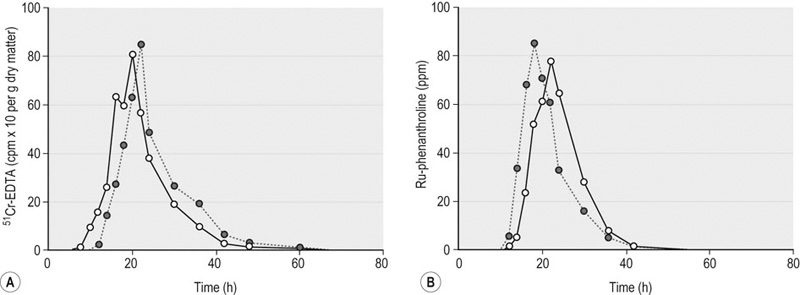
Based upon the design of its gastrointestinal tract (GIT) alone, the horse would be an unlikely animal for humans to choose as their athletic alter ego. The very large cecum and folded large colon, normally filled with ingesta, must certainly not be the easiest of structures to move around. Likewise, the anatomical arrangement and complexity of function of these fermentation vats could, theoretically at least, set the system up for any number of exercise-induced abnormalities. When left to their own devices in the wild, herbivores, whether they be the forestomach fermenters or the hindgut fermenters which include the equidae, spend the large majority of their day quietly grazing or resting.1 Instances of movement faster than a walk are usually brief and in response to attack by a predator or a rival. ‘Domesticated’ horses, on the other hand, are often subject to many situations that result in a greater percentage of their level of daily activity being much more vigorous than just walking and standing around grazing. In human medicine, physical activity is defined as any bodily movement produced by skeletal muscles that results in energy expenditure, whereas exercise is a subset of physical activity that is planned, structured and repetitive with a final objective of maintenance or improvement of physical fitness.2 While hard data concerning the effect of any form of exertion on the equine GIT, whether it be physical activity or exercise, are scarce, we have some results showing that even the anticipation of an upcoming training session on a treadmill, such as turning on the treadmill motor, will evoke an increase in intra-abdominal pressure, reflecting tensing of the abdominal muscles.3 One outstanding effect this pressure increase, and that associated with subsequent trotting or galloping, has on the stomach, for example, will be discussed later in this chapter. The point is that the intra-abdominal pressure response could have far-reaching effects on abdominal viscera, and that the character of the response could vary depending upon the type and duration of the exertion. Galloping may have a different effect than trotting, while trotting may have a different effect than pacing. Jumping may have entirely unique consequences. Effects could include distortion and/or displacement of viscera, redirection of blood flow, and release of various regulatory peptides. A study of human subjects that were ‘encouraged to perform at near-maximal effort’ indicated that runners have more complaints of gastroesophageal reflux (GERD) than do cyclists.4 Could this be because, in the bent over position in which they ride, cyclists create less abdominal muscle press than do runners? Or is it because, as one report has suggested, the physical displacement of the abdominal viscera associated with the ‘pounding of the pavement’ results in an irritation that produces symptoms – the so-called ‘cecal slap syndrome’?5 Or, could it be a question of experimental methodology, since two subsequent studies found that subjects asked to perform at up to 90% VO2max on a stationary bicycle experienced gastroesophageal reflux that was exercise-intensity dependent?6,7 Then, another study found that GERD symptoms and reflux were even more prevalent in weight lifters than either runners or cyclists.8 that is, type, duration and intensity of exercise all need to be considered from the standpoint of potentially differential effects on the gut. Finally, considering all of the things humans ask horses to do in the form of exertion, are some forms more stressful than others? In general, existent data indicate a direct relationship between plasma ACTH and cortisol concentrations and intensity and duration of exercise, respectively, in horses.9–11 But in one study, for instance, galloping caused no significant increase in pituitary venous blood corticotropin-releasing factor (CRF), although ACTH concentration did increase significantly.12 These issues must also be factored in with respect to the potential impact on the GIT. This chapter considers the effects of exercise on: Some fermentation of soluble carbohydrate occurs within the stomach, with subsequent production of lactic and volatile fatty acids (VFAs).13 Whether and to what extent these products are absorbed across the gastric wall and into the bloodstream is not known, although a large proportion should be in the non-ionized, diffusable form, given the acidic environment and they may have some ulcerogenic effects on the non-glandular (squamous) mucosa.14 Pancreatic secretion, on the other hand, while being large in volume, is relatively low in amylase and protease activity.15 The usual complement of small intestinal (brush border) disaccharide enzymes is present in horses, but small intestinal proteolytic enzymes have not been measured.16 From a comparative perspective, the one thing that makes horses unique with respect to other domestic species is the degree of cecal and colonic digestion of dietary fiber; up to 35% of daily caloric needs may be derived from the volatile fatty acids produced from the fermentation of dietary fiber.17 As indicated above, one of the most unique aspects of equine gut function is the fermentation of carbohydrates at both ends of the tract. While we still know little about where and how the products of gastric fermentation are absorbed, we do know that absorption of VFAs produced in the cecum and colon requires mucosal Na+/H+ exchange.18 A mole of water follows each mole of VFA to be absorbed. Furthermore, aldosterone plays an important role in determining the amount of Na, and the water that follows it, that is resorbed by the lower parts of the colon. It follows that disruption of any of the mechanisms involved in support of normal mucosal function, because of an exercise-related event for example, could have major consequences on water and electrolyte balance, and nutrition. In addition, under conditions where the cecum and colons are presented with a large amount of fermentable carbohydrate over a short period of time, the production rate of the VFAs may initially outstrip absorption and, through the osmotic gradient created, pull water from plasma into the colonic lumen.19 Gastric acid secretion occurs in the horse even under fasting conditions.20 The important role played by acid in gastric squamous mucosal ulcerogenesis has been demonstrated, especially as related to the high incidence of this form of gastric ulcer disease in horses under intensive training conditions.3,21–23 This association probably provides one of the clearest representations to date of how exertion can affect the equine GIT (see below). At least when the stomach is empty, small intestinal contents, made up primarily of pancreatic juice, periodically reflux into the stomach, providing some buffering of the acid present.24 Effects of exercise, if any, on this process still need to be investigated. Finally, the horse undoubtedly possesses all the intestinal secretory mechanisms that have been described in other species, but to what degree needs to be defined. What must be remembered is that, in contrast to humans and animals with relatively simple colons, a hypersecretion of strictly small intestinal origin in the horse would not manifest as diarrhea because of the ability, from a water balance perspective, of the ceco-colon to compensate for that malfunction.25 Thus, diarrhea in adult horses is indicative of a large bowel dysfunction, which could include hypersecretion at this level. As an extension of the human experience,26 horse trainers often try to schedule the training sessions for a time when they think the horse has a relatively empty stomach. Humans in general do not feel that they can perform maximally on a ‘full stomach.’ It has been shown that pedaling exercise slows gastric emptying rate in humans, although more runners than cyclists complain of GI problems.4,27–29 Effects of exercise on gastric motility and emptying function in horses are not known as they may have important implications regarding squamous mucosal ulcerogenesis, among other things. As in other species, the proximal stomach relaxes in response to active ingestion (receptive relaxation), and the degree of this relaxation is directly related to the amount of feed ingested.30 A more sustained, but less profound, post-ingestion proximal relaxation (accommodation) then occurs, along with peristaltic-like contractions that begin in the middle of the gastric body and traverse through the antrum and promote emptying of gastric contents into the duodenum. The presence of those contents within the duodenum will, in turn, have modulatory effects on the rate of gastric emptying, the degree of which will depend upon their specific composition.31 This complex regulatory process could be affected by exertion. As indicated earlier, intragastric fermentation of ingested soluble carbohydrates occurs to a considerable degree in horses and this implies gas production as a result.13 It follows that this gas must also be moved aborally into the duodenum, since horses do not normally eructate. Likewise, the large amount of gas that is generated from hindgut fermentation must be moved aborally, along with the contents, to be expelled via the anus. Thus, any condition that could adversely reduce the delivery of gas and/or contents, such as an exercise-induced dysmotility, could result in notable abdominal discomfort and/or diarrhea. Normal gut function is dependent upon a reasonably intact barrier between luminal contents and the submucosa.32–34 Maintenance of barrier integrity is dependent upon numerous factors that include blood flow, mucosal cell turnover rate and immunological tolerance to luminal antigens. Methods for experimentally evaluating the ‘leakiness’ of GI mucosa are discussed below. From the few in vivo and in vitro studies done to date, it appears that horses probably will not differ fundamentally from other species with respect to regional mucosal barrier characteristics, although this needs much more investigation.35–40 Irrespective of species, normal liver function is critical to good health, and this is highly dependent upon adequate blood supply from both arterial and portal venous sources. Blood supply is emphasized here because it is probably the aspect of liver function most prone to modification by exercise. From a species-specific perspective, it should also be kept in mind that certain detoxifying and conjugative functions of the equine liver might be prone to modulation by fasting and fever, as is the case for endogenously generated bilirubin.41 Gastrointestinal transit time can be measured by the use of indigestible, non-absorbable markers. The rate of passage is normally expressed as mean retention time (amount of marker excreted at a certain time after administration of the marker), cumulative excretion or % recovery rate. Comparison of rate of digesta passage during rest and light to moderate exercise has been evaluated in horses and donkeys. Some of the markers used in these studies were cobalt-EDTA, for liquid phase, chromium-mordanted fiber, for solid phase, and ruthenium-phenanthroline and ytterbium chloride, for particulates passage.42–44 A recent review of the subject found that published values for mean retention time (MRT) for digesta passage through the equine GIT ranged from 18 to 60 hours, depending upon many factors, including type of food and marker used, with most of the variability most likely occurring within the large intestine.45 Non-absorbable markers, such as phenol red in man,27,46,47 and horse,48 and polyethylene glycol in dog,49 have been used to measure the effect of different levels of exercise on gastric emptying (GE) of liquid meals. After ingestion of a labeled meal, gastric contents are aspirated through a nasogastric tube or recovered by drainage through a gastric cannula. The amount of marker remaining in the stomach after exercise will depend on the rate of GE of the labeled meal. Sosa et al. reported that GE of a single dose of isotonic fluid administered after intense exercise (70% VO2max) in the horse did not differ from rest, which suggests that GE is not a limitation for rehydration. However, as discussed by the authors of this study, the results were highly variable, suggesting high measurement error due to the technique.48 Apparent digestibility is calculated as the difference between specific components of the diet and those found in feces, expressed as percent utilization. Changes in efficiency of dietary fiber utilization in response to exercise may reflect changes in the passage rate of digesta, the activity or population of microbes within the gut, or mucosal integrity.50 The effect of exercise on digestibility has been studied in horses with inconsistent results. Pearson and Merritt studied the effect of a 14-km walk in donkeys, and neither GI transit nor the digestibility of hay was affected.42 On the other hand, Pagan et al. showed that 8 km of trotting and galloping by a group of Thoroughbreds resulted in a small, but significant decrease in dry matter digestibility, whereas transit rates of both a forage diet and a forage/grain diet were increased at 24 hours after the exercise.43 Orton et al. studied the effect of 12-km daily trotting in yearling horses and recorded an increase in apparent digestibility of dry matter transit rate of particulates, but decreased transit rate of the fluid phase44 (Fig. 46.1). Similarly, Goachet et al. saw an increased apparent digestibility in Arabian horses after endurance training (Table 46.1)51. As Goachet et al. point out, experimental conditions, particularly exercise regimen during measurements, may explain these contradictions as they can cause misconception between short-term effects of exercise and long-term adaptations through physical activity. Clearly, many more studies need to be done on this subject. Table 46.1 Digestibility measurements performed in Arabian horses at the control level (P01) and after conditioning periods corresponding to incremental endurance racing levels at 60 and 90 km. During the measurement periods, feed intake and diet composition were similar Means in rows with different superscripts are significantly different (P < 0.05). bwt, Bodyweight; BCS, body condition score; DMd, dry matter total tract apparent digestibility; OMd, organic matter total tract apparent digestibility; NDFd, neutral detergent fiber total tract apparent digestibility. Adapted from Goachet A-G, Varloud M, Philippeau C et al. Long-term effects of endurance training on total tract apparent digestibility, total mean retention time and faecal microbial ecosystem in competing Arabian horses. Eq Vet J 2010; 42, Suppl 38:387-392, with permission from John Wiley and Sons. The electronic barostat has been extensively used to document gastrointestinal motility in many species, including horses.3,52–54 Unlike other methods, this system is more useful for recording changes in smooth muscle tone than active, phasic contractions. The principle of the barostat is to maintain a constant pressure within a plastic bag of infinite compliance, positioned within the lumen of the segment to be studied. When the internal pressure of the organ increases for any reason (e.g. contraction, increased mural tone), the barostat aspirates air from the bag to maintain the intra-bag pressure constant. Conversely, when the internal pressure decreases, air is injected into the bag. Therefore, as the bag follows the movement of the visceral walls, changes in bag volume are an indirect measurement of changes in tone of the organ55 (Fig. 46.2). In other instances, changes in bag volume reflect changes in external pressure exerted over the organ containing the bag. As we mentioned earlier, we observed such changes in the proximal stomach of horses under a training session and related them to increased intra-abdominal pressure caused by tensing of the abdominal muscles during exercise.3 Interestingly, we also observed relaxation of the proximal stomach in some horses soon after exercise (data not published). What effect this might have on gastric emptying, for instance, could provide some useful information concerning optimal timing of feeding with respect to a given exercise schedule. Adequate local perfusion is necessary for normal gut tissue activity, which requires oxygen and nutrients supplied by peripheral blood. Exercise leads to blood flow diversion from the gastrointestinal tract to the working skeletal muscles and the skin.56 Decreased perfusion or ischemic damage may result in impairment of normal function of the gastrointestinal tissues, including mechanisms of mucosal protection, membrane secretory and absorptive functions, and motility. The effect of short-term exercise on blood flow of abdominal organs has been measured in the horse, using radionuclide-labeled microspheres.57 Once injected into the general circulation, the distribution of microspheres is proportional to the blood flow during its first transit through the circulation.58,59 In the above-mentioned study by Manohar et al. significant vasoconstriction occurred in all gastrointestinal tract tissues during short-term exercise, which caused blood flow to be diverted away from these vascular beds.57 Additionally, hepatic blood flow can be quantified by IV infusion of a marker, such as, bromsulphalein (BSP). As BSP is highly extracted by the liver, hepatic clearance of BSP is mainly dependent on liver blood flow. Decreases in blood clearance of BSP in horses have been attributed to exercise-induced splanchnic vasoconstriction, leading to decreased portal vein blood flow.60 Continuous pH monitoring of the GIT luminal contents with a self-referencing pH electrode may be useful for tracking the effects of exercise on digestive function. The stomach and distal small colon are accessible without special preparation; other parts of the tract would need to be surgically cannulated. Continuous recording of pH changes within the proximal portion of the stomach has indicated that exposure of the non-glandular (squamous) mucosa to hydrochloric acid is increased during periods when forage ingestion is reduced and during exercise.3,61 (See Fig. 46.3.) To date, there is nothing in the literature that describes pH changes within the small colon in response to exercise. Such data have the potential to provide information about the effect of exercise on mucosal secretion, net electrolyte flux and fermentation activity within the large intestine. Exercise stimulates adrenal secretion and activity of the sympathetic autonomic nervous system. This stimulation is reflected in increased plasma levels of ACTH, catecholamines, cortisol and β-endorphins, which are used as indices of physical stress.9–11,62–64 As the horse’s fitness improves, these hormonal responses to exercise diminish.65 Variations in plasma cortisol are more sensitive to duration of exercise, whereas ACTH and catecholamine levels are more closely correlated with exercise intensity.9,11,66 The effect of variations of stress hormone levels upon equine gastrointestinal function is still unknown. In other species, cortisol can have a deleterious effect on mucosal immune function and epithelial permeability,67 whereas catecholamines have the potential to delay gastric emptying, prolong transit time,68,69 and decrease perfusion of the gastric mucosa.70 Gastrin is a major regulatory peptide of acid secretion, the plasma concentration of which was increased after prolonged exercise in the horse in one study,71 but not in another.72 In a third study, gastrin concentration did not increase after short, sprint exercise, but was higher postprandially in trained horses, compared to non-trained horses.73 Whether this has any significant effect on the pH of gastric contents remains to be determined. Plasma concentrations of inhibitors of gastric acid secretion, such as somatostatin in man74 and vasoactive intestinal polypeptide (VIP) in man75 and horse,71 may increase with exercise. Additionally, somatostatin may favor colonic electrolyte transport and inhibit nutrient absorption.76 Potential exercise-induced changes in fluid absorption and secretion within the gut may reflect changes in expression of some regulatory hormones, and vice versa. Exercise increases plasma levels of secretagogues, such as VIP and glucagon, in the horse,71 and secretin, gastric inhibitory polypeptide and prostaglandins in man.74,77,78 On the other hand, atrial natriuretic peptide and aldosterone, which favor sodium absorption, also increase during prolonged exercise in horses.63,79 Agents such as corticotropin-factor (CRF), motilin,80 neuropeptide Y81 and prostaglandins78 can affect motility and are released during endurance-type exercise in man and the same could be expected in horses.10 For instance, activation of CRF signaling pathways mediates both the inhibition of upper gastrointestinal (GI) and the stimulation of lower GI motor function through interaction with different CRF receptor subtypes.82 The effect of exercise on GI motility can, therefore, be indirectly studied by measuring endogenous expression of hormones that control the motility. Finally, alterations of peptide expression may result from a decrease in GIT blood flow, leading to decreased hepatic and renal clearance.56 VIP71 and arginine vasopressin12 in horses, and endothelin-183 and angiotensin II84 in other species, are released during exercise and act as vasoconstrictors. The resulting decrease in blood flow may affect function and regulation of the GIT; for instance, mucosal integrity is compromised by splanchnic ischemia.85 In contrast to the phenol red technique mentioned above, barium contrast radiography86 and scintigraphy87
Effects of exercise on gastrointestinal function
Introduction
Digestion
Absorption
Secretion
Motility
Maintenance of mucosal barrier integrity
Liver specific functions
Available methodologies to document the effects of exercise on the equine gastrointestinal system
Methods that have been used in horses
Transit markers
Apparent digestibility of diet
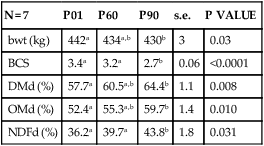
N = 7
P01
P60
P90
s.e.
P VALUE
bwt (kg)
442a
434a,b
430b
3
0.03
BCS
3.4a
3.2a
2.7b
0.06
<0.0001
DMd (%)
57.7a
60.5a,b
64.4b
1.1
0.008
OMd (%)
52.4a
55.3a,b
59.7b
1.4
0.010
NDFd (%)
36.2a
39.7a
43.8b
1.8
0.031
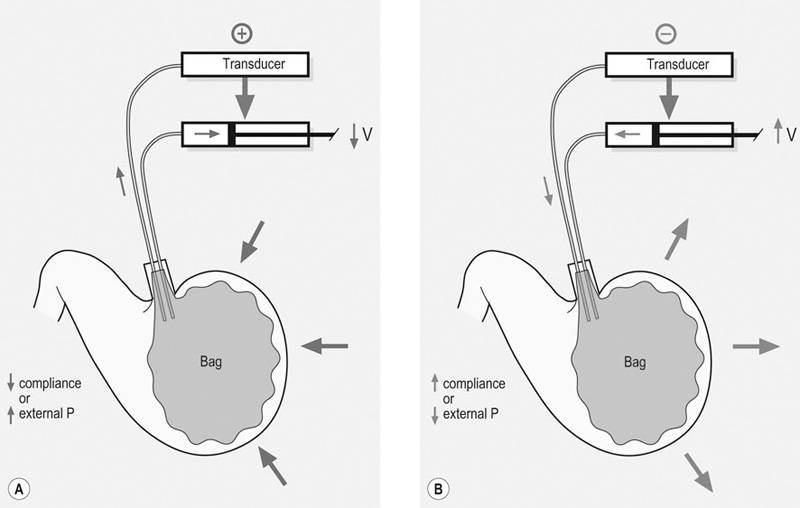
Barostat
Blood flow measurement
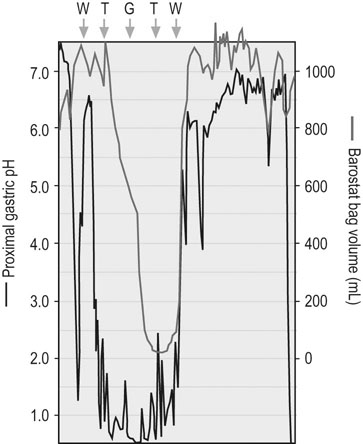
Continuous pH monitoring
Measurement of GI regulatory peptides and steroids
Methods that could potentially be useful in horses, based upon experience in other species
Gastric motility and emptying
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Effects of exercise on gastrointestinal function
