Immunologic Drugs
Learning Objectives
After studying this chapter, you should be able to
1. Explain the principles associated with vaccination.
2. Describe the differences between vaccine types.
3. Discuss the advantages and disadvantages of the different types of vaccines.
4. List common diseases that have available vaccines.
5. Describe the different routes of administration of vaccines.
Key Terms
Active immunity
Adjuvant
Anaphylaxis
Antibody
Antigen
Avirulent
Bacterin
Monovalent
Passive immunity
Polyvalent
Preservative
Recombinant DNA technology
Virulence
Principles of Vaccination
Keeping animals healthy through the proper use of immunization programs is an important aspect of veterinary medicine. Veterinary technicians must have knowledge concerning vaccine types and the diseases against which animals are vaccinated. Clients ask many questions regarding this area of their pet’s care. It should be remembered that immunization should never take the place of regularly scheduled, routine veterinary checkups (McCurnin and Bassert, 2006). As animals enter into their geriatric years, regular laboratory profiles should be done to determine the health of major organ systems. Preventive medicine also includes a good physical examination as well as procurement of a complete history on the animal. Therefore, immunization programs are only one aspect of the overall health care that should be afforded companion animals. Livestock should be properly immunized to achieve a healthy herd.
Vaccinations are an important part of the preventive health care program for companion animals and food animals alike. Vaccines are given to lessen the chance of a particular disease occurring. A patient’s response is determined by (1) health and age, (2) the type of vaccine given, (3) the route of administration, (4) concurrent incubation of infectious disease, (5) exposure to an infectious disease before complete immunity is reached, and (6) drug therapy. The ideal vaccine is safe and effective on challenge and has no undesirable side effects. Immunology is a very complex field of study. This chapter outlines only the basics of common vaccines and immunostimulants. Referencing an immunology textbook may be helpful if further information is desired.
In properly vaccinated females, antibodies are passed to their offspring in the form of maternal antibodies found in colostrum. It is important not to vaccinate very young animals when maternal antibodies are still present. The neonate’s immune system is not capable of producing an immune response when maternal antibodies are blocking this mechanism. (See the charts within this chapter for specific times that various species should be vaccinated.)
Because of the risk of vaccine-associated sarcomas when inactivated feline vaccines are administered, it is becoming increasingly important that each vaccine be given at a certain place on a cat’s body. Although the prevalence of sarcomas after vaccination has been reported at 1 case per 10,000 vaccines administered (McCurnin and Bassert, 2006), caution is warranted. The National Vaccine-Associated Sarcoma Task Force studying vaccine site tumors recommends that no vaccine be given in the intrascapular space. Rabies vaccine should be administered in the distal right rear leg, feline leukemia virus (FeLV) vaccine should be administered in the distal left rear leg, and all other vaccines should be administered in the right shoulder. It is also recommended that any vaccine lump present 3 months after the time of vaccination be removed, after a biopsy has been performed to reveal the extent of surgery that may have to be performed. Rear-leg amputation for sarcoma removal provides a better cure rate than surgery on the intrascapular space (McCurnin and Bassert, 2006). Some veterinarians follow the same vaccine site administration for dogs as is observed in cats.
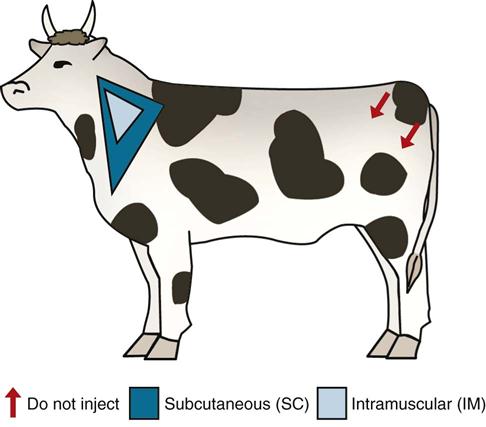
In cattle, the location of vaccine administration is also important. With the advent of the Meat Quality Assurance Program, proper administration of injections and vaccines is critical in all food animal species. All intramuscular and subcutaneous injections in cattle should be given in the neck, if possible. Administration of most vaccines requires observation of a slaughter withdrawal, sometimes up to 60 days postvaccine (Figure 17-1).
