 degrees, or it can be applied straight. Once alignment of the limb and position of the bone plate and allograft are satisfactory, the screws affixing the bone plate to the allograft are removed, and the medullary cavity of the allograft is filled with antibiotic-impregnated bone cement. The screws attaching the bone plate to the allograft are replaced during curing of the cement. Use of bone cement with cortical allografts has been shown to increase the strength of the construct and decrease screw pull-out.47 The graft and the bone plate are then affixed to the proximal radius and the third or fourth metacarpal bone. A minimum of 3 screws proximal and 4 screws distal to the ostectomy site are recommended. The plate should extend distally to just proximal to the metacarpophalangeal joint. Care is taken to avoid placing screws in the ulna, as this will promote screw loosening over time. The advantage of this technique is that it is the longest standing technique, and so it has been modified over the years to improve upon noted complications. Disadvantages of this technique include the high infection rate (approximately 50%)53 and the fact that the allograft is large and is not incorporated into the host bone within the patient’s remaining life span. Lack of incorporation leads to development of a large bone sequestrum that is prone to infection. Implant failure can occur as the result of implant fatigue and implant loosening in longer-term survivors. Another disadvantage of this technique is that it requires maintenance of a bone bank or purchase of frozen allograft. Preparation of the allograft at the time of insertion into the patient is time-consuming and increases the duration of an already lengthy procedure. These specific disadvantages and complications have led to the development of all of the other limb-sparing surgery techniques.
Musculoskeletal Neoplasia and Limb-Sparing Surgery
Diagnosis
Biopsy
Staging
Local Staging
Surgical Treatment
Repair of Pathologic Fracture
Fracture-Associated Osteosarcoma
Scapulectomy
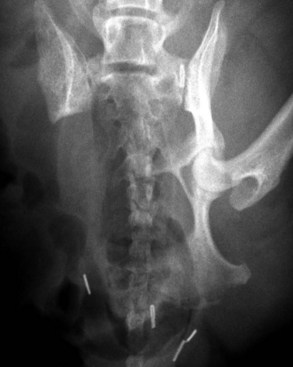
Hemipelvectomy
Limb-Sparing Surgery
Surgical Technique—Distal Radial Resection20,55
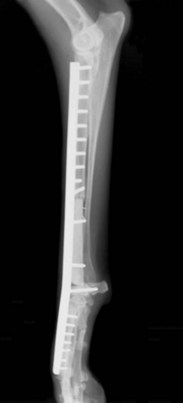
Cortical Allograft
Surgical Technique
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Musculoskeletal Neoplasia and Limb-Sparing Surgery
Only gold members can continue reading. Log In or Register to continue