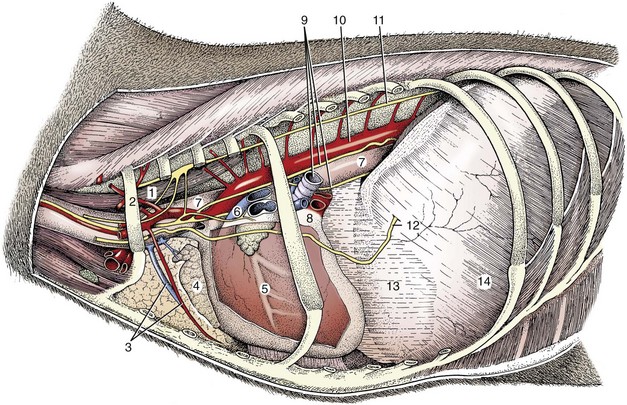
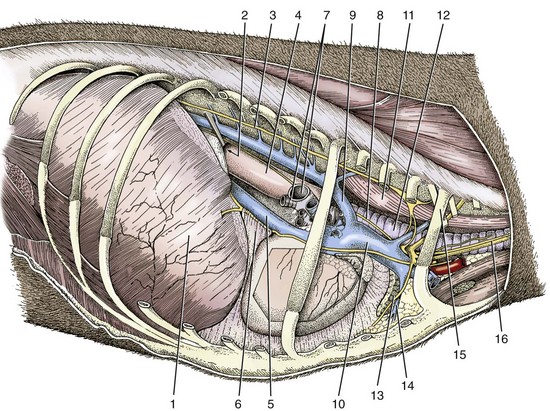
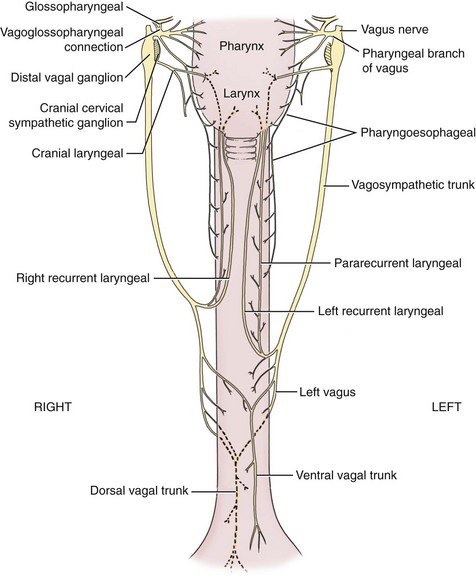
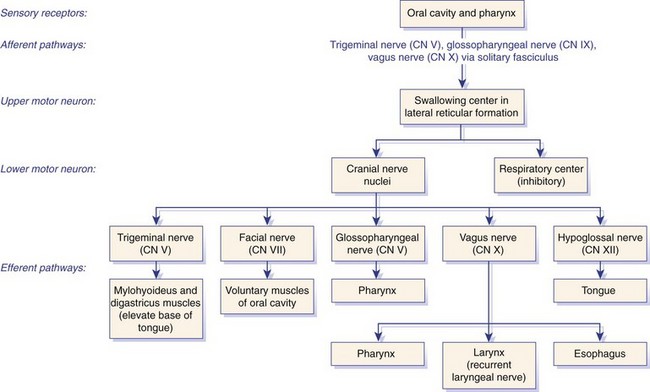
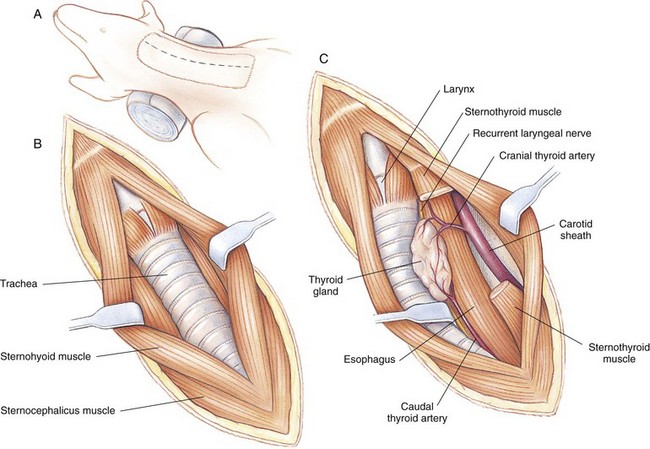
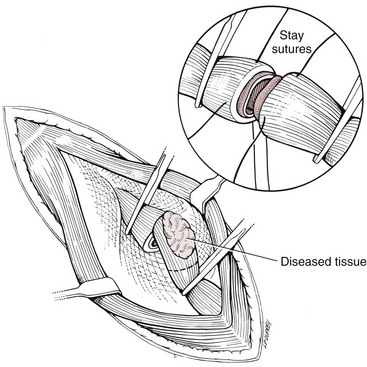
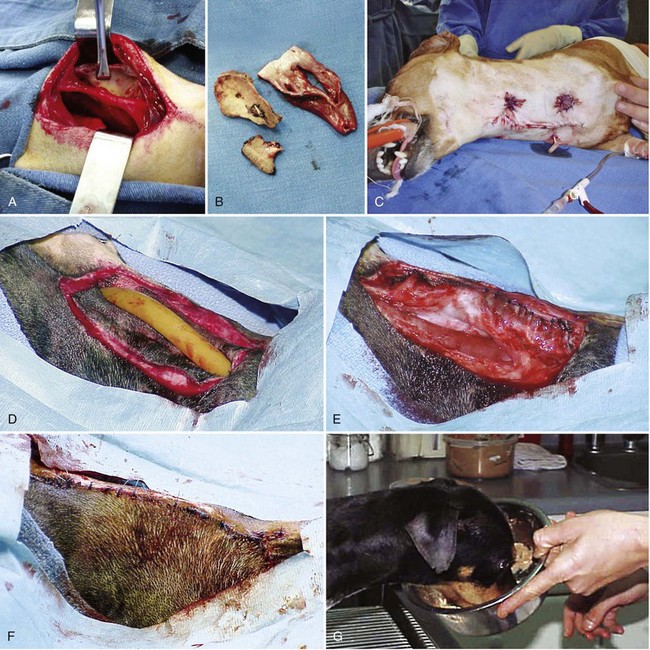
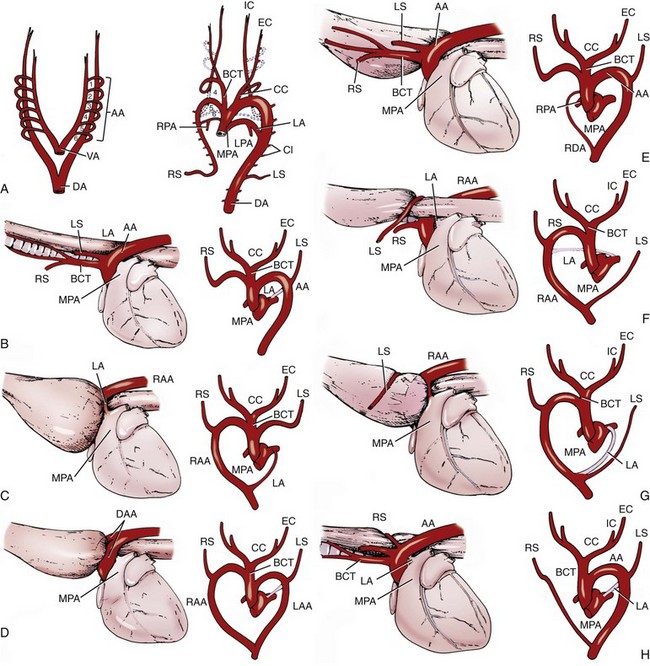
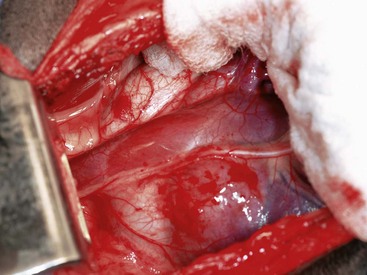
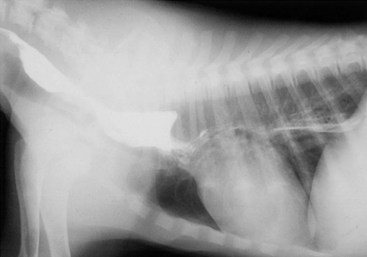
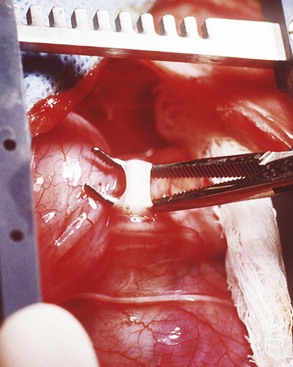
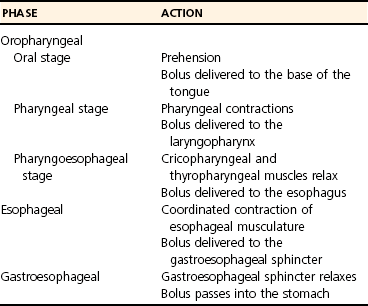
Chapter 90 The esophagus is the connecting tube between the pharynx and stomach that functions to transport ingesta and fluids.36 The cervical portion of the esophagus begins dorsal to the caudal border of the cricoid cartilage, inclines to the left of the trachea as it runs caudally, and ends at the thoracic inlet. The thoracic portion extends from the thoracic inlet, where it is located to the left of the trachea, crosses the trachea to regain its dorsal position at the tracheal bifurcation, and extends caudally to the esophageal hiatus of the diaphragm (Figures 90-1 and 90-2). The aorta obliquely crosses the left side of the midthoracic esophagus. In the caudal thorax, the dorsal branches of the left and right vagal nerves run across the side of the esophagus and unite dorsally to form the dorsal vagal trunk, and the left and right ventral branches similarly unite to form the ventral vagal trunk. Both vagal trunks enter the abdomen through the esophageal hiatus of the diaphragm. The abdominal portion of the esophagus is short and wedge shaped, extending from the diaphragmatic hiatus to the stomach. An anatomic study found that the intraabdominal portion of the esophagus in dogs could not be consistently demonstrated.129 The main arterial blood supply to the cervical esophagus is from branches of the cranial and caudal thyroid arteries. The bronchoesophageal artery is the main source of blood for the cranial two thirds of the thoracic esophagus; the remaining thoracic esophagus is supplied by esophageal branches of the aorta or dorsal intercostal arteries. The terminal portion is supplied by a branch of the left gastric artery. The veins that drain the esophagus are largely satellites of the arteries that supply it. Veins leaving the cervical esophagus drain into the external jugular veins, and those from the thoracic esophagus drain mostly into the azygous vein. The vein that accompanies the left gastric vein drains into the portal venous system. The esophageal arteries and veins form a rich, intramural plexus of anastomosing vessels in the submucosal layer. Esophageal lymphatic vessels drain into the medial retropharyngeal, deep cervical, cranial mediastinal, portal, splenic, and gastric lymph nodes. The esophagus is innervated by nerve fibers arising from various branches of the vagus, beginning with the paired pharyngoesophageal nerves followed by the recurrent laryngeal and paralaryngeal nerves and concluding with the dorsal and ventral vagal trunks (Figure 90-3). Normal swallowing is a coordinated process consisting of three phases: oropharyngeal, esophageal, and gastroesophageal. The oropharyngeal phase involves prehension of food, formation of a bolus in the mouth, and passage of the bolus into the cranial cervical esophagus. The oropharyngeal phase is subdivided into oral, pharyngeal, and cricopharyngeal stages (Figure 90-4; Table 90-1).128,169 The oral stage is a voluntary process involving prehension and mastication of food, formation of a bolus at the back of the tongue, and delivery of the bolus into the pharynx. The pharyngeal stage is the first step of the involuntary phase of swallowing and is initiated by delivery of a food bolus into the oropharynx. Pharyngeal constrictor muscles propel the bolus aborally into the cricopharynx. Contraction of the palatal and pharyngeal muscles closes the nasopharynx and prevents nasal reflux, and caudal epiglottic reflection and vocal fold adduction close the larynx and prevent laryngotracheal aspiration. The cricopharyngeal stage of swallowing involves the relaxation of thyropharyngeus and cricopharyngeus muscles and delivery of the bolus into the cranial cervical esophagus. The oropharyngeal phase of swallowing is coordinated by the trigeminal (V), facial (VII), glossopharyngeal (IX), vagal (X), and hypoglossal (XII) nerves. Table • 90-1 Data from Watrous BJ, Sutter PF: Normal swallowing in the dog: a cineradiographic study. Vet Radiol 20:99, 1979. Esophageal transit time is affected by body positioning and the type of meal.15 In healthy dogs, transit times of liquid and kibble through the cervical esophagus were 2.58 and 4.44 cm/sec, respectively, in sternal recumbency and 7.23 and 8.92 cm/sec, respectively, in right lateral recumbency. Swallows more frequently stimulated primary esophageal peristalsis in sternal recumbency than lateral recumbency. Esophageal surgery is historically associated with a higher prevalence of incisional dehiscence than surgery on other portions of the alimentary tract. Several factors may contribute to the high complication rate, including lack of serosa, the segmental nature of the blood supply, the lack of omentum, constant motion caused by swallowing and respiration, and tension at the surgical site.121 Despite these factors, esophageal surgery can be successfully performed by adhering to certain surgical principles, including gentle tissue handling, minimization of contamination, appropriate selection and application of suture materials, appropriate use of electrocautery, and accurate apposition of tissues.41,115 In the abdomen, the serosa is credited with assisting healing of viscera by the elaboration of a fibrin seal soon after surgery and by providing a source of pluripotential mesothelial cells.41,42 In the thoracic cavity, pleural mesothelium may play a similar role. However, the presence of serosa is not essential for visceral healing; experimentally, healing of intestinal anastomosis in dogs is achieved despite serosal stripping.41,52 The blood supply to the esophagus is considered segmental, with a rich, intramural plexus of anastomosing vessels in the submucosal layer that can support long segments of the esophagus.99,158,177 Thoracic esophageal anastomoses can heal successfully after ligation of the thoracic segmental blood supply, provided the intramural vascular supply and collateral circulation from the cervical and abdominal portions of the esophagus are intact.99 Simultaneous ligation of blood vessels to the cervical and thoracic esophagus, however, results in esophageal necrosis at the thoracic inlet.99 Most cases of ischemic necrosis at the esophageal incision are associated with damage to the intramural vascular supply of the esophagus rather than the segmental vessels.41,42,99 Esophageal patching with omental pedicles or other vascularized tissues may be helpful to reinforce esophageal wounds and improve vascularity. Postoperative motion of the esophageal incision with swallowing and respiration is unavoidable. After esophageal surgery, food and water are normally withheld to reduce esophageal motion and eliminate the passing of ingesta across the surgical site. Placement of a gastrostomy or enterostomy tube should be considered for postoperative enteral nutrition, particularly in patients with preoperative cachexia. Recommendations on the time period for withholding per os food and water vary between 24 hours and 7 days, depending on the author and the type of esophageal surgery.41,42,67 The cervical esophagus is approached by a ventral midline incision, separating the paired sternohyoid muscles and retracting the trachea to the right (Figure 90-5). Care should be taken to avoid damage to the recurrent laryngeal nerve during tracheal retraction. This approach can be extended via a cranial median sternotomy to expose the cranial thoracic esophagus to the level of the tracheal bifurcation. The cranial thoracic esophagus can also be exposed via a left third or fourth or a right third, fourth, or fifth intercostal thoracotomy.63,115 The esophagus is exposed from a left-sided thoracotomy by ventral retraction of the brachiocephalic trunk and subclavian vessels. On the right side, the esophagus is exposed by ventral retraction of the trachea, which can be combined with retraction or ligation of the azygous vein to increase exposure of the esophagus at the base of the heart. A left-sided seventh, eighth, or ninth intercostal thoracotomy is generally preferred for approaching the caudal thoracic esophagus to avoid the caudal vena cava. The dorsal and ventral vagal nerves should be identified and preserved. The caudal esophagus and stomach can be approached by a combined ventral midline laparotomy and diaphragmatic incision or caudal median sternotomy. Monofilament, minimally reactive, slowly absorbable suture materials, such as polydioxanone or polyglyconate, are often recommended for closure of esophageal incisions.41 Sutures should be placed approximately 2 mm from the cut edge and 2 to 3 mm apart.139 A multitude of esophageal suturing techniques have been advocated in dogs and cats,41 including double-layer appositional and single-layer inverting, everting, and appositional.114,139,144,157 Traditionally, a two-layer closure technique is used, consisting of a first layer incorporating the mucosa or submucosa with placement of the knots in the esophageal lumen and a second layer consisting of an inverting pattern in the muscularis.139 The rationale for this suture technique was based on the assumptions that the thick stratified squamous mucosal layer was the holding layer of the esophagus and that suture knots in the submucosa would provoke inflammation and fibrosis.41 In a study of the functional suture-holding layer of the canine esophagus, there was no significant difference between the tensile strength of the submucosa alone and the mucosa and submucosa combined, with the full esophageal wall having the greatest tensile strength and the muscularis alone the least.28 The author concluded that the submucosa was the functional suture-holding layer of the esophagus and therefore should be incorporated in at least one layer of sutures.28 In another study, esophagotomy incisions in healthy dogs healed adequately with double-layer simple interrupted, single-layer simple continuous, or single-layer simple interrupted patterns.114 Because incisions closed with a simple continuous single-layer pattern had the least wound strength and poorest tissue apposition, a single-layer simple interrupted or double-layer suture pattern was recommended.114 An interrupted pattern is generally preferred for esophageal anastomoses to permit esophageal dilatation and to avoid potential interference with the intramural blood supply, although a simple continuous pattern is acceptable for esophagotomy closure.41 Single-layer interrupted closure of esophagotomy and esophageal anastomoses using an absorbable suture material has been used successfully clinically in dogs and cats.144 Indications for esophagotomy include foreign body removal and closure of esophageal perforations and diverticula. Esophagotomy can be performed on any portion of the esophagus. The surgical site is isolated with moistened laparotomy sponges, and the esophagus is suctioned transorally to reduce contamination. If fluid or ingesta is present in the esophagus, the esophageal lumen can be occluded cranial and caudal to the proposed incision site with fingers, umbilical tape, or noncrushing clamps. Alternatively, stay sutures and suction can be used to minimize spillage (Figure 90-6). A stab incision is made into the esophageal lumen, and the incision is extended longitudinally as necessary. The esophagotomy incision can be closed with a one- or two-layer interrupted or continuous suture pattern. With a two-layer pattern, the first layer incorporates mucosa and submucosa, and knots are placed in the esophageal lumen. The second layer apposes the muscularis and adventitia, with knots placed extralumenally. With a one-layer closure, the suture passes through all layers of the esophageal wall, with limited penetration of the mucosa, and knots are placed extralumenally. Sutures should be placed approximately 2 mm from the cut edge and 2 to 3 mm apart.139 Integrity of the closure can be tested by distending the esophagus with saline and placing additional sutures to seal any areas of leakage. The patient should not receive food or water by mouth for at least 24 to 48 hours after surgery.67 Healing of a cervical esophagotomy incision can be delayed by contact with pharyngostomy tubes. Indications for esophageal resection and anastomosis include treatment of congenital obstructions, acquired discrete strictures, severe localized esophageal injury, esophageal diverticula that cannot be excised by simple esophagotomy, and esophageal neoplasia.115 The extent of the proposed resection should be estimated before surgery by esophagoscopy, contrast esophagography, magnetic resonance imaging, or computed tomography (CT). The length of esophagus that can be resected without undue risk of dehiscence is limited by anastomotic tension.115 Resection and primary anastomoses of up to 20% of the cervical esophagus and 50% of the thoracic esophagus have been reported in experimental dogs;99,141 in clinical patients, however, resection of more than 3 to 5 cm of esophagus has been associated with an increased risk of dehiscence.63 Esophageal substitution techniques should be considered when extensive esophageal resection is unavoidable, although clinical experience of these techniques in dogs and cats is limited. Circumferential partial myotomy may reduce anastomotic tension. The outer muscle layer is incised 2 to 3 cm proximal or distal to the anastomosis, or both, leaving the inner muscle layer intact.40,67,108 Separation of the muscle layers may be improved by injecting saline into the muscularis.108 Use of a partial myotomy prevents disruption of the submucosal vascular plexus and subsequent esophageal ischemia, which are observed with full-thickness myotomy, while providing a similar reduction in anastomotic tension.108 The myotomy heals by secondary intention without stricture or diverticulum formation. Esophageal resection and anastomosis can be performed on any portion of the esophagus. A small amount of esophageal mobilization during surgery is necessary to control spillage and reduce anastomotic tension, but excessive mobilization may compromise segmental esophageal blood supply. The esophagus should be isolated with moistened laparotomy sponges, and the lumen can be occluded with fingers, umbilical tape, or noncrushing clamps (Figure 90-7). The diseased portion of the esophagus is resected. Stay sutures facilitate gentle tissue handling and maintain alignment. The remaining segments are reapposed with a one- or two-layer suture pattern as described for esophagotomy, with the far esophageal wall sutured first. Esophageal anastomoses can also be performed with surgical stapling instruments or using a biofragmentable anastomosis ring.88,89,122,154 The stapling device can be inserted through the mouth or through a pharyngotomy, esophagotomy, or gastrotomy incision.122 In one study comparing biofragmentable anastomosis ring with an end-to-end anastomotic stapler, there was no significant difference in the complication rate or healing with either method. The mortality rate was 13.3% (four of 30 biofragmentable anastomosis ring dogs), and leaks were noted only in the biofragmentable anastomosis ring group (14.3%). Two of 18 dogs in the stapler group developed strictures within 28 days after anastomosis.89 After esophageal resection and anastomosis, fluid and nutritional requirements are provided for at least 1 week through a gastrostomy or enterostomy tube.67 Contrast esophagograms may be performed at 1 and 3 months after surgery to evaluate esophageal structure and function.115 Patches can be used to reinforce existing esophagus (“on-lay” patch) or for partial circumferential replacement of the esophageal wall (“in-lay” patch). On-lay patches are applied on esophagotomy incisions or esophageal anastomoses that are at risk for dehiscence because of reduced vascularity or anastomotic tension.41,115 The most common application for in-lay patches is for esophagoplasty after longitudinal division of an esophageal stricture.115 Materials that can be used for esophageal patching include host tissues, such as omentum, pericardium, local muscle flaps, stomach, or intestine, and xenogeneic or artificial substances, such as porcine small intestinal submucosa, lyophilized dura mater, collagen-coated Vicryl mesh, and expanded polytetrafluoroethylene patches.* Omentum is exceptional for supporting caudal thoracic esophageal wounds and closing small defects because of its vascularity and availability; esophageal stricture formation may also be minimized with omental patching.41,62,70,106 A paracostal or midline abdominal incision is necessary to create an omental pedicle flap. The right gastroepiploic artery and its branches to the omentum are ligated. The flap is brought through a small incision in the diaphragm, taking care to preserve the blood supply. The omentum is then tacked at multiple sites around the esophageal wound with absorbable suture. Pedicle grafts can be created from local muscle to support the esophagus. Cervical esophagus has been successfully patched with sternothyroideus and longus colli muscles.16,73 Experimentally, longus colli muscle patch graft in dogs reduced stricture formation when compared with primary closure of a partial circumferential cervical esophageal defect. The addition of a free buccal mucosal graft further reduced the extent of stricture formation.16 In the thoracic esophagus, a vascularized pedicle graft of external and internal intercostal muscle can be created by incision and elevation of the periosteum of two adjacent ribs, with division of either the dorsal or ventral attachment.22 Diaphragmatic pedicle grafts can be used to patch the caudal thoracic esophagus.92 Pericardial grafts significantly increase the strength of esophageal anastomoses without increasing the risk of stricture formation.69 Esophageal patches can also be created from the gastric wall74,75 or jejunum after mucosal stripping.111 The adjacent lung has been used experimentally in dogs to repair esophageal defects;104 this technique should be used with caution because of the risk of esophagopulmonary fistula formation. Patients with extensive disease from injury or neoplasia may require massive esophageal resection.41 If primary anastomosis is not feasible because of expected tension, esophageal substitution may be considered. Reports of esophageal substitution in humans and experimental animals are fairly common; however, clinical experience with esophageal replacement in small animals is minimal. The cervical esophagus can be replaced in a multistage procedure with an inverse tubed skin graft.121,161 A cranially based single pedicle skin flap is formed into an inverse tube and sutured distally to the cut end of the esophagus.121 Two weeks later, the cranial skin pedicle is cut and anastomosed cranially to the remaining end of the esophagus. Alternatively, the proximal and distal ends of the cervical esophagus can be sutured to the skin after esophageal resection to form two esophagocutaneous fistulae (Figure 90-8).161 Several weeks later, the skin is incised and inverted to form a tube connecting the two ends of the esophagus. The remaining cervical skin is closed over the top of the tube. An omocervical cutaneous island axial pattern flap, based on the omocervical vessels, has been advocated as an alternative one-stage inverse tubed skin graft for substitution of the cervical esophagus.120 Potential complications of esophageal skin graft include luminal obstruction with hair, lack of peristalsis, and indistensibility of the graft. Muscle grafts have been used experimentally in dogs for complete segmental reconstruction of the esophagus.107,156,183 The cranial thoracic esophagus was successfully replaced with a tubed intercostal musculopleural pedicle graft,107 and the caudal thoracic esophagus was replaced with a diaphragmatic pedicle graft.119 A longer segment of the esophagus was successfully reconstructed with an epithelial-lined esophageal conduit using a tubed latissimus dorsi musculocutaneous flap based on the thoracodorsal vessels.182,183 The caudal-most portion of the thoracic esophagus can be replaced by gastric advancement and esophagogastric anastomosis.23,25,122,154 Potential complications include leakage, stricture, chronic vomiting, reflux esophagitis, gastric dilatation within the thoracic cavity, and herniation of other abdominal viscera through an inadequately reconstructed diaphragm.41 Isoperistaltic and antiperistaltic gastric tubes created from the greater curvature of the stomach and based on the gastroepiploic vessels may be better alternatives for caudal esophageal replacement.65 Splenectomy and ligation of the omental branches of the gastroepiploic vessels are required with these techniques; surgery is greatly facilitated by the use of stapling instruments.65,154 An antiperistaltic, or reverse, gastric tube is created from the greater curvature of the stomach, maintaining the luminal connection at the fundus. The tube is advanced into the thoracic cavity and anastomosed to the cut end of the esophagus. An isoperistaltic gastric tube is created in a similar fashion, but the tube is separated at both ends to create a pedicle graft. The isoperistaltic tube technique maintains gastric tube peristalsis in an aboral direction but requires two anastomoses. With both techniques, a portion of the gastric tube should be maintained within the abdomen to reduce the risk of gastroesophageal reflux, and a pyloromyoplasty procedure should be performed to enhance gastric emptying because disruption of vagal innervation is likely.23,65 Animals are fed by jejunostomy tubes for at least 1 week after surgery.41 In dogs, segmental esophageal replacement with a small intestinal or colonic pedicle graft is hampered by the limited mobility of the vascular pedicle; free microvascular grafts of the intestine are therefore considered more feasible.41 Colonic autografts are more tolerant of ischemia than jejunum and thus provide a longer length of intestine for single-pedicle anastomosis. Additionally, the middle colic vessels are larger than the jejunal vessels, making the procedure less technically demanding.90 Experimentally, replacement of the canine thoracic esophagus with free intestinal grafts has been unsuccessful because of vascular thrombosis and anastomotic leakage.17,90 In the cervical region, the graft vessels can be successfully anastomosed to the carotid or cranial thyroid artery and the jugular, omobrachial, or lingofacial vein.17,55,90 In one dog with an esophageal stricture, the cervical esophagus was replaced with a free jejunal autograft.56 Complications included leakage and aspiration pneumonia. After surgery, decompression of the stomach and esophagus should be maintained with a nasogastric tube to reduce intraluminal pressure and prevent disruption of the anastomosis.56 Vascular rings are developmental anomalies of the great vessels that result in the encircling of esophagus and trachea by a complete or incomplete ring of vessels. In embryos, the great vessels are derived from the paired dorsal and ventral aortas and the six interconnecting pairs of aortic arches that correspond to the six brachial arches (Figure 90-9). The vascular system forms around the embryonic foregut and pulmonary bud, and as the embryo develops, selective involution and reconnection of the vessels result in the formation of the definitive cardiovascular system and release of the esophagus and trachea.35,41,165,181 Several types of vascular ring anomalies are described in dogs and cats (see Figure 90-9). The most common vascular ring anomaly is persistent right aortic arch with a left ligamentum arteriosum, in which the aortic arch develops from the right fourth aortic arch. Persistent right aortic arch and right ductus arteriosum would be the mirror image of the normal anatomy and would not result in a vascular ring. Persistent right aortic arch with an aberrant left subclavian artery occurs when the left subclavian artery passes dorsal to the esophagus from the right-sided aorta, resulting in an incomplete vascular ring. Persistent right aortic arch with an aberrant left subclavian artery has been described in conjunction with either a left ligamentum arteriosum, resulting in the formation of two strictures, or with a right ligamentum arteriosum, resulting in a single stricture associated with the aberrant subclavian artery.20,72,163 Double aortic arch results from persistence of both left and right fourth aortic arches and can produce significant tracheal stenosis.41,166, Persistent right ligamentum arteriosum with normal (left) aortic arch is the mirror image of a persistent right aortic arch and, although uncommon, is significant in that it cannot be corrected via a left lateral thoracotomy.68,76 Aberrant right subclavian artery with normal (left) aortic arch occurs when the right subclavian artery passes dorsal to the esophagus, resulting in an incomplete vascular ring.13,162 To the author’s knowledge, an aberrant right subclavian artery combined with a right ligamentum arteriosum has not been reported in dogs. Persistent left cranial vena cava with a non-elastic band around the esophagus has been reported.94 Vascular ring anomalies are reported less frequently in cats and include persistent right aortic arch with left ligamentum arteriosum, persistent right ligamentum arteriosum with a left aortic arch, persistent right aortic arch with a right ligamentum arteriosum and aberrant left subclavian artery, and double aortic arch.* Congenital anomalies of the great vessels have been reported in 20% of dogs and cats, but most variants are not clinically significant.149 The most common vascular ring anomaly described in humans and dogs (6%; 8 of 136 dogs) is an aberrant right subclavian artery; this anomaly frequently does not result in clinical signs.18,149 Persistent right aortic arch with left ligamentum arteriosum accounts for 95% of clinical cases of vascular ring anomalies reported in dogs.41,165 The ductus arteriosus may remain patent in up to 10% of dogs with persistent right aortic arch.165 Persistent left cranial vena cava (Figure 90-10) has been reported in up to 45% of dogs with vascular ring anomalies;165 this anomaly can complicate surgical approach. Vascular ring anomalies can occur in any breed of dog; however, 92% of dogs reported with persistent right aortic arch are of breeds with an adult body weight greater than 15 kg.148 German shepherds and Irish setters have a greater risk compared with the general clinic population.148 In cats, vascular ring anomalies may be more common in Persians and Siamese.35 There is no obvious sex predilection.148 The condition may affect multiple animals in the same litter.35,63,68 Because of familial and breed tendencies, affected individuals should be neutered.41 Affected animals are usually considered normal until weaning, with clinical signs of postprandial regurgitation developing after the animal starts to ingest solid food. In one large retrospective series, 20% and 80% of dogs were diagnosed before 2 and 6 months of age, respectively.148 Occasionally, vascular ring anomalies are not diagnosed until later in life; dogs have been diagnosed with persistent right aortic arch up to 10 years of age.40,77 The clinical signs of vascular ring anomalies are caused primarily by esophageal obstruction. Postprandial regurgitation of undigested food is first manifested some time after weaning. Initially, regurgitation usually occurs soon after eating, but later it may occur at variable time (minutes to hours).63 Affected animals may grow more slowly than their littermates and appear malnourished despite a ravenous appetite. Sometimes an enlarged cervical esophagus can be palpated, particularly after a meal or with compression of the chest or abdomen. Coughing and respiratory distress may be associated with episodes of aspiration pneumonia or with tracheal compression secondary to a double aortic arch. Signs of cardiac disease are uncommon, although vascular ring anomalies have been associated with patent ductus arteriosus and occasionally with other congenital cardiac malformations. Diagnosis of vascular ring anomalies is based on signalment, history, physical examination, and results of radiographs or esophagoscopy.41 Clinicopathologic parameters are usually normal unless there is coexisting aspiration pneumonia or the animal is severely debilitated. On survey thoracic radiographs, the esophagus cranial to the heart may be dilated with air, fluid, or ingesta. On the ventrodorsal projection, it may be possible to identify the descending aorta on the right side of the esophagus in animals with persistent right aortic arch. The position of the aorta is normal in animals with persistent right ligamentum arteriosum and a left aortic arch. Persistent right aortic arch can also cause tracheal deviation, with a characteristic marked, focal leftward curvature of the trachea near the cranial border of the heart on the ventrodorsal projection, which can be combined with a focal tracheal narrowing.19 Radiographic signs of aspiration pneumonia can also be present. Cranial esophageal dilatation and esophageal obstruction over the base of the heart can be confirmed with positive-contrast radiography using barium in a suspension or in food (Figure 90-11). The esophagus caudal to the heart is usually normal in size but can occasionally be dilated.35 Fluoroscopy can be used to document abnormal esophageal peristaltic activity. Angiography or CT, although rarely performed, may be helpful in determining the type of vascular ring anomaly.83 Esophagoscopy can be used to rule out other causes of esophageal obstruction and to evaluate the esophageal mucosa for erosions and ulcerations that are often present in the dilated cranial esophagus.41 The presence of an aortic pulse on the right side of the esophagus on esophagoscopy supports the diagnosis of a persistent right aortic arch. Persistent right ligamentum arteriosum and a left aortic arch result in a left-sided aortic pulse with concurrent extraluminal obstruction at the heart base. Long-term medical management is usually unrewarding because the degree of esophageal dilatation often increases with age.35,165 Preoperative medical management should be directed toward the secondary effects of the vascular ring anomaly. Malnourished patients are treated with elevated feeding of a slurry diet. The food is offered so that the animal eats while standing on its hind legs; this position is maintained for 10 to 20 minutes after feeding to encourage esophageal emptying. In severely debilitated animals, nutrition is provided by gastrostomy tube feeding. For patients with aspiration pneumonia, antibiotics and appropriate medical management should be initiated and the surgery delayed. Because many patients are young and malnourished, intravenous fluids with dextrose should be provided during surgery and immediately after recovery. Persistent right aortic arch with a left ligamentum arteriosum is approached through a left fourth intercostal thoracotomy in dogs; in cats, a left fifth intercostal thoracotomy may be preferred.41 The cranial lung lobe is packed off caudally with moistened sponges. The dilated cranial thoracic esophagus, tapering abruptly over the base of the heart, is usually the most obvious feature initially encountered and may obscure the ligamentum arteriosum. A left persistent cranial vena cava, if present, can be retracted dorsally and does not need to be ligated. If a prominent hemiazygos vein is also present, it should be ligated and divided as it courses close to the vascular ring.35 The mediastinum is incised longitudinally over the vascular ring, carefully preserving the vagal nerve and its branch, the left recurrent laryngeal nerve, which loops around the caudal aspect of the ligamentum arteriosum. The pulmonary artery, aorta, and connecting ligamentum arteriosum are identified. The ligamentum arteriosum is usually longer than a normal ductus arteriosus but retains its cylindrical shape.41 The ligamentum arteriosum is carefully dissected from the esophagus with right-angled forceps (Figure 90-12), double ligated with silk suture, and transected. A stiff tube or balloon catheter is then inserted through the mouth into the esophagus, and any remaining constricting fibrous bands are identified and transected carefully so as not to penetrate through the thin-walled esophagus. The tube or balloon is passed beyond the constricted area and back to ensure that adequate dilatation has been achieved. Esophageal casts may form in the dilated segment of the esophagus; if they cannot be dislodged by manual kneading, esophagotomy may be required for removal.41 Resection of the redundant esophagus and plication of the dilated cranial thoracic esophagus with nonpenetrating Lembert-type gathering sutures have been described. These techniques are not recommended because they do not improve the ability of the esophagus to propel ingesta aborally and increase the risk of complications.41,63,64 A thoracostomy tube is placed, and a local block of intercostal nerves can be administered before routine thoracic closure. Thorascopic treatment of persistent right aortic arch has been described.100
Esophagus
Anatomy
Esophageal Blood Supply and Innervation
Physiology of Swallowing
General Surgical Principals
Surgical Approaches to the Esophagus
Suture Materials and Techniques
Surgical Techniques
Esophageal Resection and Anastomosis
Esophageal Patching
Esophageal Substitution
Diseases of the Esophagus
Clinical Signs
Diagnosis
Treatment
< div class='tao-gold-member'>
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Esophagus
Only gold members can continue reading. Log In or Register to continue