The future of vaccination involves development of protocols that individualize vaccine schedules instead of having every animal vaccinated for every disease. Much discussion is ongoing in the veterinary community regarding vaccination of dogs and cats every 3 years rather than yearly. However, some state and county regulations may still mandate the yearly rabies vaccine regimen, and other states may legislate every 3 years. It is hoped that additional research will yield optimal revaccination intervals. Some studies have suggested that with some vaccines, protective immunity may last for years and annual revaccination may not be necessary. With new information and technology, the twenty-first century will see many changes in vaccines.
Common Vaccine Types That Produce Active Immunity
 Inactivated
Inactivated
Manufacture of inactivated vaccines involves killing the organisms with chemicals that leave the antigens mostly unchanged. The antigens stimulate protective immunity. Inactivated vaccines are also referred to as killed, or dead, vaccines.
Advantages
Disadvantages
Dosage Forms
See the vaccine charts within this chapter for a more comprehensive listing of the various vaccine types associated with each disease.
 Live
Live
A live vaccine is prepared from live microorganisms or viruses. These organisms may be fully virulent or avirulent. Few vaccines of this origin are in use, with the exception of several poultry vaccines.
Advantages
Disadvantages
Dosage Forms
• Brucella abortus Vaccine: Brucella abortus strain RB-51
• Ovine Ecthyma Vaccine: ovine ecthyma virus or sore mouth infection
• Chick Ark Bronc: infectious bronchitis (Massachusetts and Arkansas types)
See vaccine charts within this chapter for a more comprehensive listing of the various vaccine types associated with each disease.
 Modified Live
Modified Live
In modified live vaccines, organisms undergo a process (attenuation) to lose their virulence so that when introduced to the body via inoculation, they cause an immune response instead of disease.
Advantages
Disadvantages
Dosage Forms
See vaccine charts within this chapter for a more comprehensive listing of the various vaccine types associated with each disease.
 Recombinant
Recombinant
In recent years, vaccines produced by recombinant DNA technology have become available for veterinary medicine. These vaccines are recognized as being safe, highly specific, potent, pure, and efficacious. These attributes may be the reason why recombinant vaccines are more desirable than any other vaccine type. Recombinant vaccines are divided into three categories:
Type I recombinant (subunit) vaccines—These vaccines are derived by inserting a foreign gene from a specific pathogen into a recombinant organism (e.g., yeast, bacterium, a virus). The recombinant organism multiplies, and the product of the gene is extracted, purified, and prepared for administration as a vaccine.
Type II recombinant (gene-deleted) vaccines—The manufacturing of these vaccines involves deletion of specific genes from a pathogenic organism. This manipulation produces a vaccine that has a low risk of producing disease but can still stimulate a protective immune response.
Type III recombinant (vectored) vaccines—These vaccines are derived from the insertion of specific pathogenic genetic material into a nonpathogenic or gene-deleted organism (e.g., poxvirus). This altered organism then is propagated in vitro and is used to manufacture the vaccine (Van Kampen, 1998).
Advantages
Disadvantages
Dosage Forms
• Type I: RM Recombitek Lyme: Borrelia burgdorferi
• Type III: RM Recombitek C4: canine distemper, adenovirus type 2, parainfluenza, and parvovirus
• Type III: Raboral V-RG: oral vaccine for rabies virus (used in baiting devices for wildlife)
• Type III: Newcastle disease–fowl pox vaccine (recombinant): Newcastle disease and fowl pox
• Type III: Trovac-AIV H5: avian influenza subtype H5 and fowl pox
See vaccine charts within this chapter for a more comprehensive listing of the various vaccine types associated with each disease.
 Toxoid
Toxoid
A toxoid is a vaccine that is used to produce immunity to a toxin rather than a bacterium or a virus. The toxin is treated with heat or chemicals to destroy its damaging properties without eliminating its ability to stimulate antibody production.
An anaculture combines toxoid and killed bacteria in a single dose prepared from highly toxigenic cultures and culture filtrates.
Characteristics
Dosage Forms
Common Vaccine Types That Produce Passive Immunity
 Antitoxin
Antitoxin
An antitoxin is a specific antiserum aimed at a toxin that contains a concentration of antibodies extracted from the blood serum or plasma of a hyperimmunized, healthy animal (usually a horse).
Characteristics
Dosage Forms
 Antiserum
Antiserum
An antiserum is a serum that contains specific antibodies extracted from a hyperimmunized animal (usually a horse) or an animal that has been infected with microorganisms that contain antigen.
Characteristics
Dosage Forms
Other Types of Vaccines
 Autogenous Vaccine
Autogenous Vaccine
An autogenous vaccine contains organisms isolated from an infected animal on a farm where a disease outbreak is occurring. This carefully prepared vaccine contains antigens needed for protection at that particular location.
 Mixed Vaccine
Mixed Vaccine
A mixed vaccine contains a mixture of different antigens. It is also referred to as a polyvalent vaccine. Each component of a mixed vaccine is required to achieve an immune response comparable with that of a vaccine containing a single antigen (monovalent vaccine).
Administration of Vaccines
The intramuscular and subcutaneous routes are by far the most common methods for vaccine administration. These routes are easily accessible and provide systemic immunity, which is important in many diseases. Some diseases also respond well to local immunity. Vaccines against feline rhinotracheitis and calicivirus, canine infectious tracheobronchitis, and infectious bovine rhinotracheitis may be administered intranasally, or, in some cases, intraocular administration may be used to provide local immunity. After administration of these vaccines, the animal may experience a slight bout of watery eyes and occasional sneezing for a few days.
All of the previously mentioned routes of vaccine administration necessitate that each animal be handled individually. When a large number of animals require vaccination, these routes may not be feasible. Some vaccines may be mixed with drinking water or feed. Others can be aerosolized and inhaled by the animal. For example, on mink ranches, vaccine for canine distemper and mink enteritis may be administered in this manner, or poultry houses may vaccinate for Newcastle disease by aerosolization. The margin for incomplete vaccination is greater when aerosolization or mixing with feed or water is used. Some animals may not drink or eat enough to acquire adequate protection, or the aerosolized vaccine may not distribute equally throughout the room. Vaccine failure may be implicated if these animals contract the disease, whereas in reality, the animal did not receive enough vaccine to gain adequate immunity.
When conventional measures are used for vaccination, it is very important to carefully read the insert provided with the vaccine. Some vaccines may be administered intramuscularly or subcutaneously, but others may be administered by only one route. For example, some rabies vaccines require administration by an intramuscular route to be most effective. Subcutaneous injections should be given according to the manufacturer’s instructions. Care should be used when one is vaccinating a cat to prevent vaccine-induced tumors.
If a vaccine requires reconstitution, this should be done with the diluent provided by the manufacturer. The vaccine should not be reconstituted until just before it is administered (see Chapter 2 for the proper reconstitution procedure). The full recommended dose should be given. Splitting a vaccine dose may result in an animal’s failure to develop an adequate immune response and may lower its protection.
Mixing different vaccines to minimize the number of injections the animal receives is not recommended. This procedure can cause antigen blocking, resulting in one component interfering with the action of another; in this case, the animal does not receive adequate antigen to attain an effective immune response. Mixing of different vaccines may cause an increased chance of an allergic response. When different types of vaccines are administered, each vaccine should be administered at a separate site. It is also advisable to note the locations of administration and vaccine lot numbers on the patient’s medical record. If a reaction or a problem develops later, a reference will be available to aid in evaluation of the problem.
When food animals are vaccinated, several factors must be considered. Almost all vaccine labels contain information advising not to vaccinate within 21 days of slaughter. Vaccines such as those for B. abortus are subject to federal limitations and regulations, and complete records are maintained on administration of these vaccines. Brucellosis vaccines are restricted to use by or under the direction of a licensed veterinarian. Carcass destruction is also a factor that involves food animal producers. Injection site lesions may cause damage to muscle tissue, requiring that area to be trimmed and discarded. If a vaccine may be administered intramuscularly or subcutaneously, the subcutaneous route would produce less tissue reaction and would eliminate muscle damage. This is important when one is dealing with animals used for meat consumption. Most vaccines on the market today can be given subcutaneously.
Biologic Care and Vaccine Failure
Biologics (especially modified live and live vaccines) are sensitive to inactivation by heat or sunlight. Clients purchasing vaccines should be provided with a cold pack if needed and should be warned against leaving such biologics in vehicles or in sunlight, where they may become warm and inactivated. Even the performance of killed products can be altered if proper handling and storage measures are not practiced. When these vaccines are shipped from the manufacturer, cold packs are put in the box to provide some refrigeration during shipment. In some areas, it may be advisable to anticipate how much vaccine may be needed during the hot summer months and to stock up on that amount during early spring to prevent shipments from overheating during summer transportation. Once a shipment is received, it should be quickly unpacked and placed under refrigeration. Vaccines should never be frozen because cells may rupture when the vaccine thaws, releasing toxins that can damage tissue or cause tissue death.
Inappropriate care of vaccines may lead to inactivation of the vaccine and may be perceived as a vaccine failure. Actual vaccine failure is relatively uncommon. If vaccines are purchased from a reputable manufacturer, one can be fairly sure that the vaccine provided will be effective. Failure usually occurs because of improper handling, storage, or administration.
Live vaccines are especially affected by concurrent antibiotic therapy. Live and modified live vaccines can be inactivated by chemicals used to clean or sterilize syringes and by the use of excessive alcohol or other disinfectants to swab the skin before injection. As was mentioned earlier, the route of administration may affect the ability of an animal to achieve an adequate immune response. Immunosuppressed, parasitized, stressed, or malnourished animals and those incubating disease are not able to mount an adequate immune response to prevent disease. Clients should always be advised that such problems can occur. In most cases, an adequate immune response is not achieved before 10 to 14 days. An 8-week-old puppy may not develop a strong immune response to protect against an infectious disease if challenged by maternal antibodies because it is not feasible to check immune titers to determine the presence of maternal antibodies. Therefore, it is recommended that puppies receive boosters every few weeks until they are about 4 to 5 months of age. Boosters allow vaccines to produce an optimum immune response. Clients often find it difficult to understand why they need to bring their pet in for boosters. If the reasons are explained and if clients are advised about why they should isolate their pet from animals with questionable vaccination histories, many cases of infectious disease would be prevented among young animals. Clients often perceive one vaccine to be enough or do not understand that their animal is not protected immediately after an injection has been received. Technicians should include this information when educating clients on animal and pet care.
Adverse Vaccination Responses
The most notable risks involving vaccination include residual virulence and toxicity, allergic reactions resulting from hypersensitivity, disease in immunosuppressed animals, possible effects on a fetus, and abortion. The veterinarian assesses these risks before a vaccine is administered. In most cases, the benefits of vaccination far outweigh the risks, but it may occasionally be necessary to omit or delay vaccination because of some of the factors just mentioned.
One of the most common reactions noted with vaccine administration is the sting felt by the animal after injection. This is most often caused by inactivating agents used in manufacturing the vaccine. Manufacturers are constantly researching ways to decrease these undesirable effects while still producing a quality product. This stinging reaction is short lived and does not usually cause a problem unless the animal reacts violently. Other common but not usually serious reactions include a slight fever, lethargy, and soreness at the injection site. These usually subside within 1 day. Hypersensitivity may be caused by several factors, including immunizing antigens, antigens acquired during the manufacture of the vaccine, and reactions to adjuvants used in the vaccine. Some animals may experience an anaphylactic shock reaction after receiving a vaccine, although this is uncommon. Clinical signs of anaphylaxis include vomiting, salivation, dyspnea, and incoordination. Epinephrine is usually the antidote of choice in cases of anaphylactic shock and should always be on hand when horses are vaccinated (McCurnin and Bassert, 2006). Other possible adverse side effects include vaccine-associated fibrosarcoma in cats and immune-mediated hemolytic anemia in dogs (Ford, 1998). The possible causes for these effects are under investigation.
Vaccinations for Preventive Health Programs
Canine
As was stated earlier, vaccination is an important part of any preventive health program. Many vaccines may be available as a monovalent or polyvalent product. For dogs, a common polyvalent vaccine includes canine distemper (D), respiratory diseases caused by adenovirus type 2 (A2), canine parainfluenza (P), leptospirosis (L), canine parvovirus (P), and coronavirus (C). It should be noted that vaccination with the A2 virus also protects the dog against infectious canine hepatitis (ICH). This vaccine may be referred to as DA2PPL/C. Many different combinations and product names are available. Most veterinarians choose a particular manufacturer from which to buy vaccine products. This helps to lessen the confusion caused by different names used to designate the manufacturers’ products. Other available canine vaccines include those given for canine infectious tracheobronchitis (B. bronchiseptica), rabies, and Lyme disease (B. burgdorferi). Manufacturer recommendations should be followed regarding age, route of administration, and follow-up boosters needed for each individual vaccine. Box 17-1 provides an example of a vaccination program for dogs.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree