Chapter 37 Anthelmintic Agents
There are a number of cestodes, nematodes, and trematodes that are associated with gastrointestinal disease in dogs and cats. Chapters 55 to 58 provide an in-depth discussion of the clinical applications of anthelmintic drugs for these disease states. Some helminths can also be associated with clinical illness in humans and therefore management of infections in dogs and cats is part of a comprehensive public health program. This is particularly important for some of the hookworm (e.g., Ancylostoma spp., Uncinaria spp.) and roundworm (e.g., Toxocara spp., Baylisascaris procyonis) species. Approximately 3% to 5% of client-owned dogs and cats in the United States are currently shedding roundworm or hookworm eggs, with much higher shedding rates being detected in specific regions.1–5
This chapter provides information concerning the drugs most frequently used to remove or control helminth infections in dogs and cats. The majority of the compounds discussed are available in multiple countries. However, formulations and combinations of drugs may vary between countries. In addition, some of the drugs are used off-label for some parasites and label claims and dosages may change. Thus readers should always consult recent publications for potential changes. The majority of the comments in this chapter are written using resources and experiences from practice in the United States. Information is also drawn from the recommendations of the Companion Animal Parasite Council in the United States (www.capcvet.org) and the Compendium of Veterinary Products (CVP) drug label database in the United States.
Benzimidazoles
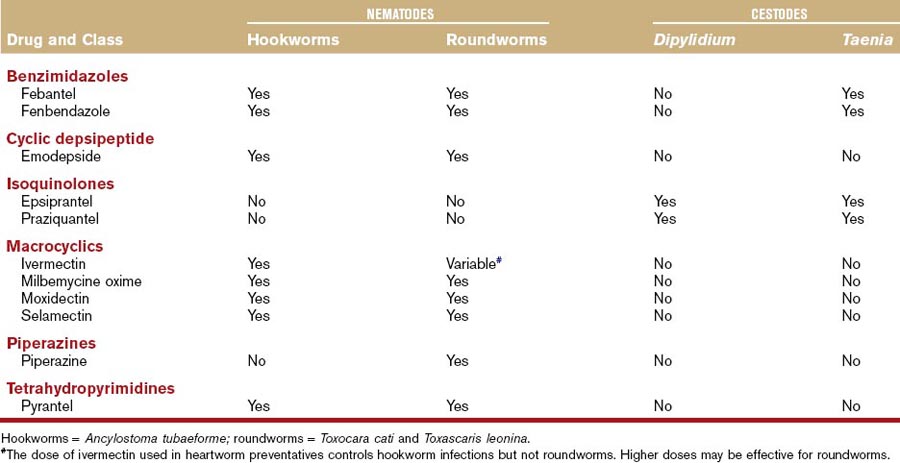
Historically, many drugs in the benzimidazole class (albendazole, fenbendazole, flubendazole, mebendazole, oxibendazole, thiabendazole) or probenzimidazole drugs (febantel) have been used as broad-spectrum anthelmintics in dogs and cats.6–11 There is no information available to suggest that albendazole, flubendazole, mebendazole, oxibendazole, or thiabendazole are more effective against gastrointestinal parasites in dogs or cats than fenbendazole or febantel. Mebendazole is associated with hepatic necrosis in dogs and albendazole is associated with bone marrow suppression in dogs and cats, which are additional reasons fenbendazole and febantel are prescribed more frequently.12,13 These drugs are considered among the best broad-spectrum agents because of activity against a variety of different parasites of the gastrointestinal system and other systems (Tables 37-1 and 37-2).
Table 37-1 Drugs Used in the Management of Common Helminth Infections of the Gastrointestinal System of Dogs
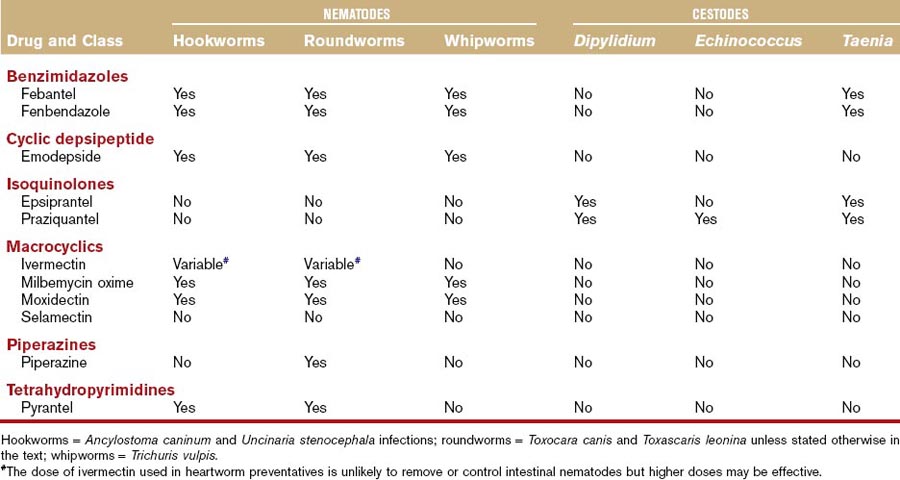
Table 37-2 Drugs Used in the Management of Common Helminth Infections of the Gastrointestinal System of Cats
Dose and Toxicity
If a labeled product containing fenbendazole or febantel is available, the label should be followed for each respective parasite. In general, fenbendazole at the dosage of 50 mg/kg, PO, daily for 3 days, is considered safe and effective against most of the target helminths of both dogs and cats (see Tables 37-1 and 37-2). In the United States, febantel is labeled only for oral administration in dogs. However, the drug has been administered safely to cats in several studies at dosages as high as 56.5 mg/kg (adult cats).11,14 Side effects at the recommended dosages in dogs and cats are rare, and if side effects occur, generally salivation or vomiting, they are usually mild and self-limiting.
Clinical Application
Fenbendazole and febantel are prescribed frequently for the target helminths as discussed (see Tables 37-1 and 37-2). However, neither compound is effective for the treatment of Dipylidium caninum or Echinococcus spp. infections. Febantel is combined with praziquantel (see “Isoquinolones” section), which effectively treats the other tapeworms and pyrantel (see “Tetrahydropyridimines” section), which increases its activity against hookworms and roundworms (Drontal Plus, Bayer Animal Health). Benzimidazoles also treat Trichuris vulpis infections, which is an advantage over some of the drugs in other classes with anthelmintic activity.
Fenbendazole or febantel are also commonly prescribed to dogs or cats with small bowel diarrhea for potential activity against Giardia spp. (see Chapter 57).14–17 Fenbendazole is one of the anthelmintics that is believed to have activity against the trematodes that can be associated with gastrointestinal disease (Alaria spp., Nanophyetus salmincola, Heterobilharzia americana, Platynosomum fastosum).18 However, optimal protocols have not yet been established and fenbendazole is not labeled for this use.
Cyclic Depsipeptide
Emodepside is the only drug in this class currently prescribed to dogs or cats. It has been evaluated for oral (dogs) or topical (cat) administration and is considered safe and effective for use in the management of several nematode infections in dogs or cats (see Tables 37-1 and 37-2).19–23 Emodepside is combined with praziquantel (Profender, Bayer Animal Health). The oral formulation for use in dogs is not currently available in the United States.
Dose and Toxicity
Based on the label in the United States, the emodepside-containing topical solution should be applied to cats older than 8 weeks of age and greater than 1 kg of body weight. The drug has been applied to small numbers of pregnant queens and 4-week-old kittens in a research study without apparent side effects.19 Adverse effects associated with administration of the topical solution to 606 cats in a controlled, double-masked field safety study reported by Bayer Animal Health were infrequent, mild, and self-limited. The reactions reported were licking/excessive grooming in 18 cats (3.0%), scratching treatment site in 15 cats (2.5%), salivation in 10 cats (1.7%), lethargy in 10 cats (1.7%), alopecia in eight cats (1.3%), agitation/nervousness in seven cats (1.2%), vomiting in six cats (1.0%), diarrhea in three cats (0.5%), eye irritation in three cats (0.5%), respiratory irritation in one cat (0.2%), and shaking/tremors in one cat (0.2%).21–23 Salivation, vomiting, and mild neurologic effects induced by administering the topical solution orally to cats were self-limited. Side effects induced by administration of the oral preparation to dogs have also been minimal.21–23
Clinical Application
In addition to the label indications, administration of emodepside to a small number of pregnant queens that were experimentally infected with Toxocara cati apparently blocked lactogenic transmission to the kittens.19 Combining emodepside with praziquantel expands the spectrum of activity to include cestodes and, potentially, trematodes (see “Isoquinolones” section).23 Emodepside also has effects against helminths of other body systems like feline lungworm.24
Isoquinolones
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


