Chapter 18 Icterus
Definition
The term icterus is used interchangeably with jaundice to refer to yellow discoloration of skin, mucous membranes, and sclerae caused by an accumulation of bilirubin pigment in plasma (hyperbilirubinemia) and tissues. Icterus generally becomes visible when serum bilirubin concentration exceeds 2 to 3 mg/dL (35 to 50 µmol/L), or greater than five- to 10-fold above reference range. Unlike other nonspecific clinical signs of hepatobiliary disease (e.g., inappetence, lethargy, weight loss, vomiting, diarrhea, and dehydration), icterus correlates with hyperbilirubinemia and is therefore a highly specific clinical sign consistent with decreased excretion of bilirubin in association with hepatobiliary disease or increased formation in association with severe hemolysis. Despite excellent specificity, icterus and hyperbilirubinemia are relatively insensitive indicators, found only in moderate to severe hepatic insufficiency and overall in less than 50% of dogs and cats with hepatic disease.1
Pathophysiology and Mechanisms
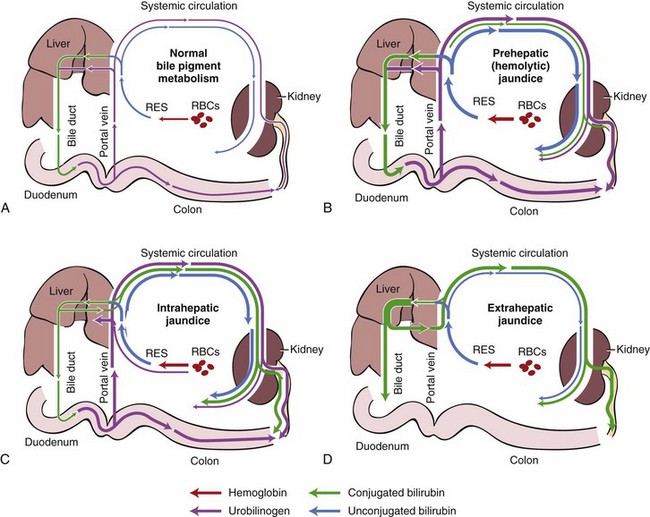
Bilirubin is an end product of hemoglobin metabolism. Icterus occurs when bilirubin formation exceeds hepatobiliary excretion. Three pathophysiologic mechanisms of hyperbilirubinemia and icterus are termed (a) prehepatic, which results from accelerated red blood cell destruction (hemolysis) and increased bilirubin production; (b) hepatic, which is caused by intrinsic hepatocellular disease and reduced hepatocyte uptake, conjugation, and secretion of bilirubin; and (c) posthepatic, which is caused by extrahepatic cholestasis and disruption of bile flow through the extrahepatic biliary system (Fig. 18-1).1,2 This mechanistic classification helps to explain how different categories of disease can cause hyperbilirubinemia and icterus, although all three mechanisms may be operative in any individual.
Normal Bilirubin Metabolism
Bilirubin is a yellow pigment derived mainly from the degradation of red blood cell hemoglobin (see Fig. 18-1A). Under normal conditions approximately 80% of bilirubin comes from erythrocyte hemoglobin, while the remaining 20% is derived from the breakdown of other heme-containing proteins, such as myoglobin, P450 cytochromes, peroxidase, and catalase.2 In macrophages of the mononuclear phagocytic system of the spleen, liver, and bone marrow, heme from senescent erythrocytes is cleaved by microsomal heme oxygenase to yield iron and biliverdin, a green pigment, which is then reduced to bilirubin, a yellow pigment, by biliverdin reductase. Daily production of bilirubin in adult mammals is approximately 3 to 5 mg/kg.3 Unconjugated bilirubin, also called free bilirubin or indirect-reacting bilirubin, is insoluble in plasma. As it is released into the circulation it is tightly bound to albumin for transport to the liver, which completes the prehepatic phase of bilirubin metabolism.2
In the hepatic phase, bilirubin metabolism depends upon hepatocyte uptake, conjugation, and secretion, followed by posthepatic excretion in bile (see Fig. 18-1A).2 Unconjugated bilirubin is removed from circulation by the liver (uptake), which involves dissociation from albumin at the sinusoid-hepatocyte interface and transport across the cell membrane by a carrier protein. Uptake capacity of hepatocytes greatly exceeds excretory capacity.4 Once inside the hepatocyte, bilirubin is bound to ligandin, a cytosolic transporter protein that regulates the rate of bilirubin uptake.2 Within the hepatocyte glucuronyl transferase catalyzes conjugation of bilirubin with glucuronic acid to form water-soluble bilirubin diglucuronide.5 Conjugated bilirubin, also called water-soluble or direct-reacting bilirubin, is then secreted into the bile canaliculi which is both an energy-dependent process and rate-limiting step in the transfer of bilirubin from plasma to bile.2 In the posthepatic phase, conjugated bilirubin is then transported with bile through the extrahepatic bile duct system, stored in the gallbladder, and excreted into the intestines through the common bile duct. In dogs, intestines and kidneys can also conjugate and excrete a small amount of bilirubin.6
Conjugated bilirubin that enters the intestinal tract with bile is poorly absorbed and passes largely into the large intestine where colonic bacteria convert it to colorless derivatives called urobilinogens.2 A small portion of bilirubin within the intestine is deconjugated by enteric bacteria, reabsorbed by enterocytes, and returned to the liver by the enterohepatic portal venous circulation. The majority of intestinal urobilinogen is oxidized and passed in the feces as stercobilin, a pigment that imparts the normal brown color to feces. Normally, a small amount of intestinal urobilinogen (10% to 20%) is reabsorbed into the enterohepatic circulation and handled once again by the liver, except for approximately 1% to 5% of absorbed urobilinogen that is excreted into urine by the kidneys.7
Prehepatic Icterus
Icterus is characterized as prehepatic when the primary abnormality is overproduction of bilirubin resulting from hemolysis (see Fig. 18-1B). Accelerated erythrocyte destruction increases formation of bilirubin from heme. If this bilirubinemia overwhelms the liver’s functional capacity for uptake, conjugation, and secretion, bilirubin is refluxed from the liver into the circulation resulting in hyperbilirubinemia and icterus.2 The hemolytic event must be acute and severe to overwhelm the large reserve capacity of the normal liver. Severe hemolytic events can be complicated by anemic hypoxia of the liver, which can secondarily compromise hepatic function and contribute to intrahepatic cholestasis. Icterus and bilirubinemia associated with severe hemolysis are typically a mixture of unconjugated and conjugated bilirubin fractions resulting from combination of overproduction of bilirubin and intrahepatic cholestasis.8 Increased excretion of conjugated bilirubin into the intestinal tract increases urobilinogen formation, which results in increased urobilinogen in urine and increased stercobilins in feces, which imparts a dark orange–brown color to the feces.1
Hepatic Icterus
Icterus is characterized as hepatic when the primary abnormality is intrahepatic cholestasis associated with hepatocellular injury, necrosis, or dysfunction (see Fig. 18-1C). Moderate to severe hepatic disease can impair various steps in handling of bilirubin particularly at the level of the canaliculi and intrahepatic bile ductules. Intrahepatic cholestasis and icterus develop most readily with hepatic parenchymal lesions that affect the periportal region of the liver lobule because bile flows from the centrilobular region toward the intrahepatic bile ductules in the portal triad.3,9 Intrahepatic cholestasis causes reflux of conjugated bilirubin into the circulation. Unconjugated bilirubin also refluxes into circulation because increased bilirubin accumulation within hepatocytes interferes with the conjugation process. In cholestatic hepatic disease, concurrent retention of bile acids may further damage hepatocytes. Hyperbilirubinemia that occurs in most diseases of the liver is a mixture of conjugated and unconjugated bilirubin in varying proportions.8 Some of the circulating conjugated bilirubin is excreted by kidneys and is detected in urine along with urobilinogen.
Posthepatic Icterus
Icterus is characterized as posthepatic when the primary abnormality is impaired excretion of bilirubin caused by extrahepatic biliary disease that interrupts or obstructs the flow of bile (see Fig. 18-1D). Extrahepatic cholestasis primarily causes reflux of conjugated bilirubin into the circulation, but bile retention also interferes with conjugation and injures hepatocytes which can cause secondary unconjugated bilirubinemia. Thus posthepatic icterus is characterized by mixed hyperbilirubinemia with the conjugated fraction predominating. Some of the circulating conjugated bilirubin is excreted by the kidney and detected in urine; however, urobilinogen is absent in urine because obstructive extrahepatic biliary disease prevents delivery of precursor bilirubin into the intestine. In complete bile duct obstruction, failure of bile to enter the intestinal tract can result in pale gray–colored acholic feces.1,10
Differential Diagnosis
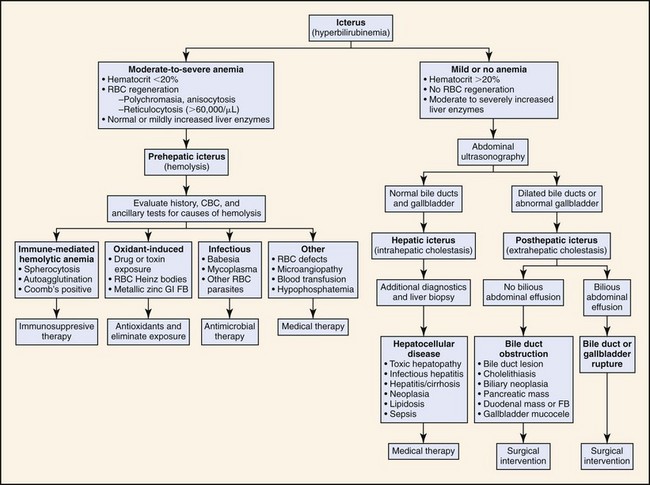
In dogs and cats, hyperbilirubinemia and icterus can be associated with many diseases that cause either hemolysis or intra- or extrahepatic cholestasis (Box 18-1). Figure 18-2 outlines an algorithmic approach for differential diagnosis of icterus. The first step is to consider prehepatic (hemolytic) icterus, which is usually indicated by the presence of moderate to severe anemia (hematocrit <20%) and evidence of red blood cell (RBC) regeneration (polychromasia, anisocytosis, and reticulocytosis) in the absence of blood loss.11 The cause of hemolysis may be suggested by other hematologic findings such as autoagglutination, spherocytosis, Heinz bodies, RBC parasites, or RBC fragments (schistocytes). In icteric patients without clinical and hematologic evidence of severe hemolysis, diagnostic evaluation should focus next on identifying a primary hepatobiliary disease that can cause either intra- or extrahepatic cholestasis. Ultrasonography is especially useful for evaluating the liver, gallbladder, and bile ducts. Posthepatic icterus is unlikely if ultrasonography fails to reveal evidence of obstruction (bile duct distention), rupture (bile peritonitis), or other disease (e.g., cholelithiasis, gallbladder mucocele) in the extrahepatic biliary tract. Ultrasonography also helps to identify pancreatic or duodenal masses that could be obstructing the common bile duct. Once prehepatic hemolysis and posthepatic disruption of bile flow are excluded, diagnostic evaluation should focus on intrinsic hepatic parenchymal diseases that can cause icterus. Hepatic biopsy is required in many such cases.
Box 18-1
Differential Diagnosis of Icterus and Hyperbilirubinemia
Prehepatic (Hemolytic) Icterus
• Immune-mediated hemolytic anemia
• RBC oxidative injury by toxins and drugs (Heinz body anemia)
• Envenomation (spiders, snakes, bees)
• Hypophosphatemia (insulin therapy, refeeding syndrome)
• Microangiopathic RBC fragmentation
• Incompatible blood transfusion
Hepatic (Hepatocellular) Icterus
Posthepatic (Obstructive) Icterus
• Cholelithiasis, choledocholithiasis
• Bile duct inflammation, stricture, or cyst
• Biliary fluke infestation (Platynosomum concinnum, Amphimerus pseudofelineus)
• Gallbladder disease (cholecystitis, mucocele)
• Pancreatic disease (carcinoma, abscess, pancreatitis)
• Duodenal disease (neoplasia, foreign body, duodenitis)
DIC, Disseminated intravascular coagulation; RBC, red blood cell.
Evaluation of the Patient
Icterus is sometimes noticed by an observant pet owner and quite readily detected on physical examination. Following a complete history and physical examination, initial diagnostic evaluation of the icteric patient should include a complete blood count (CBC), serum chemistry, urinalysis, and diagnostic imaging of the abdomen, especially ultrasonography. This minimum database usually enables classification of icterus as prehepatic, hepatic, or posthepatic in origin (see Fig. 18-2). Hepatic cytology and biopsy are often indicated for further evaluation of hepatic parenchymal disease. In most icteric patients coagulation status should also be evaluated. Abdominocentesis and fluid analysis are indicated in patients with abdominal effusion, especially if gallbladder or bile duct rupture is suspected. Laparoscopic or surgical intervention is usually the most effective way to confirm, characterize, and decompress posthepatic biliary obstruction.
History
Icterus is an important clinical sign of hemolysis and hepatobiliary disease in dogs and cats. Some owners may report changes in the color of urine or feces. Severe bilirubinuria can cause orange or green discoloration of urine (pigmenturia), whereas intravascular hemolysis can cause dark red or brown urine (hemoglobinuria).1 Feces may appear dark orange–brown or green with increased excretion of bile pigments into the intestinal tract. Prolonged absence of bile pigments in the intestines in complete bile duct obstruction can result in pale gray–colored acholic feces.10
Other clinical manifestations of hepatobiliary disease can include inappetence, lethargy, weight loss, vomiting, diarrhea, polyuria-polydipsia, hepatic encephalopathy, abnormal bleeding tendencies, and abdominal distention related to hepatomegaly or ascites.1,12 Genetic and breed predispositions for specific diseases (e.g., copper hepatopathy) should be also considered (see Chapters 61 and 62). Owners should be questioned about potential for exposure to infectious agents, hepatotoxins (e.g., xylitol, aflatoxin-contaminated dog food, amanita mushrooms, Microcystis blue-green algae, sago, or cycad palm), or medications that might cause hepatotoxicity (e.g., nonsteroidal antiinflammatory drugs, phenobarbital, and many others) (see Chapter 61).
Patients with hemolytic icterus typically have a history of exercise intolerance, lethargy, weakness, and pallor of mucous membranes.11 When hemolysis is suspected, history should ascertain potential for recent exposure to blood transfusions, oxidant substances, vector-borne hemotropic RBC parasites, venomous snakes and insects, zinc ingested with metallic objects, or zinc oxide sun protectants (see Box 18-1).11 Breed predispositions to hereditary RBC defects should be taken into account. Signs that could indicate an underlying cause of microangiopathic hemolytic anemia should also be considered. Insulin therapy in ketoacidotic diabetic patients or intensive hyperalimentation can precipitate hypophosphatemic hemolysis when serum phosphorus is less than 1.5 mg/dL.11
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree