Chapter 55 Esophagus
Structure and Function
The esophagus conveys food from the pharynx to the stomach. This relatively narrow tube begins dorsal to the cricoid cartilage of the larynx and accompanies the trachea, at first inclining to the left but regaining a median position above the trachea before or shortly after entering the thorax. Within the thorax it runs in the mediastinum and, continuing beyond the tracheal bifurcation, it passes over the heart before penetrating the esophageal hiatus of the diaphragm to join the stomach at the gastroesophageal junction (the cardia).1 The cardia is pulled caudally and may be found in the abdomen, but this is not a consistent finding in all canine breeds.2
Structure of the Esophagus
Macroscopic Structure
The structure of the esophagus conforms to a pattern that is common to the alimentary canal (see Chapter 1). The outer coat of loose connective tissue, the adventitia, is present in the cervical part but is largely replaced by serosa in the thorax. In dogs, the muscle of the esophagus is striated from the cricopharyngeus muscle to the junction with the stomach. In cats, the striated muscle is replaced by smooth muscle within the thorax, such that roughly the proximal one-third is striated muscle and the remainder is smooth muscle. The muscular layer of the esophagus is composed of two strata, both of which are spiral, and they wind in opposite directions in the first part of the esophagus. Closer to the stomach the outer stratum becomes more longitudinal and the inner more circular.1,3 Two sphincters are suggested on the basis of functional studies; a cranial (upper) and a caudal (lower) sphincter, but these are less convincingly demonstrated morphologically. The cranial sphincter is provided by fibers of the cricopharyngeal muscle. In the cat the cricopharyngeal muscle and the esophageal muscle are not morphologically separate.3 A thickening of the esophageal muscle mass at the junction of the esophagus with the stomach suggests a sphincter, but food is impeded slightly more cranial to this—just cranial to the diaphragm.1 In dogs, the thickness of the circular striated muscle layer increases from the distal esophagus to the gastroesophageal junction and cardia.3
The inner part of the wall of the esophagus consists of submucosa and mucosa, divided by the fenestrated muscularis mucosae, which is more prominent in the thoracic part of the esophagus. The submucosa loosely connects the mucosa and the muscularis. This structure allows the relatively inelastic mucosa to be thrown into longitudinal folds when the esophagus is contracted. The submucosa contains blood vessels, nerves, and glands. Glands are found over the entire length of the mucosa in dogs, but only in the cranial one-third of the esophagus in cats.3 In both dogs and cats, the muscularis mucosae is more prominent in the thoracic esophagus. The mucosa is composed of keratinized, stratified squamous epithelium, which contains the openings, at about 1-mm intervals, of the ducts of the esophageal glands. The cat has transverse folds in the mucosa in the caudal half of the esophagus. These folds are permanent and independent of the state of contraction of the esophagus.3
Microscopic Structure
The outer coat of the esophagus, the adventitia, consists of connective tissue with elastic fibers. In dogs the muscularis of the esophagus is entirely striated muscle, whereas in cats the striated muscle is replaced by smooth muscle within the thorax. Fiber type composition has been studied by histology and immunohistochemistry, and myosin type has been studied by electrophoretic peptide mapping and two-dimensional gel electrophoresis, respectively.4
In both dogs and the cats, the striated esophageal muscle was composed of a small percentage of type I and IIC fibers, but the predominant type was very different histochemically and immunohistochemically from all fiber types (I, IIA, IIB, IIC) present in skeletal muscles. This esophageal fiber type (IIoes) had an acid- and alkaline-stable m-adenosine triphosphatase (ATPase) activity and a moderate histochemical Ca-Mg actomyosin ATPase activity, and it reacted weakly with anti-IIA and anti-IIB myosin sera. Although the light chains of the IIoes were the same as the light chains of a mixture of IIA and IIB myosin, their respective heavy chains gave different peptide maps. Greater differences were observed between the heavy chains of IIoes and other striated muscle myosins.4 It was concluded that this predominant fiber type in the dog and cat striated muscle is of the “fast” type, and contains a distinct isoform of myosin similar, but not identical, to the other fast-type myosins. Histochemically and immunohistochemically, the muscle of the canine esophagus was similar to that of cats.4
The histologic characteristics of the lower or gastroesophageal sphincter (LES) were compared with those of the smooth muscle of the distal esophagus in three cats. Numerous thin, annulospiral elastic fibers were found in the circular layer of the LES and the distal esophagus. These fibers were not observed in other areas.5
In dogs the esophageal glands were found over the entire length of the esophagus, whereas in cats they were found only in the cranial one-third of the esophagus.3 Scanning electron microscopy (SEM), light microscopy, and morphometric analyses were used to study the morphology of the mucosa and submucosa of the cervical, thoracic, and abdominal esophagus in the dog.6 Apart from the absence of a lamina muscularis mucosae in the cervical part of the canine esophagus, little regional variation was detected. There was, however, morphologic variation associated with age. The number and complexity of microplicae on surface epithelial cells observed by SEM increased with age, particularly between 1 and 21 days of age. Although SEM revealed typical duct openings from submucosal glands in 1-day-old dogs, light microscopy revealed few functional glands at that age. However, there was a marked increase in the volume fraction occupied by glands between 1 and 161 days of age, followed by some decrease between 161 and 337 days of age.
Ciliated cells were observed in the esophagus of 1-day-old dogs, being most abundant in the abdominal part, but by 21 days of age, groups of ciliated cells were no longer observed in any part of the esophagus.6
Vessels of the Esophagus
The arterial blood supply to the cervical part of the esophagus in dogs and cats is supplied by the thyroid arteries and esophageal branches of the carotid arteries.7,8 The esophageal portion of the bronchoesophageal artery is the main supply to the cranial two-thirds of the esophagus and the remaining part is supplied by esophageal branches of the aorta.7
Venous drainage is via the external jugular and azygos veins; adjacent veins anastomose with each other on the esophagus.7,8 Lymph vessels from the esophagus drain into the medial retropharyngeal, deep cervical, cranial mediastinal, bronchial, portal, splenic, and gastric lymph nodes.7
Nerves of the Esophagus
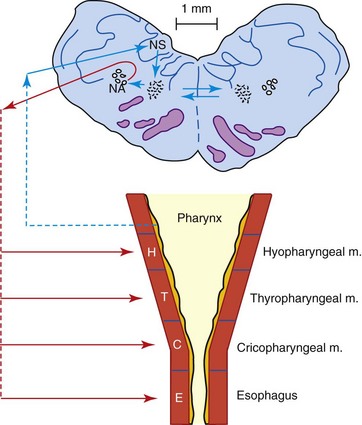
Innervation of the striated muscle of the esophagus in dogs and cats is provided by special visceral efferent neurons from the bilateral nucleus ambiguus in the medulla oblongata. Axons are carried in the vagus nerves and distributed with the pharyngoesophageal nerves, the recurrent laryngeal nerves, and the vagal trunks. In cats the smooth muscle innervation, via general visceral efferents, arises from the rostral parts of the bilateral nucleus ambiguus and is also distributed via the branches of the bilateral vagus nerves. Three major regions of innervation can be recognized in the esophagus of dogs: a cervical region, supplied by paired pararecurrent laryngeal nerves; a cranial thoracic region, supplied mainly by the left pararecurrent laryngeal nerve; and a caudal thoracic and abdominal region, supplied by the vagal trunks.7,9
Function of the Esophagus
The function of the esophagus is to transport food and liquid from the pharynx to the stomach. The esophageal stage is the last stage of the swallowing action and although linked to the pharyngeal stage, is under distinct neuronal control. Swallowing is a physiologic phenomenon that occurs many times daily. Although it may be initiated consciously as a voluntary act during eating, most swallowing occurs subconsciously between meals, without apparent cerebral participation. Swallowing irrespective of eating occurs about once a minute in conscious individuals and is driven by salivation, which stimulates the sensory receptors in the mouth and pharynx.10 Studies in humans show that both salivation and swallowing virtually cease during sleep.
Swallowing is a complex and complicated function that can be divided into four overlapping stages: oral preparatory, oral, pharyngeal, and esophageal. The first two are voluntary and the last two are involuntary. In the oral preparatory stage, food is chewed and prepared for the oral stage (see also Chapter 54). In the oral stage the food bolus is moved from the front of the oral cavity to the faucial arch, where the pharyngeal stage is initiated. The pharyngeal stage has four defined components: palatopharyngeal closure, peristalsis of the pharyngeal constrictor muscles, airway protection, and cricopharyngeal relaxation. The larynx is elevated and closed, protecting the airway. These reflex movements occur in sequence and overlap one another. They are mediated by the reticular formation in the brainstem and follow a fixed pattern. The interneuronal network involved in this pattern is termed the central pattern generator (CPG) for swallowing. Interconnections between the left and right CPGs coordinate their activity (Figure 55-1). There is coordination with the respiratory center as well.
In the final stage of swallowing, the bolus is moved through the esophagus by the peristaltic contractions of the esophageal musculature.11 The swallowing action during this stage is best illustrated by contrast videofluorography.12 After the prehension of food by the stripping action of the tongue, a bolus is formed at the base of the tongue and is passed into the oropharynx. This is immediately followed by contractions of the pharyngeal muscles, lifting the food bolus up and passing it through the relaxed cranial esophageal (cricopharyngeal) sphincter into the cervical part of the esophagus. At the same time, the pharyngeal egresses (i.e., nasopharynx, oral cavity, and laryngeal opening) are closed, preventing regurgitation and leakage into the trachea. After entry of the bolus into the esophagus, the cranial esophageal sphincter immediately closes and the bolus is transported by one slow and regular peristaltic wave from the cervical region to the gastroesophageal junction, where it passed slowly through the relaxed sphincter into the stomach.
The passage of food through the cricopharyngeal sphincter is usually followed immediately by esophageal peristalsis. However, in some dogs, especially those that bolt their food, not every swallowed bolus is picked up immediately by esophageal peristalsis; sometimes, peristaltic activity starts only after a second or even a third bolus entered the esophagus. Another frequent phenomenon is the stopping of a bolus of food somewhere in the esophagus until it is carried along with the next bolus. Similarly, a bolus sometimes stops at the gastroesophageal sphincter and does not enter the stomach until it is carried in with the next bolus. Reflux of food from the stomach into the terminal portion of the esophagus occurs occasionally, but it is always followed by secondary peristalsis that clears the esophagus. The pharynx and esophagus are always completely cleared of food at the end of the meal.12
Neurophysiologic Control of the Esophageal Stage of Swallowing
Esophageal peristalsis initiated by the pharyngoesophageal action is termed primary peristalsis. Secondary peristalsis occurs when a bolus remains in the esophagus. It is initiated by receptive fields in the esophagus which are stimulated by the persistent bolus.13
The motor sequence of the esophageal stage depends, as does the pharyngeal stage, on the activity of medullary interneurons in the swallowing center. Through excitatory and inhibitory connections, these interneurons program the sequential excitation of motor neurons and vagal preganglionic neurons responsible for the entire motor sequence, in both striated and smooth muscles of the esophagus. The activity of the medullary swallowing neurons occurs without feedback phenomena, and is thus entirely dependent on a central network.14
Components of the Sensory Pathway in the Esophageal Stage of Swallowing
The esophageal stage of swallowing requires sensory feedback to control and modulate its function. The importance of sensory feedback is suggested by the finding that the peristaltic waves were slower, greater in amplitude, and longer in duration during swallowing of water than during dry swallowing.14,15 The influence of stimulation of the afferent fibers of the esophagus was shown to increase the discharge of the vagal motor fibers supplying the esophagus.15–17 The afferent fibers of the esophagus are in the glossopharyngeal and vagus nerves. The cell bodies of these cranial nerves lie in their associated sensory ganglia. Branches of these sensory nerves pass rostrally to innervate the nerve cells in the nucleus of the solitary tract in the medulla oblongata. The neurons of this nucleus connect with interneurons in the dorsal area around the nucleus tractus solitarius. This dorsal region contains the first synaptic sites for the sensory input that evokes swallowing and the interneurons that are excited switch on the specific timing during pharyngeal and esophageal swallowing. Once triggered, bursts of sequential activity in interneurons in the dorsal region may function without feedback. They are driven by central neurons, the “master” interneurons, which set up the sequential activation of specific motor neurons. These master neurons control in particular the pharyngeal swallowing pattern. They are linked to interneurons in the ventral region around the nucleus ambiguus, which control the esophageal stage of swallowing and activate the motor neurons in the nucleus ambiguus. The interneurons in the ventral region connect with the contralateral region involved in swallowing and thus unilateral activation of swallowing may activate nucleus ambiguus motor neurons bilaterally. It is not clear how the dorsal and ventral regions connect with the motor neurons of the dorsal motor nucleus of the vagus.13
Components of the Motor Pathway in the Esophageal Stage of Swallowing
The motor neurons for the esophageal stage of swallowing are in the left and right nucleus ambiguus in the brainstem. The motor neurons innervating both the striated and smooth muscles of the esophagus are located in the rostral part of the nucleus ambiguus. The dorsal motor nucleus of the vagus nerve, which would be expected to harbor preganglionic neurons innervating the smooth muscles of the distal esophagus, are apparently not of major importance to the swallowing action of the esophagus in the cat. The axons of the motor neurons in the nucleus ambiguus associated with esophageal swallowing proceed through the vagus nerve and directly innervate esophageal striated muscles, or they pass to ganglia near the esophagus from which postganglionic excitatory cells then innervate the smooth muscles.13 As mentioned previously, the timing of esophageal swallowing is controlled by the CPG, a group of interneurons in the reticular formation and around the nucleus ambiguus, under influence of the sensory input that elicits swallowing.13
Peripheral Neural Control of Esophageal Peristalsis
Esophageal peristalsis is normally a smooth, uninterrupted contraction wave that traverses the esophagus. It consists of a rapid wave of relaxation followed by a slower wave of contraction. A rapidly descending wave of inhibition sequentially relaxes the upper esophageal sphincter, the body of the esophagus, and the LES, so that they are prepared for passage of an oncoming bolus. In recumbent subjects, transport of the bolus is exclusively the result of pharyngoesophageal peristalsis, while in upright subjects it is assisted by gravity.10,18
Neural control of esophageal peristalsis differs between dogs and cats in that the end organ in dogs is striated muscle and in cats it is both striated and smooth muscle. The striated muscle of the canine esophagus is a relatively slow-contracting muscle, as shown by direct electrical stimulation in vitro.13,19 Electrical stimulation of the vagus nerve to the esophageal striated muscle caused contraction of the muscles,13,20 and curare, which antagonizes the neurotransmitter acetylcholine, blocks the vagal excitation of the canine striated esophageal muscle. Curare blocks the peristaltic contractions of the entire esophagus in dogs.13,21 Pharmacologic studies show that the striated esophageal muscle is under central neural control via motor fibers that release acetylcholine to stimulate nicotine receptors, the same receptors as in somatic muscles. In the same comparison with somatic muscles, inhibition of the striated esophageal muscles depends on a decrease in discharges from the vagal efferent fibers. No separate inhibitory mechanism for striated esophageal muscles has been found.13 Esophageal peristalsis in the dog is produced by a descending sequence of efferent neural discharges, generated by the central swallowing program.
In the cat, the esophageal striated muscles are innervated and function in the same way as in the dog. In the smooth muscles of the distal part of the esophagus in the cat, peristalsis can develop with or without central efferent signals via the vagus nerve.13,22 As peristalsis reaches the distal part of the striated muscle segment, the central swallowing program stimulates the onset of peristalsis in the proximal part of the smooth muscle segment. Esophageal peristalsis is most likely initiated by neural excitation. This originates centrally in primary peristalsis and in cats it also does so in secondary peristalsis.10 The organization of esophageal muscle peristalsis was shown to persist after experimental transection of the vagus, and the smooth muscle segment of the esophagus can also function without central control.
Acetylcholine is an important transmitter in the ganglia or at the neuromuscular junction of the esophageal smooth muscle.13,23 It is postulated that the timing of sequential activation could depend on the amount and ratio of neural transmitters at each segmental level, the timing of their release, and the density of smooth muscle receptors.13,24 It is probable that peristalsis in the smooth muscle segment of the esophagus in the cat functions via a neuronal network through the ganglia, with or without the smooth muscle providing control for a sequential contraction, and thus with or without sequential activity descending in the vagal efferent fibers.
Peripheral Neural Control of the Gastroesophageal Sphincter Function
During swallowing, the neural control of the LES in the dog and cat is similar to the neural control of peristalsis in the distal third of the esophagus. In dogs, relaxation and contraction of the LES follows the activity of the distal esophagus and is under control of the central pattern generator for primary peristalsis in the distal esophagus. Vagal nerve fibers directly innervate the striated muscle cells, their neurons being located in the nucleus ambiguus. In the cat, neural control of the sphincter is similar to control of peristalsis in the distal third of the esophagus and is under control of the central pattern generator. The smooth muscle of the sphincter is directly innervated by postganglionic cells and in part by mechanisms intrinsic to the esophagus itself.10
Other Influences on Gastroesophageal Sphincter Function
Also influencing LES function are the receptor responses to intrinsic or extrinsic pharmacologic agents. Most studies of these agents have been concerned with better understanding of the causes of reflux esophagitis in humans, but several of the studies have contributed to our understanding of the function of the LES in dogs and cats. In conscious dogs the LES response to a meal consists of tonic contractions of the sphincter. These appear to be mainly regulated by muscarinic receptors. Nitric oxide can inhibit postprandial contractions of the LES.25
In dogs, gastric distention induces transient relaxation of the LES. The basal LES pressure fell progressively during distention of the antrum but not during distention of the fundus alone. The antrum was considered to be primarily responsible for triggering transient relaxation of the LES induced by distention.26 Radiofrequency energy applied to the LES in dogs inhibited the triggering of transient relaxations and this reduced the resulting gastroesophageal reflux. Basal LES pressure and LES relaxation during swallowing were unchanged.27 In cats, swallowing and balloon distention of the esophagus both caused relaxation of the LES.28 Microinjections of bicuculline (γ-aminobutyric acid [GABA] antagonist) in the dorsal motor nucleus of the vagus in the medulla oblongata in cats caused a decrease in the LES pressure. This effect was abolished by vagotomy. These results indicate that the dorsal motor nucleus of the vagus influences the functional regulation of the LES in cats.29 It is to be expected that more detailed information about the function of the LES in relation to gastric motility will become available, since better understanding of the dysfunction of the LES in reflux disease is of high priority in human gastroenterology.
Diagnostic Evaluation
History and Signalment
Regurgitation
Regurgitation is the hallmark of esophageal disease and is defined as the passive evacuation of food or fluid from the esophagus into the environment. Regurgitation results primarily from local mechanical events within the esophagus and must be distinguished from vomiting, which is a centrally mediated reflex characterized by the active evacuation of gastroduodenal contents preceded by nausea, hypersalivation, retching, and abdominal contractions. The history should determine the age of onset of regurgitation, the duration of signs, the timing of regurgitation in relation to eating, and a description of the regurgitated material. Videorecordings of eating and drinking behavior are often very informative and may help to differentiate esophageal and upper airway signs. Chapter 21 has additional discussion of regurgitation and a diagnostic algorithm.
Breed predispositions for esophageal disease should be considered, especially in young puppies and kittens with persistent regurgitation of undigested food that begins shortly after weaning, which is suggestive of vascular ring anomaly, congenital idiopathic megaesophagus, or congenital esophageal stenosis.1 The congenital form of megaesophagus is heritable in Wire-haired Fox Terriers and Miniature Schnauzers and an increased prevalence has been noted in Irish Setters, Great Danes, German Shepherds, Labrador Retrievers, Newfoundlands, Chinese Shar-Peis, and Siamese cats.2–4 In addition, acquired megaesophagus and oropharyngeal dysphagia may be associated with several types of myopathy (e.g., myasthenia gravis, muscular dystrophies, and polymyositis) that have increased incidence in many breeds of dogs and cats.5,6 Vascular ring anomalies have been reported in many breeds of dogs and cats, but German Shepherds, Irish Setters, and Labrador Retrievers appear to be at increased risk.7,8 Late-onset regurgitation can occasionally be seen with mildly obstructive vascular ring anomalies that have gone undetected until as old as 8 years of age.9 Young Chinese Shar-Pei dogs are predisposed to congenital hiatal hernia resulting from malformation of the esophageal hiatus.10,11 Bulldogs and other brachycephalic breeds with upper airway obstruction have a high prevalence of concurrent hiatal abnormalities, reflux esophagitis, esophageal deviation, gastroduodenal disease.12
The regurgitated material is usually composed of undigested food (often tubular in shape), water, and white to clear frothy liquid (mucus and saliva). In comparison, vomitus usually consists of partially digested food mixed with yellow bile-stained fluid. Dogs and cats with megaesophagus usually regurgitate both solids and liquids. Tolerance of liquids but not solid foods is more typical of obstructive esophageal diseases.1 Fresh red blood is occasionally seen with severe mucosal trauma or erosions, especially from foreign bodies. Putrefaction of ingesta may occur after prolonged retention in a diverticulum or dilated esophagus, which can cause halitosis. Both regurgitation and vomiting can occur concurrently in animals with dysautonomia, hiatal hernia, gastroesophageal intussusception, or vomiting disorders complicated by secondary reflux esophagitis.
Dysphagia, Odynophagia, and Salivation
Dysphagia, or difficult swallowing, more often connotes oropharyngeal disease and cricopharyngeal dysfunction, but may be seen with cranial esophageal diseases such as foreign body, stricture, or esophagitis. Dysphagia is characterized by repeated, often unproductive attempts to swallow with extension of the head and neck during swallowing, and is often accompanied by gagging, retching, odynophagia (pain on swallowing), and ptyalism (excessive salivation). Anorexia and unexplained salivation may be the only clinical signs in some patients with painful esophageal disease, especially esophagitis. Esophageal dysphagia and salivation must be differentiated from oropharyngeal dysphagia and salivary gland diseases, such as sialadenitis and sialadenosis. Chapters 13 and 22 provide additional discussions of dysphagia and salivation, respectively, and diagnostic algorithms.
Historical Findings Indicative of Esophageal Injury
The history is important for identifying potential causes of esophageal injury that could culminate in severe esophagitis or esophageal stricture. These include esophageal foreign bodies, gastroesophageal reflux (secondary to general anesthesia, hiatal hernia, or malpositioned esophageal feeding tubes), acid or peptic injury from persistent vomiting, irritating oral medications (e.g., doxycycline, clindamycin), ingestion of corrosive chemicals, radiation injury, thermal injury from overheated (microwaved) food, and prior esophageal surgery.1,13–17 Clinical signs of esophagitis usually develop within 1 to 3 days following injury and include regurgitation, dysphagia, salivation, odynophagia, retching, gagging, and repeated swallowing.1,17 In mild esophagitis, clinical signs may be subtle or absent. Severe esophagitis or foreign-body impaction can lead to serious complications of esophageal necrosis, perforation, fibrosis, and stricture. The possibility of esophageal perforation may be suggested by a recent history of foreign body ingestion. Iatrogenic perforation can also occur after endoscopic removal of foreign bodies, dilation of esophageal strictures, laser ablation of tumors, and esophageal surgery. Penetrating injuries of the cervical esophagus can be caused by bite wounds or gunshot wounds.
Esophageal strictures result from deep circumferential injury of the esophagus that heals by intramural fibrosis, resulting in narrowing of the lumen and reduced distensibility.1,14,17 Regurgitation (solids more than liquids) and other clinical signs attributable to esophageal obstruction usually begin within 1 to 4 weeks after the inciting esophageal injury.14,18–22 Despite the severity of the inflammation, many animals will have preservation of appetite because of the caloric deprivation and worsening malnutrition.
Owners should be questioned about the likelihood of ingested foreign objects. Esophageal foreign bodies are common, especially in small dogs (weighing <10 kg), and include objects such as bones, chew toys, plastic, rubber, pins, needles, fishhooks, string, and trichobezoars. Trichobezoars are particularly important in cats.23–29 In one report, 12% of dogs with moderate to severe foreign-body–induced esophagitis subsequently developed strictures.25 Esophageal foreign-body obstruction caused by dental chew treats resulted in strictures in six of 25 dogs (24%) in another report.24 Esophageal perforation and esophagobronchial fistula are also potential complications of foreign-body trauma.
Overall, 65% of esophageal strictures in dogs and cats occur following general anesthesia.18–22,30,31 This is presumably a result of the reflux of gastric (H+, pepsin) and/or intestinal (bicarbonate, bile salts, and pancreatic proteases) content associated with relaxation of the gastroesophageal sphincter (GES), and the delayed clearance of the refluxate from the esophagus in the anesthetized animal.31,32 Clinical signs of esophagitis usually develop within 2 to 4 days of the anesthetic procedure, followed by progressive worsening of regurgitation if fibrosis takes place.
Hiatal hernia is a protrusion of a portion of the stomach through the esophageal hiatus of the diaphragm, which can be the result of congenital or acquired laxity in the hiatal opening.1,13,33,34 Clinical signs in the acquired form are often intermittent and attributable to secondary reflux esophagitis caused by displacement of the GES into the thorax. Clinical signs in the congenital form are often more frequent and more severe due to extreme laxity at the esophageal hiatus. Reflux esophagitis can occur independently of hiatal hernia, is idiopathic in some dogs and cats, and presumably results from GES incompetence or dysmotility.
Corrosive injury to the esophagus can result from several oral medications and ingested chemicals. The severity of the injury depends on chemical concentration, pH, duration of contact with the mucosa, and distribution of the lesion (e.g., focal vs. diffuse injury). Oral tablet and capsule medications with high acidity, especially doxycycline and clindamycin in cats, can cause severe focal esophagitis and strictures.35–39 The corrosive effects of these medications are worse in cats because of the slower esophageal propagation velocity in this species (compared with the dog), and prolonged mucosal contact time when tablets or capsules are administered as a “dry swallow.”40,41 Ingested chemical irritants are usually concentrates that have highly acidic, alkaline, or oxidant properties. This should be suspected when esophagitis is accompanied by stomatitis and oropharyngeal ulcerations (mucositis). Examples include chlorine granules, button batteries, and undiluted quaternary ammonium disinfectants such as benzalkonium chloride that cats can lick from their paws and haircoat.42,43
Other Clinical Signs
Other clinical signs can be seen in dogs and cats with esophageal disease. Weight loss and poor body condition can occur secondary to persistent regurgitation and inability to retain ingested food. An otherwise healthy animal with persistent regurgitation may have a ravenous appetite. This is common in animals with megaesophagus, vascular ring anomaly, and some esophageal strictures. In contrast, anorexia can occur in animals that have painful and difficult swallowing associated with severe esophagitis, stricture, esophageal foreign body, or neoplasia. Anorexia can also occur in conjunction with cough, tachypnea, dyspnea, and fever in animals that have secondary aspiration pneumonia, esophageal perforation, mediastinitis, or bronchoesophageal fistula. The owner should also be questioned about signs of neurologic or neuromuscular dysfunction that could indicate an underlying cause of acquired megaesophagus.1,2,4
Laryngeal paralysis (stridor and change or loss of voice) can be associated with acquired megaesophagus in dogs.4 Disorders of the esophageal hiatus (e.g., hiatal hernia, reflux esophagitis) can be associated with chronic obstructive airway conditions such as laryngeal paralysis and brachycephalic syndrome, presumably from enlargement of the esophageal hiatus and laxity of the surrounding support structures caused by abnormally high negative intrathoracic pressure.12 Increased intraabdominal pressure associated with vomiting or blunt abdominal trauma can also cause hiatal hernia. Severe gastroesophageal reflux can cause signs of chronic laryngitis (stridor, change of bark) from exposure of the larynx to gastric refluxate. Mediastinal tumors (lymphoma, thymoma) that cause regurgitation from periesophageal compression of the esophagus can also cause dyspnea (pleural effusion) and Horner syndrome.
Physical Examination
Depression, fever, cough, tachypnea, dyspnea, mucopurulent nasal discharge, and abnormal pulmonary auscultation can indicate complicating aspiration pneumonia, or rarely, bronchoesophageal fistula. In animals with esophageal foreign body, a rigid stance with fever and depression can indicate mediastinitis secondary to esophageal perforation. Pleural effusion, Horner syndrome, and a noncompressible cranial thorax can be found in animals with periesophageal cranial mediastinal tumors (e.g., lymphoma or thymoma). Neurologic deficits or muscle weakness, atrophy, or pain may indicate an underlying neuromuscular disorder as a cause of esophageal hypomotility and secondary megaesophagus. A cranial nerve examination is also important in animals with abnormal oropharyngoesophageal motility. Dysautonomia, which is a generalized autonomic neuropathy that can cause megaesophagus, is suggested by mydriasis and loss of pupillary reflexes, decreased tear production, dry mucous membranes, bradycardia, dysuria, decreased anal tone, and diarrhea or constipation.1,2
Laboratory Evaluations
In dogs and cats with megaesophagus and motility dysfunction, additional laboratory evaluations should be considered to identify an underlying cause.1–344 Primary megaesophagus can be either congenital or acquired. Acquired megaesophagus is often idiopathic, but this is a diagnosis of exclusion. Esophageal motility dysfunction and megaesophagus can also occur secondary to various underlying neuromuscular disorders that impair esophageal motility, including myasthenia gravis, polymyositis, muscular dystrophy, other polymyopathies, peripheral neuropathies, central nervous system disease, dysautonomia, botulism, tick paralysis, tetanus, anticholinesterase toxicity, lead toxicity, hypoadrenocorticism, and possibly hypothyroidism.2–444 Measurement of serum acetylcholine receptor antibody for the diagnosis of acquired myasthenia gravis is especially important, even in the absence of generalized muscle weakness, because focal myasthenia can account for 25% of acquired canine megaesophagus cases.5,45
Radiography
Survey and contrast radiographic evaluations are useful for the diagnosis of many esophageal diseases (see Chapter 26 for techniques). Survey thoracic and cervical radiographs are obtained initially to evaluate the entire esophagus and to identify complications of esophageal disease, such as aspiration pneumonia (alveolar lung opacity) and esophageal perforation (pneumothorax, pneumomediastinum, mediastinitis, pleural effusion). The esophagus is not normally visible unless it contains air, fluid, food, or foreign material, so survey radiographs can often identify radiopaque foreign bodies, megaesophagus, diverticula, esophageal masses, and esophageal dilation outlined by air, fluid, or ingesta cranial to an obstructive lesion.46,47 A small amount of air in the esophagus from aerophagia can be an incidental finding caused by excitement, nausea, dyspnea, or anesthesia. Hiatal hernia and gastroesophageal intussusception are usually identified as a mass effect cranial to the hiatus in the dorsocaudal mediastinum if the stomach is displaced at the time of the radiograph.
Contrast radiography of the esophagus is indicated in animals with signs of esophageal disease that have normal or inconclusive survey radiographs.46 Static image barium contrast radiography (i.e., without fluoroscopy) is useful for documenting radiolucent foreign bodies, megaesophagus, diverticula, and obstructive diseases (e.g., vascular ring anomalies, strictures, tumors, periesophageal masses). Hiatal hernia and gastroesophageal intussusception can be confirmed if the stomach is displaced at the time of the radiograph.
Potential complications of contrast esophagraphy are aspiration of contrast medium or leakage from an esophageal perforation. Perforation should be suspected when survey radiographs reveal cervical soft tissue emphysema, pneumomediastinum, pneumothorax, mediastinitis, or pleural effusion. In such cases a water-soluble, nonionic, iodinated contrast agent such as iohexol (Omnipaque; Amersham Health) is preferred over barium because it is less irritating to periesophageal tissues and more readily reabsorbed.48 For demonstrating acquired esophagobronchial fistulas, which sometimes develop after foreign-body perforation of the caudal esophagus, a dilute barium mixture (20% to 30% w/v [weight per volume]) is preferred, as iohexol is hypertonic and can cause pulmonary edema if it enters the lung.49
Continuous videofluoroscopic recording of swallowing is required to characterize complex motility disorders of the esophagus, as exact timing of exposures is unlikely with static-image barium radiographs.47,50 Contrast videofluoroscopy provides a dynamic evaluation of the oropharyngeal, esophageal (both primary and secondary peristalsis), and gastroesophageal phases of swallowing (see Chapter 26).50 Sequential three-phase contrast videofluoroscopy can be performed in the unsedated animal using barium liquid, barium paste, and barium-soaked food (kibble, canned, or both).51 Sternal positioning may be preferable to lateral positioning for videofluoroscopic evaluations of swallowing and esophageal transit.52 Videofluoroscopy can be combined with esophageal manometry,53 electromyography, and pH monitoring to provide additional information on pressure–time relationships, neuromuscular integrity, and gastroesophageal sphincter function, respectively (see “Esophageal Structure and Function” section).
Megaesophagus and Motility Disorders
Megaesophagus is characterized by severe diffuse esophageal hypomotility and flaccid dilation of the esophagus. Primary megaesophagus is usually idiopathic and can be either congenital or acquired.2,3 Survey radiographs are usually diagnostic of a severely enlarged esophagus distended with air, fluid, or food.2 If survey radiographs are inconclusive, a static-image barium contrast esophagram can be used to confirm esophageal dilation and reduced esophageal clearance. Contrast videofluoroscopy reveals aperistalsis and impaired esophageal transport of contrast medium in the absence of obstruction. Less severe esophageal motility dysfunction without overt megaesophagus usually requires barium swallow videofluoroscopy to assess the intensity and coordination of peristalsis and the coordinated functioning of the sphincters.47,54 Diffuse or segmental esophageal hypomotility can also occur secondary to esophagitis, hiatal hernia, and obstructive esophageal lesions, such as strictures, vascular ring anomalies, and leiomyomas.4,10,44
Esophageal Diverticulum
Esophageal diverticula are large pouch-like sacculations of the esophageal wall that interfere with the orderly movement of ingesta through the esophagus. Diverticula can be identified by radiography or endoscopy.55–57 Survey thoracic radiographs show an air-, fluid-, or food-filled esophageal sacculation or pouch, and contrast radiography demonstrates pooling of barium in the diverticulum. Most diverticula occur in the cranial mediastinal and epiphrenic regions of the esophagus.55–57 Congenital diverticula are rare abnormalities of embryogenesis that permit herniation of the esophageal mucosa through a defect in the muscularis. Acquired diverticula can result from external traction and distortion of the esophagus caused by periesophageal inflammatory adhesions (traction diverticula), or most often from increased intraluminal pressure and food impaction (pulsion diverticula) associated with esophageal injury (esophagitis), hypomotility (megaesophagus), or obstruction (e.g., vascular ring anomaly, foreign body, stricture, and tumor). Radiographs should be evaluated carefully for underlying causes when an acquired diverticulum is identified. A redundant flexure or deviation of the esophagus at the thoracic inlet is a common incidental finding in clinically normal brachycephalic and Chinese Shar-Pei dogs and should not be mistaken for a diverticulum.58 These false diverticula lack pooling of food and fluid and decrease or disappear with extension of the neck.
Vascular Ring Anomaly
Vascular ring anomalies are congenital malformations of the great vessels and their branches that entrap the intrathoracic esophagus and other intrathoracic structures, and cause clinical signs of esophageal obstruction.1,7,59–62 Persistent right aortic arch accounts for 95% of vascular ring anomalies. In persistent right aortic arch, the ligamentum arteriosum continues to develop from the left side and forms a fibrous band that crosses over the esophagus to connect the main pulmonary artery and the anomalous aorta. The esophagus becomes entrapped and constricted circumferentially by the ligamentum, aorta, and base of the heart. Survey thoracic radiographs usually show a severe dilation of the esophagus with food and fluid cranial to the heart. A characteristic leftward deviation of the trachea near the cranial border of the heart is a consistent finding on ventrodorsal or dorsoventral views.7 The normal shadow of the aortic arch to the left may be absent. Barium contrast esophagram will confirm obstruction of the esophagus at or just cranial to the base of the heart. Nonselective computed tomography (CT) angiography can be used to diagnose rare atypical vascular ring anomalies.7,62
Esophageal Foreign Body
Esophageal foreign bodies are common and lodge most often at the thoracic inlet, base of the heart, or gastroesophageal sphincter,23–29 which are the least-distensible regions of the esophagus. Survey thoracic and cervical radiographs are usually diagnostic for radiopaque foreign bodies, such as bones or metal objects (pins, needles, fishhooks, etc.). Radiolucent objects can be identified if outlined by air, or they may require barium contrast radiography or esophagoscopy. Foreign bodies may injure the esophageal mucosa and cause complications such as esophagitis, perforation, or stricture. If perforation is suspected, iohexol should be used as a contrast agent instead of barium.
Hiatal Disorders
Hiatal hernia is a congenital or acquired protrusion of a portion of the stomach through the esophageal hiatus of the diaphragm.1,10,11,13,33,34 In many cases the herniation and clinical signs are intermittent. A sliding hiatal hernia, which is most common, is a cranial displacement of the abdominal segment of the esophagus, gastroesophageal junction, and cardia region of the stomach through the esophageal hiatus of the diaphragm into the thorax. A paraesophageal hiatal hernia occurs when a portion of the stomach (usually the fundus) herniates through the hiatus into the caudal mediastinum alongside the caudal thoracic esophagus.63
Gastroesophageal intussusception is an invagination of the stomach into the lumen of the caudal esophagus.1,13,64–67 Gastroesophageal intussusception can be diagnosed by radiography or esophagoscopy. If needed to confirm the diagnosis, barium esophagram will identify gastric rugae within the lumen of the esophagus and severe esophageal obstruction. Like hiatal hernia, it can be a consequence of laxity of the esophageal hiatus and may occur intermittently. It is most often a complication of esophageal hypomotility or megaesophagus.64,66,67
Esophageal and Periesophageal Neoplasia
Primary esophageal tumors are rare.1,13 Squamous cell carcinoma is the most common primary malignant tumor in cats.68,69 In regions where S. lupi is an endemic parasite, dogs can develop fibrosarcomas and osteosarcomas from spirocercal granulomas.70,71 Leiomyomas, which are the most common benign tumors of the esophagus, most often arise near the gastroesophageal junction of dogs and usually are incidental findings unless large enough to partially obstruct the lumen.72 Leiomyosarcoma, lymphoma, and metastatic carcinoma are rare tumors of the esophagus. Survey thoracic radiography may be normal or may identify a soft-tissue mass in the region of the esophagus. The degree of luminal obstruction depends on the size and position of the mass, and the esophagus may be distended cranial to the tumor site. Barium esophagram may reveal stricture with irregular intraluminal filling defects, but endoscopic biopsy will be required for definitive diagnosis. Survey radiographs in dogs with S. lupi-induced sarcoma may reveal a dorsocaudal mediastinal mass, spondylitis of the caudal thoracic vertebrae, and hypertrophic osteopathy.70,71
Esophagoscopy
Esophagoscopy is indicated for the diagnostic evaluation of many dogs and cats with signs of esophageal disease, especially when radiography is inconclusive. Esophagoscopy enables thorough visual examination for diseases that disrupt the mucosa or obstruct the lumen, for example, foreign bodies, esophagitis, strictures, tumors, and gastroesophageal intussusception.1,13,73 Esophagoscopy is less reliable for diagnosing megaesophagus and other functional motility disorders. Some diseases of the esophagus, stomach, and intestine cause overlapping signs or involve multiple regions of the digestive tract simultaneously; thus, most animals that undergo esophagoscopy should have a complete endoscopic examination of the entire upper gastrointestinal tract (esophagogastroduodenoscopy). It can be difficult to obtain good quality endoscopic biopsies of normal esophageal mucosa, but adequate biopsy specimens can usually be obtained from diseased mucosa with increased friability (esophagitis) and from neoplastic tissue. Endoscopic cytology specimens collected using a guarded (sheathed) cytology brush are sometimes useful for diagnosing esophageal neoplasia, unusual forms of esophagitis caused by infectious agents (pythiosis, candidiasis, and spirocercosis), and eosinophilic esophagitis. Esophagoscopy can also be used for therapeutic intervention to remove esophageal foreign bodies, guide balloon catheter placement with esophageal strictures, assist in the deployment of esophageal stents, place indwelling gastrostomy and esophagostomy feeding tubes, and ablate neoplastic tissue with lasers.13,73 Chapter 27 describes the equipment and techniques for performing esophagoscopy.
Megaesophagus
Esophagoscopy is less reliable than radiography for confirming megaesophagus, but it can help rule out obstructive causes of esophageal dilation (GES abnormalities, vascular ring anomalies, strictures, tumors) and evaluate for the esophagitis that develops in many cases.2,4 Esophageal motility and lumen size are difficult to assess with endoscopy because the normal esophagus becomes flaccid and dilated with general anesthesia and insufflation of air. The typical endoscopic appearance of megaesophagus is a markedly dilated, flaccid esophagus throughout its entire length, with variable amounts of saliva, fluid, and food residue in the lumen.73 On initial insertion of the endoscope the normal esophagus in a fasted animal is usually empty, so a motility, hiatal, or obstructive disorder should be suspected when the esophagus contains a pool of fluid and ingesta. Megaesophagus must be differentiated from segmental dilations or sacculations that develop cranial to obstructions caused by vascular ring anomalies, strictures, tumors, or periesophageal masses. A diverticulum of the cranial thoracic esophagus sometimes develops as a complication of chronic megaesophagus.
Esophageal Diverticulum
In esophageal diverticulum, esophagoscopy reveals a saclike outpouching of the esophageal lumen, often with erosive esophagitis of the mucosa lining the diverticulum. Food, fluid, or hair may have to be removed from the diverticulum before it can be adequately visualized.57 If a diverticulum is small, the only obvious finding may be a focal pooling of fluid. Because of the thin, weakened wall of the diverticular sac, caution must be used to avoid perforation.
Vascular Ring Anomaly
Esophagoscopy is useful for diagnosing vascular ring anomaly when radiography is inconclusive or unable to distinguish it from other causes of esophageal obstruction, such as stricture. Vascular ring anomaly causes entrapment and extraluminal compression of the esophagus, yielding a characteristic endoscopic appearance. Pulsations of the major vessels against the wall of the esophagus are seen at the level of the narrowed lumen, and the outline of the band-like ligamentum forms a distinct indentation that crosses over the left dorsolateral wall of the esophagus.13,73 In some cases the obstructed lumen is only a tiny slit-like opening. The esophageal entrapment at the level of the heart base causes the cranial thoracic esophagus to be dilated, and often distended with ingesta and fluid at the time of endoscopy. In some cases vascular ring anomaly leads to sacculation of the cranial thoracic esophagus and secondary diverticulum formation.
Esophageal Foreign Body
Esophagoscopy is the definitive diagnostic procedure for esophageal foreign body. Most esophageal foreign bodies lodge at the thoracic inlet, base of the heart, or gastroesophageal sphincter, which are the least distensible regions of the esophagus. Depending on the type of object, its size and shape, and the length of time it is in contact with the mucosa, foreign bodies can injure the esophageal mucosa during passage or through impaction and pressure necrosis when they become entrapped.23–29 Foreign bodies with sharp edges and points, especially bones, can become deeply embedded and lacerate or even perforate the esophagus, and with time tightly wedged objects can cause circumferential pressure necrosis.23–25 In cats, large trichobezoars expelled from the stomach during vomiting may become lodged in the esophagus causing severe erosive esophagitis and stricture formation, presumably through the effects of pressure and prolonged mucosal contact with gastric acid and pepsin absorbed into the impacted hairball.73 Chapter 27 describes techniques for the endoscopic retrieval of foreign bodies. After an esophageal foreign body has been removed, the mucosa should be examined for esophagitis, perforation, and esophagobronchial fistula.
Esophagitis
Esophagitis is an endoscopic diagnosis based on mucosal abnormalities that can include hyperemia, increased friability, ease of bleeding when rubbed by the endoscope tip, granular surface texture, accentuated folding, erosions, ulcers, focal necrosis, pseudomembranes, pale white areas of fibrosis with indistensibility, strictures, and GES abnormalities.13–1773 Inflammatory lesions in the caudal thoracic esophagus are indicative of reflux esophagitis, especially erythematous or erosive streaks radiating from the GES accompanied by a wide open sphincter and pooling of gastrointestinal contents (food, fluid, or bile) in the distal esophagus.73 Reflux of gastric contents may be observed during endoscopy. In reflux esophagitis microscopic inflammation and epithelial changes may be evident before abnormalities can be seen with an endoscope, so if chronic reflux is suspected, mucosal biopsies should be taken adjacent to the GES for evaluation of hyperplastic, dysplastic, and metaplastic epithelial changes.15,16,74
Eosinophilic esophagitis is rare form of esophagitis diagnosed by endoscopic cytology and biopsy.75 P. insidiosum is a waterborne fungus that can cause severe necrotizing pyogranulomatous esophagitis.76 A presumptive diagnosis of pythiosis is based on finding characteristic broad, poorly septate hyphae with the Grocott methenamine silver stain in esophagoscopic biopsies. The diagnosis is supported by a positive serologic test for antibodies or by culture with polymerase chain reaction identification. Candida albicans causes white mucosal deposits of yeast, which develop secondary to retention of decomposing food residue in megaesophagus, diverticulum, or vascular ring anomaly. S. lupi is a nematode parasite that causes large granulomatous nodules in the caudal thoracic esophagus, which are transformed into fibrosarcoma or osteosarcoma.70
Esophageal Stricture
Esophagoscopy is the most reliable method for diagnosis of esophageal stricture and for determining the luminal diameter, stricture length, and presence of associated esophagitis. Diagnostic esophagoscopy is usually followed by endoscopic-guided mechanical dilation of the stricture using balloon catheter dilation or bougienage techniques. These procedures result in a favorable outcome in 70% to 88% of stricture patients.18–22 Approximately 80% to 90% of strictures in dogs and cats are located in the intrathoracic esophagus, particularly between the heart base and gastroesophageal junction.18–22 The degree of narrowing varies, but the luminal diameter averages 5 mm.18,20,22 The stricture length averages 1 cm, but it can vary from a thin band, web, or ridge of fibrotic tissue to a segmental narrowing up to 10 to 15 cm in length.18,20,31 In some cases diffuse esophagitis results in multiple strictures. Single strictures are found in 80% of cases and two or three strictures in the remaining 20%.18–22
Most strictures appear as focal circumferential narrowings formed by smooth, glistening-white, fibrotic rings or ridges.18–22 Occasionally strictures form a web or imperforate membrane across the lumen. Concurrent esophagitis in 40% of cases causes the adjacent mucosa to be erythematous, hemorrhagic, friable, or ulcerated.18–22 With chronicity, contraction of deep fibrotic tissue can distort the tubular axis of the esophagus so it appears to be angular or spiral shaped rather than straight. The degree of dilation of the esophagus cranial to the stricture depends on the duration and extent of obstruction. Benign strictures must be differentiated from other causes of esophageal obstruction, such as neoplasia or extraluminal compression caused by a vascular ring anomaly or periesophageal mass. Mucosal biopsy is recommended to determine if an atypical appearing stricture is benign or malignant.
Esophageal Perforation and Fistula
Esophageal perforation is a rare complication of esophageal foreign bodies, especially objects with irregular or sharp edges such as bones, or chronically lodged foreign bodies that cause deep-pressure necrosis.23,25,27–29 Iatrogenic esophageal perforation can also occur during endoscopic foreign-body extraction or stricture dilation, and as a complication of esophageal surgery or laser treatment. Esophageal perforation occurs in 4% to 9% of dogs and cats undergoing stricture ballooning or bougienage.18–22 Penetrating injuries of the esophagus can result from bite wounds, gunshot injuries, and stick impalements. In general perforation of the intrathoracic esophagus has more serious consequences than perforation of the cervical esophagus because of the potential for tension pneumothorax and leakage of contaminated fluids into the thorax. During esophagoscopy, hemorrhagic fluid that bubbles from a deep defect in the thoracic esophagus in synchrony with respirations is indicative of acute perforation. If perforation occurs during endoscopic manipulation of a foreign body or stricture, life-threatening tension pneumothorax may occur that is accentuated by insufflation. This situation requires immediate thoracocentesis.
An esophageal fistula is a communication between the esophageal lumen and adjacent structures (e.g., mediastinum, trachea, bronchi). A fistula appears endoscopically as a small erythematous opening in the esophageal wall that drains purulent, bloody, or frothy fluid.13,49 Esophagobronchial fistulas are the most common and often are accompanied by an esophageal diverticulum. Chronic perforations can also form fistulas that extend deep into the mediastinum.
Hiatal Disorders
Endoscopic findings consistent with hiatal hernia include pooling of fluid or ingesta in the caudal esophagus; enlargement of the esophageal hiatal opening; dilation and cranial displacement of the gastroesophageal sphincter into the thorax; rugal folds of the stomach protruding through the hiatus into the caudal thorax as viewed from the esophageal and gastric retroflex positions; and evidence of esophagitis in the caudal esophagus.73 A pseudopouch can be seen if the stomach is herniated at the time of insertion of the endoscope.73 This effect is created by narrowing of the lumen as the scope advances through the cranially displaced gastroesophageal junction followed by a dilated region of the lumen lined by rugal folds.
Gastroesophageal intussusception is diagnosed endoscopically when the rugal folds of the invaginated stomach form a bulging intraluminal mass that fills the caudal thoracic esophagus.1,13,64–67 Concurrent findings can include esophagitis, esophageal obstruction, and megaesophagus. If intussusception is encountered, the endoscopist should maximally insufflate the esophagus and occlude it in the cervical region to retain the air. The combination of inflation pressure and advancement of the endoscope tip against the invaginated rugae may reduce small intussusceptions and allow entry into the repositioned stomach.
Esophageal and Periesophageal Neoplasia
Endoscopy and biopsy are indicated for the definitive diagnosis of esophageal neoplasia.68,69,73 The intrathoracic esophagus is most often involved and the degree of luminal obstruction varies. Endoscopically, a carcinoma usually causes eccentric stenosis and appears as a proliferative mass with a friable, lobulated, or ulcerated surface. In dogs with S. lupi–associated esophageal granuloma or sarcoma, nematodes may be seen protruding from the nodular lesions and eggs may be identified by fecal examination.70 Forceps biopsies should be obtained from all esophageal masses and proliferative lesions identified at endoscopy.
Endoscopy is useful for evaluating the extent of obstruction and determining whether masses are extramural or intramural. A stenotic region of esophagus with normal-appearing mucosa suggests extraluminal compression by a periesophageal mass rather than stricture or primary esophageal tumor.73 Transmural endoscopic-guided fine-needle aspiration of periesophageal masses can be obtained for cytologic examination using a 21- to 23-gauge through-the-scope sclerotherapy needle.
Other Diagnostic Methods
Videofluoroscopy is the most clinically useful tool for evaluating all phases of swallowing in dogs and cats with esophageal motility dysfunction (see “Radiography” earlier in this chapter).46,47 Other specialized procedures include nuclear scintigraphy, manometry, pH monitoring, and electromyography, but these studies are difficult and not routinely performed in dogs and cats.
Nuclear Scintigraphy
Scintigraphic evaluation of swallowing of a radioisotope such as technetium-99 m (99mTc) sulfur colloid or 99mTc-pertechnetate enables noninvasive quantitative assessment of esophageal transit time and clearance.77 Nuclear scintigraphy can also be used to identify gastroesophageal reflux after intragastric instillation of the radioisotope via stomach tube. Scintigraphy can identify esophageal perforations and fistulas while avoiding the risks of extravasated contrast medium.
Esophageal Manometry
Continuous pull-through infusion manometry has been used extensively in people and research animals to record the pressure–time relationships during the cricopharyngeal, esophageal, and gastroesophageal phases of swallowing. Manometry techniques and normal values for dogs and cats have been described along with the effects of various drugs and neuroendocrine substances on gastroesophageal sphincter pressure.78–84 Unfortunately, normal reference ranges in these studies have varied widely and individual dogs show considerable day-to-day variation, thus routine clinical application of manometry in animal patients has been limited. In addition manometry in dogs and cats may require anesthesia, which affects the measurements, except in some studies where research dogs have been trained to swallow manometric tubes while awake.84,85 Esophageal manometry has been performed in dogs with idiopathic megaesophagus.85 New high-resolution manometry catheters used in people have up to 128 circumferential pressure sensors that enable complex computer analysis of swallowing in new ways.
Esophageal pH Monitoring
Continuous ambulatory monitoring of the caudal esophageal pH and impedance with an intraluminal pH probe is the gold standard for verifying gastroesophageal reflux in people. Esophageal pH monitoring has not found clinical application yet in dogs and cats, but it has been used as a research tool to assess gastroesophageal reflux in anesthetized dogs.86,87 A recent innovation in pH monitoring is a catheter-free pH sensor capsule (Bravo pH Monitoring System; Medtronic) that is attached endoscopically to the esophageal mucosa where it transmits continuous pH data wirelessly (by telemetry) to a mobile receiver. The pH data are then downloaded to a computer for analysis. An esophageal acid clearance test was evaluated in healthy awake dogs using a pH probe in the caudal esophagus that recorded the number of swallows and time required for esophageal peristalsis to increase the pH to 4.0 following instillation of a 10-mL bolus of 0.1 N hydrochloric acid.88 The mean clearance time was 5 minutes (range: 2.5 to 8) after eight swallows (range: 4 to 12).
Electromyography
Electromyography (EMG) can be used to evaluate striated muscle activity associated with swallowing, and it can be integrated with manometric and videoradiographic findings.53 Abnormal EMG findings include fibrillation potentials, positive sharp waves, increased insertional activity, and complex repetitive discharges. In myopathic and neuropathic esophageal diseases, the EMG may reveal abnormalities in the muscles of the pharynx, cricopharyngeal sphincter, and cranial esophagus.50,83 When esophageal disease is associated with polymyopathy, EMG of the masticatory and limb muscles may also be abnormal.6
Inflammation
Esophagitis
Etiology
Esophagitis is an acute or chronic inflammatory disorder of the esophageal mucosa that occasionally involves the underlying submucosa and muscularis. It may result from ingestion of caustic or corrosive substances, injury from esophageal foreign bodies, gastroesophageal reflux, radiation injury, idiopathic generalized megaesophagus, and inflammation associated with malignancy.1–5
Pathophysiology
The esophageal mucosa has several important barrier properties to withstand caustic substances, including stratified squamous epithelium with tight intracellular junctions, mucus gel, and surface bicarbonate ions. Disruption of any of these barrier properties may result in inflammation, erosion, and/or ulceration of the underlying structures.2,6–8 Clinical signs are related to the type of chemical injury, the severity of inflammation, and the involvement of structures underlying the esophageal mucosa, for example, the muscularis.9 Although esophagitis may occur at any age, young animals with congenital esophageal hiatal hernia may be at increased risk for reflux esophagitis.10,11 Cats appear to be particularly susceptible to doxycycline-associated esophagitis and esophageal stricture.12–14 Anesthesia, poor patient preparation, and poor patient positioning during anesthesia places other animals at risk for gastroesophageal reflux and esophagitis.15–19 Upper respiratory syndrome in brachycephalic breeds predisposes to reflux esophagitis.20,21
Diagnosis
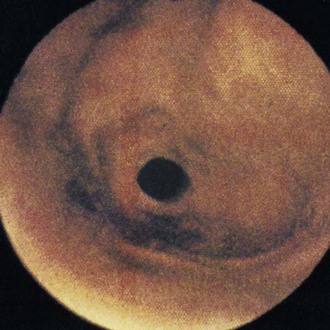
Leukocytosis and neutrophilia may be found in animals with severe esophagitis or aspiration pneumonia. Results of routine hematology, serum biochemistry, and urinalysis are otherwise nonremarkable. The esophagus often appears normal on survey thoracic radiographs unless there is some secondary dilation. Aspiration pneumonia may be evident in the dependent portions of the lung. Barium contrast radiographs include irregular mucosal surface, segmental narrowing, esophageal dilation, and/or diffuse esophageal hypomotility. Stricture formation may also be observed in chronic undiagnosed or untreated esophagitis. Endoscopy and biopsy are the most reliable means of diagnosing this disorder. In severe cases of esophagitis, the mucosa will appear hyperemic and edematous with areas of ulceration and active bleeding (Figure 55-2). Milder cases of esophagitis may appear endoscopically normal. Endoscopic biopsy of the esophageal mucosa is technically more difficult than other parts of the gastrointestinal tract,22 but mucosal biopsy will be necessary to confirm the diagnosis in mild to moderate cases. Esophagitis will have several important differential diagnoses, including mechanical injury as a result of esophageal foreign body, esophageal stricture, hiatal hernia, megaesophagus, esophageal diverticulum, and vascular ring anomaly. Each of these disorders can be differentiated by survey or contrast radiography, or endoscopy.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree