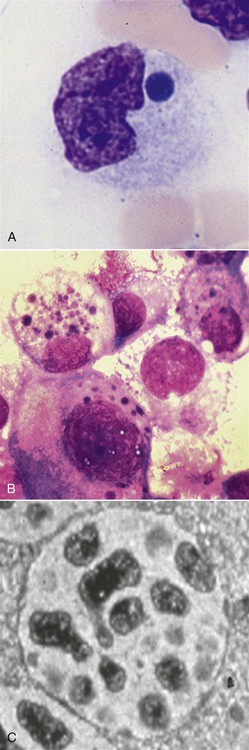
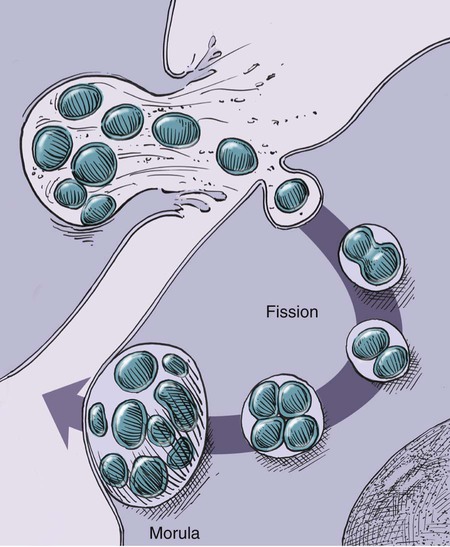
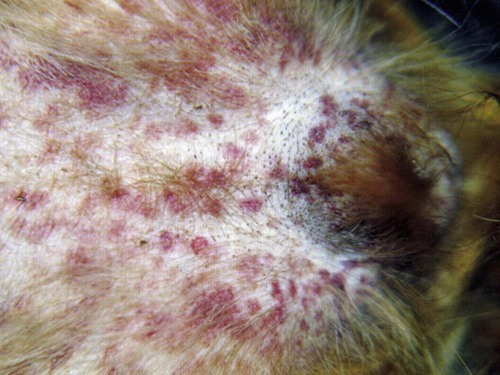
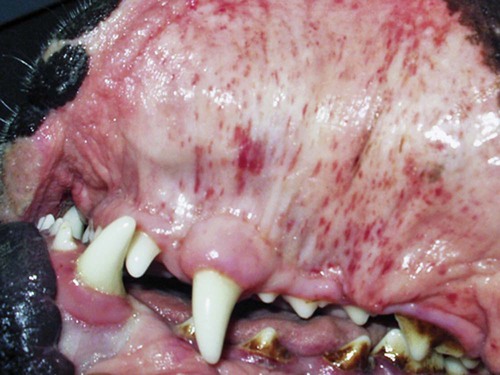
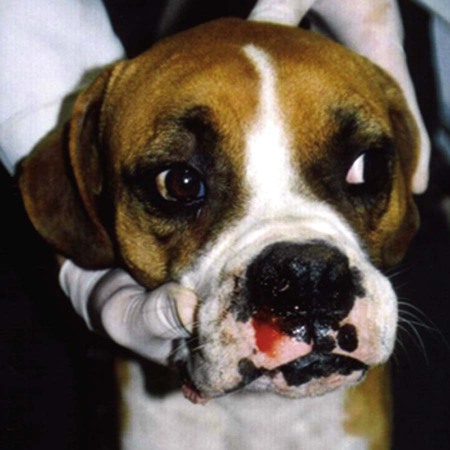
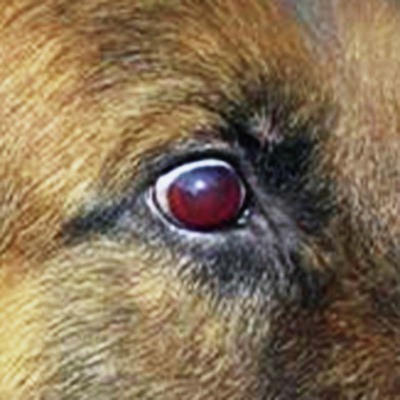
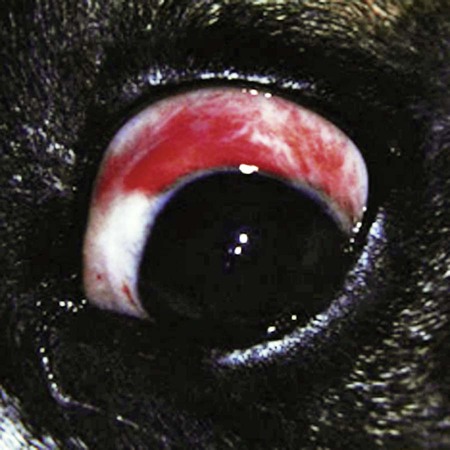
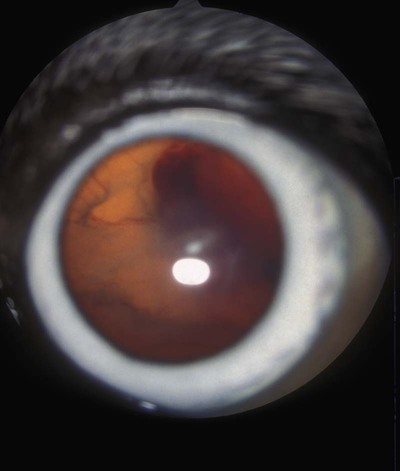
Rickettsiae that cause ehrlichiosis and anaplasmosis are classified as bacteria within the group α-proteobacteria. They are intermediate between viruses, which are obligate intracellular organisms, and bacteria, which use oxygen, have metabolic enzymes, are susceptible to some antibacterial drugs, and possess cell walls. Members of α-proteobacteria, within the families Rickettsiaceae and Anaplasmataceae of the order Rickettsiales, underwent reclassification in 2001. Reclassification was indicated after the molecular analyses of bacterial 16S rRNA and groESL gene sequences were elucidated.243 The genera Ehrlichia and Wolbachia (see Chapter 25) were moved from the family Rickettsiaceae to the family Anaplasmataceae. The genera Rickettsia and Orientia (see Chapter 27) remained in the family Rickettsiaceae. In addition, species that were previously included within the genus Ehrlichia (Ehrlichia phagocytophilum, Ehrlichia equi, and Ehrlichia platys) have been moved to the genus Anaplasma, and Ehrlichia risticii and Ehrlichia sennetsu have been moved to the genus Neorickettsia (see Chapter 25). A new proposed genus, Neoehrlichia, contains and organism Candidatus Neoehrlichia mikurensis, that has been isolated from rodents and a human with septicemia.276a Other closely-related organisms, identified in ticks and wildlife, have not yet been classified. The genus Ehrlichia consists of tick-transmitted gram-negative obligate intracellular bacteria that infect primarily leukocytes (monocytes, macrophages, granulocytes). These acid-sensitive, pleomorphic coccobacilli of approximately 0.5 µm diameter are aerobes that lack a glycolytic pathway.675 Ehrlichia canis, Ehrlichia chaffeensis, Ehrlichia ewingii, and Ehrlichia muris remained members of the genus Ehrlichia. Ehrlichia ruminantium (previously named Cowdria ruminantium), the cause of heartwater disease of cattle in Africa, has been added to this genus. The family Anaplasmataceae is composed of obligatory gram-negative intracellular organisms that parasitize leukocytes, erythrocytes, endothelial cells, or platelets. These agents are naturally transmitted to humans and a wide variety of domestic animals including dogs and cats from specific ticks that acquire their infections by feeding on mammalian wildlife reservoirs. The designation of Anaplasma phagocytophilum as a species group now comprises the past designations of the following: all strains of human granulocytotropic Ehrlichia (HGE) described worldwide; Ehrlichia equi in the Western United States; and the European agent infecting ruminants and causing tick-borne fever, Ehrlichia phagocytophila/um. They appear biologically similar, except for geographic, host, and pathogenic adaptations. Subtle genetic differences, described later in the A. phagocytophilum section, help explain these variants. Anaplasma bovis, Anaplasma platys, and A. phagocytophilum have all been classified within the genus Anaplasma because of their close genetic relationship to Anaplasma marginale. Molecular genetic data suggests that dogs in Japan may be infected with A. bovis.749 This chapter discusses canine and feline diseases caused by the organisms just described. Historically, they have been named according to the genus of the infectious agent, the target cell parasitized (preferentially but not exclusively) and the hosting animal (e.g., canine monocytotropic ehrlichiosis [organisms predominantly residing in monocytes and macrophages], canine/feline granulocytotropic anaplasmosis [organisms predominantly in neutrophils and eosinophils], and canine thrombocytotropic anaplasmosis [organisms in platelets]) (Table 26-1). With the advent of molecular genetic testing to confirm the genus and species of infecting organisms, and to avoid ambiguity, this chapter uses the specific organism labels where possible. TABLE 26-1 Monocytotropic Ehrlichia and Anaplasma Species Infecting Domestic Animals Endothel, Endothelial cells; lymph, lymphocytes; macro, macrophages; mono, monocytes; neut, neutrophils; ?, uncertain. aNaturally infected hosts have also been experimentally infected; however, they are always listed in this column. Some of these experimental infections are subclinical or transient. bOnly experimental infections are documented with this vector. cPreliminary identification of E. ewingii in Brazil632 and Cameroon602 by molecular genetic analysis requires further studies to determine the extent of its occurrence and significance in those regions. Shimon Harrus, Trevor Waner, and T. Mark Neer Canine monocytotropic ehrlichiosis (CME) is caused by the obligate intracellular organism E. canis (see Table 26-1).243 This small pleomorphic gram-negative coccoid bacterium appears intracytoplasmically within monocytes and macrophages in clusters of organisms called morulae (Fig. 26-1). The organism was initially identified in dogs by Donetein and Lestoquard in Algeria in 1935.223 However, the disease did not gain considerable attention until large numbers of American military dogs, many of which were highly susceptible German shepherds, died from the disease during the Vietnam War. E. canis received further attention in the late 1980s when the rickettsia was erroneously suspected to infect humans. However, in 1991, a new species of the Ehrlichia genus, E. chaffeensis, was found to be the cause of human monocytotropic ehrlichiosis.27 E. canis is established as a ubiquitous cause of morbidity and mortality in dogs. It has a worldwide distribution including Asia, Africa, Europe, and the Americas. Australia appears to be free of E. canis infection.545 The disease has been reported in Japan.827 Vertebrate hosts for E. canis include members of the family Canidae. The coyote, fox, and jackal, in addition to the domestic dog, are considered reservoir hosts (see Table 26-1). A single publication suggests that E. canis or a closely related organism might also infect cats (see Feline Monocytotropic Ehrlichiosis, later).111 The arthropod vector of E. canis is the brown dog tick, Rhipicephalus sanguineus (Fig. 26-2). This one-host tick preferentially feeds on dogs in all three stages of its life cycle and can live indoors in domiciled environments in which dogs are housed. Experimentally, infection has also been transmitted by Dermacentor variabilis, the American dog tick.419 The mode of transmission is transstadial, whereby the infection is transmitted to subsequent tick stages, but not to the maternal eggs of the next generation. Ticks acquire E. canis as either larvae or nymphs by feeding on rickettsemic dogs and transmit infection for at least 155 days thereafter to susceptible dogs.342 This capability permits the pathogen to overwinter in the tick and then in the following spring allows the tick to infest and infect susceptible dogs. Most CME cases occur during the warm season when the vector ticks are abundant; however, the disease may occur all year long as a result of the prolonged subclinical period in chronically infected animals. The minimal time required for tick attachment to transmit E. canis infection has yet to be determined.439 Dogs living in or traveling to endemic regions are candidates for the disease. Sequencing of the complete genome of E. canis has helped to identify genes involved in the course of the disease allowing for a better understanding of the pathogenesis of CME. The genome consists of a single circular chromosome of 1,315,030 nucleotides. A notable finding is its small size (984 genes) relative to other ehrlichiae with the presence of a similar number of predicted genes. In accordance with the organism’s obligate intracellular existence, the genome has undergone marked loss of metabolic pathway enzymes.562 Because of the close genetic similarity between E. canis and E. chaffeensis, it is reasonable to assume that scientific findings from either organism may be applicable to both agents. Of the two organisms, E. chaffeensis has been more extensively studied because of its human health importance. Immunoreactive surface proteins suspected to be adhesins have been identified in E. chaffeensis and E. ruminantium that possess repeat units with high serine and threonine content. It has been suggested that the adhesins are involved in the ehrlichial attachment and entry into the host cell.562,676,676 Because of the lack of many genes involved in metabolism, the organism must take up various nutrients from the surrounding milieu through pores or channels in its outer membrane. Outer membrane proteins P28 and OMP-1F in E. chaffeensis organisms possess porin activities that would allow for the organism to regulate nutrient uptake during intracellular development.467 Analysis of the 16S rDNA and antigen-coding genes has shown that multiple clades of E. canis exist.388a,389a This information may be beneficial in the future to characterize isolates for epidemiologic purposes. The incubation period of CME is 8 to 20 days. The organisms multiply in macrophages of the mononuclear phagocyte system by binary fission (see Fig. 26-1, C) and spread throughout the body (Fig. 26-3). Infection is thought to spread between cells through exit and uptake via adjacent cytoplasmic projections.846a Replication in the host takes place in secluded membrane-bound vacuoles protected from the host immune surveillance system, lysosomes, and oxygen-reactive intermediates. A mechanism for adaptation that allows ehrlichiae to reside within vacuoles and communicate with the host cell through the endoplasmic reticulum has been identified in a group of ankyrin genes encoding proteins that are suggested to mediate specific protein-protein interactions.562 Ankyrin proteins also affect proinflammatory cytokine expression and the downregulation of cell cycle regulators. Ehrlichiae can be released to infect new cells, by host cell membrane rupture at a late stage of morulae formation.846a Like other ehrlichiae, E. chaffeensis lacks enzymes for the biosynthesis of peptidoglycans and lipopolysaccharide (LPS) that provide strength to the outer membrane.504 The absence of LPS and peptidoglycans also has important implications for the infection and survival of ehrlichial organisms in both tick and mammalian hosts. The tick’s immune system is competent in responding to the presence of LPS, and therefore the absence of LPS in ehrlichial organisms gives them a survival benefit. In the mammalian host, macrophages or neutrophils use pattern-recognition receptors, such as Toll-like receptors, to bind molecules with conserved pathogen-associated molecular patterns such as LPS or peptidoglycan. This reaction elicits an intense innate immune response intended to eliminate offending microorganisms; therefore, the absence of LPS and peptidoglycan gives E. chaffeensis, and other closely related rickettsial organisms, added advantage in intraleukocyte survival. The incubation period is followed by three consecutive stages: acute, subclinical, and chronic. The acute phase may last 1 to 4 weeks. Most dogs recover from the acute disease with adequate treatment. Untreated dogs and those treated inappropriately may recover clinically but then enter the subclinical phase, where only platelet counts may be subnormal.356,890 Dogs in this phase may remain persistent “clinically healthy” carriers of E. canis for months and even years as described in a 3-year follow-up study.356 Results of this experimental infection indicate that the spleen is most likely to harbor E. canis organisms during the subclinical phase of CME and is probably the last organ to accommodate the rickettsia before its elimination. Furthermore, in natural infections, detection of morulae was more likely from splenic aspirations than from buffy coat smears of peripheral blood.276 The spleen is believed to play an important role in the pathogenesis and resultant clinical expression of the disease. Splenectomized dogs experimentally infected with E. canis were shown to undergo a mild clinical illness compared with more severe illness in nonsplenectomized dogs.363 Persistently infected dogs may recover from the disease spontaneously, or may develop chronic severe disease. Not all dogs progress to the chronic phase of CME, and the conditions leading to the development of this phase remain unclear. Comparison of 16S rRNA from ehrlichiae isolated from dogs in the acute and chronic phases of the disease did not reveal any differences, indicating that strain differences of the ehrlichiae were not responsible for the different clinical syndromes seen in dogs.783 However, because the 16S rRNA is a conserved gene, the comparison of other genes should be commenced. Severe pancytopenia, which occurs as a result of bone marrow hypoplasia, is a typical finding of the chronic disease. Significantly lower leukocyte and platelet counts and hematocrit were found to be a high risk for mortality. Severe leukopenia, severe anemia, prolonged activated partial thromboplastin time (aPTT) and hypokalemia have each found to predict mortality with a probability of 100%.781 Death may occur as a consequence of hemorrhages and/or secondary infections. Hematologic changes in CME are associated with inflammatory and immune processes triggered by the infection. Thrombocytopenia, the most common hematologic abnormality of dogs infected with E. canis, occurs in all phases of disease.359,365,365 The greater the magnitude of the thrombocytopenia, the greater the possibility for detection of E. canis 16S rRNA in the blood of dogs living in endemic regions.131 Various mechanisms are involved in the pathogenesis of thrombocytopenia, which include increased platelet consumption and decreased platelet half-life, probably as a result of splenic sequestration and immune mediated destruction. Platelet-bound and circulating antiplatelet antibodies have been detected in whole blood and serum, respectively, of dogs in the acute phase after artificial and following natural infections with E. canis.893 In addition, a serum cytokine, platelet migration-inhibition factor (PMIF), has been found to exist in dogs with ehrlichiosis, and its level is inversely related to the platelet count.1 Higher levels of PMIF were associated with more virulent strains of E. canis. PMIF inhibits platelet migration and is produced by lymphocytes when they are exposed to infected monocytes. Decreased production of platelets as a result of hypoplastic bone marrow is considered to be the mechanism responsible for the thrombocytopenia in the chronic phase.359 Despite the marrow failure and pancytopenia observed in chronically infected dogs, myelofibrosis does not occur.597 The thrombocytopenia is also accompanied by platelet dysfunction in infected dogs. Platelet dysfunction, in conjunction with the low platelet count, contributes to the hemorrhages seen in CME.360, 365 Anti-platelet antibodies may be involved in the thrombocytopathy in CME by competitive binding to the platelet receptors.360 Most dogs infected with E. canis develop hyperproteinemia due to hypergammaglobulinemia, which is usually polyclonal but may be monoclonal in some dogs.358,912 The role of the circulating E. canis–specific antibody in eliminating intracellular ehrlichial infections is minimal. High E. canis antibody titers do not provide protection when animals are challenged.65,729 Moreover, they may have a detrimental effect on progression of the disease because of their immunopathologic consequences.729 Dogs with monoclonal gammopathy may develop hyperviscosity with associated clinical signs and pathologic lesions (e.g., subretinal hemorrhage and retinal detachment leading to acute blindness). Increasing evidence supports the assumption that hyperimmune mechanisms may be involved in the pathogenesis of CME. Some of these mechanisms have been elucidated. These include extensive plasma cell infiltration of the bone marrow and parenchymal organs; polyclonal hypergammaglobulinemia that does not parallel the magnitude of corresponding specific E. canis–antibody titers; positive Coombs’ and autoagglutination test results; the induction of antiplatelet antibody production after natural and experimental infection; and the presence of circulating immune complexes in dogs naturally and experimentally infected with E. canis.340,352,352 Although some pathologic and clinical manifestations in CME appear to be immune-complex mediated, antinuclear antibodies have not been found in dogs naturally or experimentally infected with E. canis.352 Anti–E. canis IgG antibodies generally appear about 15 days after experimental infection.891 IgG2 antibody reaction to E. canis is the principal response in all phases of the CME.364,566 It has been proposed that isotype switching to IgG2 subclass antibodies in dogs is associated with a T-helper type 1 response and a corresponding production of interferon (IFN)-γ.566 This proposition has been strengthened by the finding of persistent expression of IFN-γ and tumor necrosis factor (TNF)-α messenger RNA (mRNA) from days 2 to 8 after infection of dogs with the Oklahoma strain of E. canis and continuing to day 56 postinoculation (PI).832 Furthermore, IFN-γ and TNF-α exert an anti-rickettsial effect via the induction of nitric acid synthesis.280 Apparently, T-cell-induced immunity and IFN-γ secretion play a predominant role in recovery from ehrlichial infections.119,891 IgM antibody response in CME is inconsistent and may not be observed at all in the acute phase of the disease.566 German shepherd dogs were found to be more susceptible to CME than other breeds, displaying a more severe disease with a higher mortality rate. The cellular immune response of German shepherd dogs has been shown to be depressed compared to that of beagle dogs, whereas no significant differences have been recorded in the humoral response between the two breeds.617 This finding supports the major role of cell-mediated immunity during E. canis infection. Persistence of E. canis is achieved by evasion of the host immune system. This occurs through constant alterations of the organism’s surface antigens and the expression of different protein variants. In this regard, proteins with tandem repeats play an important role in the pathogenicity and pathogen-host cell interaction. Twelve proteins containing tandem repeats have been identified in the E. canis genome, and with the exception of three proteins that appear specific to E. canis, all the others have orthologs in other genomes of Rickettsiales.562 Persistent immune evasion may also involve downregulation of major histocompatibility complex class II receptors. In vitro studies in mononuclear cell cultures have indicated downregulated expression.362 This probably leads to impairment of signaling pathways affecting antigen presentation, cell-cell adhesion, cytokine production, humoral reactions, isotype switching, and antirickettsial activity. Survival and multiplication of ehrlichial organisms in infected cells are further enhanced by the organism’s ability to inhibit phagosome-lysosome fusion, as shown for Neorickettsia risticii. Oxytetracycline has been shown to restore the host’s phagocytic ability, probably by inhibiting the synthesis of a bacterial fusion-hindering protein.902 CME is a multisystemic disorder. E. canis infections can be acute, subclinical, or chronic in dogs. Common clinical signs include depression, lethargy, anorexia, weight loss, and hemorrhagic tendencies. The bleeding is usually exhibited by dermal petechiae or ecchymoses, or both (Figs. 26-4 and 26-5). Epistaxis is frequently noticed in CME (Fig. 26-6). Physical examination may also reveal lymphadenomegaly and splenomegaly in 20% and 25% of patients, respectively.925 Because the organism is transmitted by R. sanguineus, the disease may be complicated by co-infection with other pathogens such as Babesia canis vogeli and Hepatozoon canis transmitted by the same vector.306,638,638 Dogs may show changes in eye color or appearance or may develop blindness due to paraproteinemia, systemic hypertension, hyphema, subretinal bleeding, and retinal detachment (Figs. 26-7 through 26-9).355 Anterior uveitis and retinal disease, such as chorioretinitis, papilledema, retinal hemorrhage, retinal perivascular infiltrates, and bullous retinal detachment, are the more common findings in CME.457,643 The neurologic manifestations of ehrlichiosis are primarily the result of meningitis or meningeal bleeding, or both, and result from damage to the adjacent central nervous system (CNS) or peripheral nervous system tissues.643 Seizures, stupor, ataxia with upper or lower motor neuron dysfunction, acute central or peripheral vestibular dysfunction, anisocoria, cerebellar dysfunction, intentional tremors, and generalized or localized hyperesthesia have all been observed. In rare cases, morulae have also been found in cells present in cerebrospinal fluid (CSF).577 Ticks can harbor multiple pathogenic organisms, resulting in co-infections in the infected dog.186,306,306 Therefore, positive results for antibody reactivity or nucleic acid detection methods should be interpreted with caution with regard to defining E. canis as the sole cause of a clinical illness unless other potential tick-borne pathogens have been ruled out. In addition, dogs with immune aberrations resulting from ehrlichiosis can become secondarily infected with opportunistic bacterial, fungal, or protozoal diseases.236 Evidence exists that ehrlichiosis could be a risk factor for myocardial injury, as has been suggested by the finding of increased levels of cardiac tropinin 1 in dogs with acute E. canis infection.230 In one case, apparent electrocardiographic changes were detected in association with E. canis infection.894 The diagnosis of ehrlichiosis is usually based on a combination of anamnesis (i.e., living in endemic area, travel history, tick infestation), clinical signs, hematologic abnormalities, and serologic findings. Polymerase chain reaction (PCR) is now being used in the clinical setting to obtain a diagnosis (refer to Molecular Genetic Detection section in this chapter). Because E. canis is transmitted by R. sanguineus, co-infection with other tick-borne pathogens transmitted by this same tick vector should always be investigated.418 In the acute phase, typical hematologic abnormalities include a characteristic moderate to severe thrombocytopenia, a mild anemia, and leukopenia. The thrombocytopenia is the hallmark of the acute stage and is characterized by megathrombocytosis.892 The anemia seen during the acute phase of CME is considered as “anemia of inflammatory disease” and is classically a mild to moderate normocytic, normochromic and nonregenerative.888 In chronically infected dogs, hemosiderin deposition in the marrow is reduced,597 suggesting that iron deficiency associated with chronic blood loss may be involved in the anemia, rather than it being completely a function of inflammatory sequestration or myeloid failure. Dogs recovering from the acute disease, either those that have not received treatment or those receiving incomplete or inadequate treatment, may enter the subclinical phase. The most pronounced hematologic finding comparing preinfected dogs to subclinical CME dogs is the presence of thrombocytopenia in the latter. In one study, 8 of 9 dogs examined were mildly thrombocytopenic (fewer than 200,000 platelets/µL).890 A concomitant increase in mean platelet volume was also seen in all thrombocytopenic dogs. In experimental studies, E. canis–infected dogs that were co-infected with E. platys developed significantly lower platelet counts than dogs with either infection alone.313 Other statistically significant hematologic parameters included a decline in total leukocyte count in 7 of 9 dogs, a decline in absolute neutrophil count in 5 of 9 dogs, a decline in the hematocrit and hemoglobin concentration in 3 of 9 dogs, a decline in the mean corpuscular volume in 5 of 9 dogs, and an increase in the mean corpuscular hemoglobin concentration in 4 of the dogs tested. The chronic severe form of CME is characterized by bone marrow hypoplasia and impairment of all bone marrow cells, thus resulting in pancytopenia. The prognosis for dogs in the chronic severe form is grave. Hematologic and resultant clinical alterations in these dogs do not resolve with doxycycline treatment.354 The severe chronic phase occurs more often in German shepherd dogs,925 and potentially in spitz hound dogs.781 Granular lymphocytosis has been recognized as an occasional feature of E. canis infection.370 Affected dogs had absolute lymphocyte counts ranging from 5200 to 17,200 cells/µL with cytoplasmic granularity typical of well-differentiated lymphocytic leukemia. Some of these dogs also had monoclonal gammopathies, which may further lead to a misdiagnosis of lymphocytic leukemia. Therefore, ehrlichial testing should be performed on dogs that possess a well-differentiated lymphocytosis on complete blood (cell) count (CBC) results. The most frequent serum chemistry abnormalities include hyperproteinemia, hyperglobulinemia, hypoalbuminemia, and elevated alanine aminotransferase and alkaline phosphatase activities.353,600,600 The hyperproteinemia results from elevated globulin levels, but no direct correlation exists between the levels of serum globulins and serum E. canis antibodies. Serum electrophoresis generally shows a polyclonal hyperglobulinemia,358,868 although monoclonal gammopathies may occur. Therefore, an E. canis antibody titer should be measured in all dogs with a monoclonal gammopathy. Dogs with E. canis often have plasmacytosis in the bone marrow or other tissues, and this can be confused with plasma cell myeloma.567 Infected dogs with pancytopenia generally have lower serum γ-globulin concentrations compared with nonpancytopenic dogs.358,868 Other clinicopathologic findings include prolonged bleeding time, pulmonary interstitial radiopacity ranging from a mild linear pattern to marked interstitial infiltration with peribronchial opacities, hematuria, and proteinuria. Experimentally, peak urine protein loss, consisting principally of albumin, is observed 2.5 to 3.5 weeks after inoculation and may persist for a variable time after infection.188,189 During the peak loss, urine protein/creatinine ratios ranged from 4.5 to 23.2 (reference ratio less than 1.0). A corresponding decrease in serum albumin concentrations was noted (mean 2.1 g/dL). CSF analysis in dogs with signs of CNS disease has revealed increased protein concentrations and predominantly a lymphocytic pleocytosis similar to that found in viral infections. Comparable CSF findings have been noted in human monocytotropic ehrlichiosis.702 Prognostic indicators for survival and risk factors for mortality in dogs with CME have been studied. Severe leukopenia, severe anemia, hypokalemia, and prolonged aPTT were found to be predictors of death with a 100% probability. On the other hand, total leukocytes above 5.8 × 103/µL, platelet counts above 89.5 × 103 µL, hematocrit above 33.5%, aPTT below 14.5 seconds, and potassium levels above 4.75 mmol/L were found to be predictors of survival with 100% probability.781 A diagnosis of CME can be made by demonstration of morulae in monocytes in blood smears or macrophages from tissue aspirates such as spleen, lung, and lymph node. The finding of morulae is difficult and time consuming but may be optimized by performing buffy coat smears or examining thin blood smears made from a peripheral capillary bed of the ear margin. Morulae may be visualized within monocytes present in peripheral blood smears, within synovial fluid, or rarely in the CSF. However buffy coat smears have been shown to have a high sensitivity for detection of morulae.598 Platelets, lymphocytic azurophilic granules, lymphoglandular bodies, and phagocytosed nuclear material can all be confused with ehrlichial inclusions. The indirect fluorescent antibody (FA) test is the most widely used and is considered the serologic gold standard test, indicating exposure to E. canis. The antigenic conservation of immunoreactive proteins has been demonstrated in a number of studies showing strong and consistent immunoreactivity of multiple immunoreactive proteins in dogs with E. canis infection diagnosed by indirect FA test.7,564,564 However, infections with other members of the genus Ehrlichia, and sometimes Anaplasma, may cause positive test results. Furthermore, the E. canis indirect FA test has not yet been standardized between veterinary diagnostic laboratories. Therefore, variations in the indirect FA procedure, as well as variations in the quality and quantity of E. canis antigen used to prepare slides, result in significant variability in test results between different laboratories.619 Early diagnosis of E. canis infection during the acute phase followed by proper treatment or natural elimination of the organism by host immune defenses usually results in complete recovery. Suspicion of present or past exposure of dogs to E. canis is based on the finding of serum anti–E. canis IgG-indirect FA test results with titers at or above 40 to at or above 80, depending on the reference laboratories. Interpretation of indirect FA results must take into account the history, clinical signs, and laboratory findings (Table 26-2). Based on studies that have defined the subclinical phase of CME, it is apparent that the presence of IgG antibodies may only be an indication of past exposure to E. canis. The possibility exists that dogs may be manifesting clinical signs of another disease (infectious or noninfectious) and still be infected with and have a titer to E. canis.890 For example, a dog in the subclinical phase of CME suffering from a concurrent unrelated disease or condition may have high E. canis antibodies but yet have no overt signs of CME. This possibility is supported by the results of previous serosurveys that indicate a high percentage of clinically healthy dogs being seroreactive to E. canis.57,102,102 There is also the possibility that a dog may have a concurrent unrelated disease after successful treatment of E. canis, but yet still have persistence of serum antibodies to E. canis.298 In both of these circumstances, it is not possible to differentiate E. canis infection as a cause of the clinical signs or illness from another disease simply by the use of a single E. canis antibody titer. A repeat indirect FA test or PCR testing 1 or 2 weeks later may be helpful in interpretation of serologic results in these circumstances; however, changes in titers are not often seen in this short time interval. TABLE 26-2 Serologic Test Evaluation Errors and Suggested Resolution for Ehrlichia canis Infection in Dogsa
Ehrlichia and Anaplasma Infections
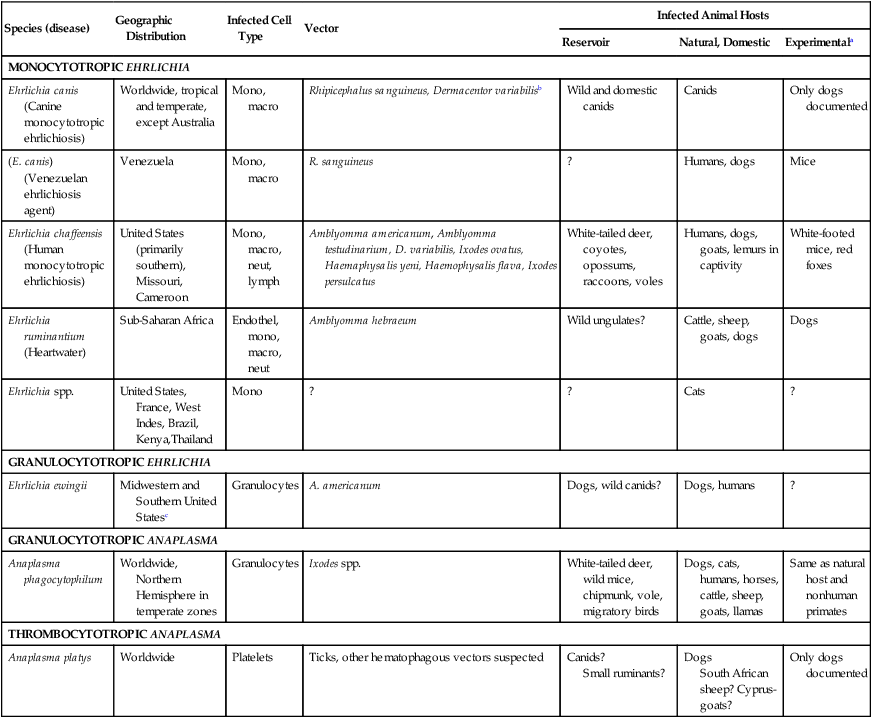
Species (disease)
Geographic Distribution
Infected Cell Type
Vector
Infected Animal Hosts
Reservoir
Natural, Domestic
Experimentala
MONOCYTOTROPIC EHRLICHIA
Ehrlichia canis (Canine monocytotropic ehrlichiosis)
Worldwide, tropical and temperate, except Australia
Mono, macro
Rhipicephalus sanguineus, Dermacentor variabilisb
Wild and domestic canids
Canids
Only dogs documented
(E. canis) (Venezuelan ehrlichiosis agent)
Venezuela
Mono, macro
R. sanguineus
?
Humans, dogs
Mice
Ehrlichia chaffeensis (Human monocytotropic ehrlichiosis)
United States (primarily southern), Missouri, Cameroon
Mono, macro, neut, lymph
Amblyomma americanum, Amblyomma testudinarium, D. variabilis, Ixodes ovatus, Haemaphysalis yeni, Haemophysalis flava, Ixodes persulcatus
White-tailed deer, coyotes, opossums, raccoons, voles
Humans, dogs, goats, lemurs in captivity
White-footed mice, red foxes
Ehrlichia ruminantium (Heartwater)
Sub-Saharan Africa
Endothel, mono, macro, neut
Amblyomma hebraeum
Wild ungulates?
Cattle, sheep, goats, dogs
Dogs
Ehrlichia spp.
United States, France, West Indes, Brazil, Kenya,Thailand
Mono
?
?
Cats
?
GRANULOCYTOTROPIC EHRLICHIA
Ehrlichia ewingii
Midwestern and Southern United Statesc
Granulocytes
A. americanum
Dogs, wild canids?
Dogs, humans
?
GRANULOCYTOTROPIC ANAPLASMA
Anaplasma phagocytophilum
Worldwide, Northern Hemisphere in temperate zones
Granulocytes
Ixodes spp.
White-tailed deer, wild mice, chipmunk, vole, migratory birds
Dogs, cats, humans, horses, cattle, sheep, goats, llamas
Same as natural host and nonhuman primates
THROMBOCYTOTROPIC ANAPLASMA
Anaplasma platys
Worldwide
Platelets
Ticks, other hematophagous vectors suspected
Canids?
Small ruminants?
Dogs
South African sheep? Cyprus-goats?
Only dogs documented
Ehrlichia Canis Infection
Etiology and Epidemiology
Pathogenesis
Clinical Findings
Multisystemic Signs
Ocular Signs
Neuromuscular Signs
Concurrent or Secondary Infections
Cardiologic Signs
Diagnosis
Clinical Laboratory Findings
Cytology
Serologic Testing
Fluorescent Antibody Testing
Test for Ehrlichia canis
Lowest Level Significant Seroreactivity Used
Reasons for False Positive
Reasons for False Negative
Suggested Further Serologic and Genetic Testing Options to Resolve Potential False Results
4-Dx point-of-care serum antibodies dot ELISA
SNAP 4-Dx Plus Test (screening test)
>160
Ehrlichia chaffeensis or Ehrlichia ewingii exposure or infectionb
Previous cleared (past exposure) E. canis
Level of antibody below cutoff of 160
Do indirect FA titer to determine antibody level and for future monitoring titer level after treatment
Do PCR, species specific, or do immunoblot
Microscopic FA serum antibodies to whole-organism antigen (screening test)
Generally ≥64 (varies with lab)
Infection or past exposure to Neorickettsia helminthoeca, E. ewingii, E. chaffeensis, Anaplasma phagocytophilum
Very early infection
Level below cutoff of 64
Titers ≥2048 generally due to E. canis371
Do 4-Dx to help eliminate all but E. chaffeensis
Do PCR, genus-specific, and if positive, then organism-specific
Do PCR if titers remain persistently high after treatment, but need sufficient waiting period or do immunoblot
Immunoblot serum antibodies (confirmatory test)
Distinguish E. canis from E. ewingii by reactivity to separated protein antigens as well as from other rickettsial infections
Common protein antigens among different organisms
Too early sampling, at least 21 days postinoculation required
Do PCR to resolve whether antibody titers represent present or past infection and to help resolve suspected co-infections
Whole blood PCR (confirmatory test)
Determine genetically which species is present. Quantitative (real-time) may allow for monitoring therapeutic effectiveness
Sample contamination, nonspecific amplification
Improper sample extraction; lab technique problems; inappropriate sample selection; low level of infection; sensitivity issue, which seems more frequent in natural vs experimental infections; prior therapy with antibacterial drugs
Do PCR of splenic aspirates
Do real-time (quantitative) PCR to determine organism levels for significance or severity or monitoring effect of therapy?![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree