Chapter 13 Dysrhythmias
assessment and medical management
INTRODUCTION
Cardiac dysrhythmias occur frequently in horses and can be associated with a wide range of cardiac and noncardiac diseases. They can be associated with valvular disease,1 congenital defects, pericardial disease and primary myocardial pathology,2 for example, myocarditis,3 myocardial fibrosis,4 myocardial ischaemia,5 cardiomyopathy,6 ionophore toxicosis,7–9 myocardial trauma or myocardial neoplasia.10 However, more commonly, horses develop dysrhythmias in association with hypoxia, metabolic acidosis and electrolyte disturbances,11,12 alterations in local or circulating catecholamine concentrations and autonomic tone,13 septicaemia,14 endotoxaemia,15,16 or various drugs including some that are used to treat dysrhythmias such as quinidine and digoxin,17,18 and the reader is referred to Chapter 6 for a detailed description of arrhythmogenesis.
IDENTIFICATION AND ASSESSMENT OF DYSRHYTHMIAS
An ECG must be performed to definitively diagnose any dysrhythmia (see Chapter 6). The normal components of the surface ECG are:
• P wave: generated by atrial depolarization
• PR segment: representing the duration of atrioventricular (AV) conduction
• QRS complex: produced by ventricular depolarization
• ST segment: representing the duration of the ventricular refractory period
Thus, the surface ECG provides a “road map” of electrical events within the heart and allows the origin of any abnormalities to be identified. A variety of options for lead placement are available (Table 13.1). However, a base-apex lead is often the only lead needed to accurately diagnose the rhythm disturbance present, because this lead produces large, easy to read complexes and is usually well tolerated by the horse (see Chapter 6). It is conventional to display the base-apex lead with negative QRS complexes; however, some clinicians, particularly in the UK, prefer to invert the leads to produce positive QRS complexes. Either is appropriate and the choice is dictated by personal preference. The base-apex lead is the most convenient monitoring lead for critically ill patients, for example, during antidysrhythmic therapy or while performing a pericardiocentesis or other invasive procedure. Occasionally it may be difficult to determine if a rhythm disturbance is of ventricular or supraventricular origin from just one ECG lead, particularly with supraventricular dysrhythmias. In these horses at least two different leads are needed to determine the origin of the abnormal depolarization (see Table 13.1).
Table 13.1 Electrode placement for complete 12-lead electrocardiogram
| Lead 1: LA–RA | Left foreleg (left arm) electrode placed just below the point of the elbow on the back of the left forearm – right foreleg (right arm) electrode placed just below the point of the elbow on the back of the right forearm |
| Lead II: LL–LA | Left hindleg (left leg) electrode placed on the loose skin at the left stifle in the region of the patella – left foreleg (left arm) electrode placed just below the point of the elbow on the back of the left forearm |
| Lead III: LL–RA | Left hindleg (left leg) electrode placed on the loose skin at the left stifle in the region of the patella – right foreleg (right arm) electrode placed just below the point of the elbow on the back of the right forearm |
| aVr: RA–CT | Right foreleg (right arm) electrode placed just below the point of the elbow on the back of the right forearm – the electrical centre of the heart or central terminal × 3/2 |
| aVl: LA–CT | Left foreleg (left arm) electrode placed just below the point of the elbow on the back of the left forearm – the electrical centre of the heart or central terminal × 3/2 |
| aVf: LL-CT | Left hindleg (left leg) electrode placed on the loose skin at the left stifle in the region of the patella – the electrical centre of the heart or central terminal × 3/2 |
| CV6LL: Vl–CT | Vl electrode placed in the 6th intercostal space on the left side of the thorax along a line parallel to the level of the point of the elbow – the electrical centre of the heart (central terminal) |
| CV6LU: V2–CT | V2 electrode placed in the 6th intercostal space on the left side of the thorax along a line parallel to the level of the point of the shoulder – the electrical centre of the heart (central terminal) |
| V10: V3–CT | V3 electrode placed over the dorsal thoracic spine of T7 at the withers – electrical centre of the heart. The dorsal spine of T7 is located on a line encircling the chest in the 6th intercostal space (central terminal) |
| CV6RL: V4–CT | V4 electrode placed in the 6th intercostal space on the right side of the thorax along a line parallel to the level of the point of the elbow – the electrical centre of the heart (central terminal) |
| CV6RL: V5–CT | V5 electrode placed in the 6th intercostal space on the right side of the thorax along a line parallel to the level of the point of the shoulder – the electrical centre of the heart (central terminal) |
| Base-apex: LA–RA | Left foreleg (left arm) electrode placed in the 6th intercostal space on the left side of the thorax along a line parallel to the level of the point of the elbow – right foreleg (right arm) electrode placed on the top of the right scapular spine |
Echocardiography is used to identify valvular disease, pericarditis congenital defects and myocardial pathology although a normal echocardiogram does rule out the presence of myocardial disease (see Chapter 9). Assays of serum cardiac troponin I and other biomarkers (see Chapter 12) may also provide evidence of myocardial disease while haematology, blood biochemistry, serum electrolyte and blood gas analysis, and other laboratory assessments such as blood culture, virus isolation and serology are also helpful in identifying possible contributing factors in horses presenting with dysrhythmias.
CLASSIFICATION OF DYSRHYTHMIAS
There are several different classifications of rhythm disturbances. From a clinical perspective, it is useful to categorize cardiac dysrhythmias by their site of origin (supraventricular or ventricular) and by their rate (bradydysrhythmias or tachydysrhythmias)19–21 as these factors determine their clinical significance and most appropriate management. In horses, most bradydysrhythmias are physiological and associated with high vagal (parasympathetic) tone while most tachydysrhythmias are abnormal. Profound bradydysrhythmias and rapid tachydysrhythmias, particularly those of ventricular origin, are the rhythm disturbances most likely to need immediate treatment to control the ventricular rate and relieve the clinical signs of cardiovascular collapse.
Bradydysrhythmias
Bradydysrhythmias are commonly detected in healthy horses and are associated with high parasympathetic tone. Second-degree atrioventricular (AV) block is the most common bradydysrhythmia detected in resting horses. Sinus arrhythmia, sinus bradycardia, sinoatrial (SA) block and SA arrest also occur in normal horses with high vagal tone.20,22 These normal dysrhythmias are present at rest and should disappear with a decrease in parasympathetic tone and/or an increase in sympathetic tone such as occurs with excitement or exercise.
Advanced second-degree AV block
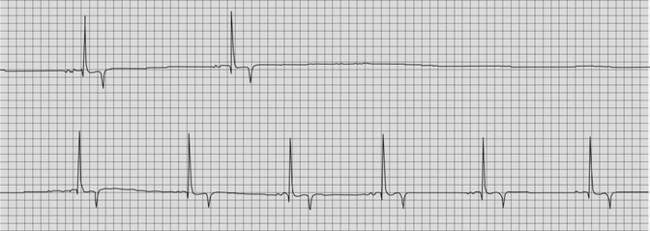
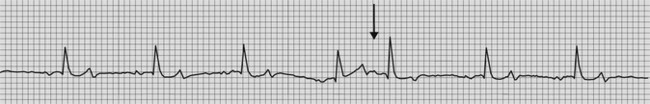
Advanced second-degree AV block, rare in horses, is a pathological form of conduction block at the AV node and can be caused by electrolyte imbalances, digitalis toxicity and AV nodal disease (inflammatory or degenerative).2,20 Horses with advanced second-degree AV block usually have severe exercise intolerance and may collapse. A complete cardiovascular examination, biochemistry screen and complete blood count should be performed on all horses presenting with advanced second-degree AV block to attempt to determine the underlying cause of the dysrhythmia. Affected horses have a slow heart rate (usually 8–24 bpm). On auscultation S1 and S2 is regularly spaced with an audible S4 preceding each S1. S4 is heard in the diastolic pauses with one, or more, S4 for each period of second-degree AV block.
The ECG reveals a slow heart rate with a regular R–R interval between episodes of AV block and normally configured QRS complexes. The QRS complexes are each preceded by a P wave at a normal to near normal P–R interval, evidence of AV conduction. The P–R interval may be slightly prolonged as first-degree AV block may also be present. The P–P interval is regular and the atrial rate is rapid with numerous (two or more) P waves not followed by QRS complexes (Fig. 13.1). Appropriate treatment should be instituted as soon as possible, based upon the probable aetiology of the dysrhythmia to hopefully prevent the progression of the conduction block to complete AV block (see treatment of complete heart block below).
Complete (third-degree) AV block
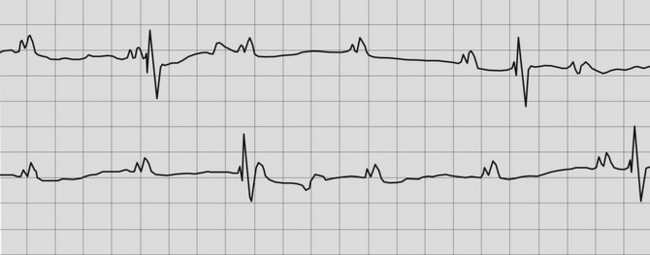
Complete heart block is rare in horses and is usually associated with inflammatory or degenerative changes in the AV node,23 although it may occur with electrolyte imbalances or other metabolic abnormalities such as is seen with foals with uroperitoneum, particularly when anaesthetized.24,25 Complete heart block has also been observed as a congenital defect26 and associated with mediastinal lymphoma27 and rattlesnake envenomation.28 Horses with third-degree AV block will typically have severe exercise intolerance and frequent syncope. The resting heart rate (ventricular rate) is very slow (usually ≤20 bpm) but regular with a more rapid, independent atrial rate. The heart rate does not increase appropriately with a decrease in vagal tone or an increase in sympathetic tone. The S1 and S2 are usually loud and regularly spaced with more rapid independent S4 (usually ≤60/minute), which are also regularly spaced. Occasional “bruit de cannon” sounds, caused by the summation of S4 with another heart sound (S1, S2 or S3) are detected. An irregular rhythm may be detected in some horses with concurrent paroxysms of ventricular tachycardia.
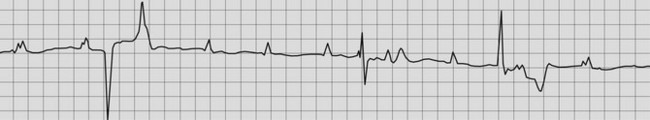
The ECG usually reveals regular P waves that are not followed by QRS complexes (i.e. there is no evidence of AV conduction) and QRS complexes that are widened and bizarre in their appearance because they are originating from an idionodal or idioventricular pacemaker (Fig. 13.2). If the QRS complexes are regularly spaced, they should all look alike because they are originating from the same idionodal or idioventricular pacemaker. These are known as escape complexes because they represent the ventricles’ attempt to escape in the absence of AV conduction. If there is other ventricular ectopy present, there will be more than one QRS configuration and the R–R interval may vary (Fig. 13.3). The P–R intervals will be of varying lengths with no consistent relationship between the P waves and the QRS complexes (see Figs. 13.2, 13.3). The atrial rate is usually very rapid with a regular P–P interval and there are many more P waves than QRS complexes.
Treatment of complete heart block in horses should be initiated as soon as the diagnosis is made. If the underlying cause of the complete heart block cannot be corrected or removed, the definitive treatment for third-degree AV block is a pacemaker (see Chapter 14). Corticosteroids, for example, dexamethasone at 0.05–0.2 mg/kg IV, are usually indicated because the most common treatable cause of complete heart block is inflammatory disease in the region of the AV node. If an active viraemia is suspected, corticosteroids should be used with care. The small risk of the horse developing laminitis associated with prolonged administration of high doses of corticosteroids must also be weighed against the risk of the horse remaining in third-degree AV block, requiring the implantation of a cardiac pacemaker for long-term survival.23,26 As emergency measures, vagolytic drugs, atropine or glycopyrrolate, can be administered (see Table 7.1). If ventricular ectopy is present, sympathomimetic drugs should be used with care, or not at all, because these drugs may exacerbate ventricular dysrhythmias. If no concurrent ventricular ectopy is present sympathomimetic drugs may be effective. Dopamine has β1-adrenergic effects at moderate doses (3–5 µg/kg/minute) and has been effective in treating advanced AV block.25,28 Alternatively, isoproterenol can be administered as continuous rate infusion (CRI; see Table 7.1). However, rapid tachydysrhythmias can occur with the administration of sympathomimetic drugs. If pharmacological intervention is not successful in restoring sinus rhythm, a temporary transvenous pacemaker may be necessary until a permanent transvenous pacemaker can be inserted23,26 (see Chapter 14).
Sinus bradycardia, sinus arrhythmia, SA block and SA arrest( SBR)
SBR)
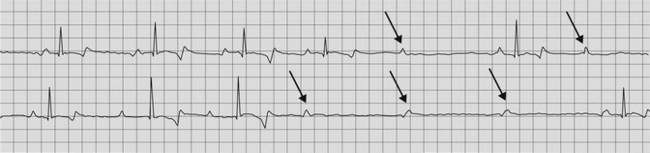
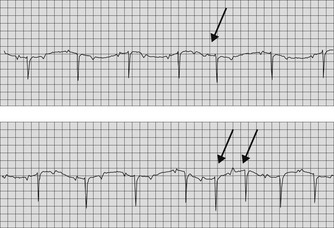
Sinus bradycardia, sinus arrhythmia, SA block and SA arrest occur in normal fit horses associated with high vagal tone but are less common than second-degree AV block (Fig. 13.4).21 These dysrhythmias can occur in combinations: sinus arrhythmia is usually also present in horses with sinus bradycardia, SA block can occur in conjunction with second-degree AV block29 and on resting ambulatory ECG, many normal horses will display a variety of physiological bradydysrhythmias throughout a 24-hour period. Auscultation reveals regular S1 and S2 with a pause in the rhythm (SA block or arrest) or rhythmic variation of diastolic intervals (sinus bradycardia and sinus arrhythmia) with heart rates of 20–30 bpm. An S4 precedes each S1 and there are no isolated S4 in the diastolic pauses representing the period of SA block.
The atrial and ventricular rates are slow or low normal and normal QRS complexes are detected associated with the preceding P waves, evidence of AV conduction. The P–P and R–R intervals are rhythmically irregular with sinus arrhythmia. SA blocks are characterized by pauses of less than or equal to two P–P intervals, whereas sinus arrest is present if SA activity ceases for longer than two P–P intervals. These rhythms are normal manifestations of high vagal tone and disappear with exercise or the administration of a vagolytic drug, for example, atropine or glycopyrrolate (see Table 7.1).
Sick sinus syndrome
Prolonged periods of SA arrest, profound sinus bradycardia or high-grade SA block may be indicative of sinus node disease, termed sick sinus syndrome.30 Ventricular escape rhythms may occur during prolonged pauses. Sinus node disease is rare in horses but inflammatory and degenerative changes must be considered possible aetiologies. Affected horses may have a history of collapse or weakness. These horses should be carefully evaluated with exercising electrocardiography and the response of the horse to vagolytic and sympathomimetic drugs determined. It may be possible to increase the heart rate and abolish the dysrhythmia during exercise tests, although the maximal heart rate may be reduced and the dysrhythmia may recur shortly after cessation of exercise.30 Corticosteroids should be initiated for horses with life-threatening abnormalities of sinus rhythm, in the hope that pacemaker implantation will not be necessary. Definitive treatment of sick sinus syndrome is pacemaker implantation30 (see Chapter 14).
Tachydysrhythmias
Tachydysrhythmias can be defined as either supraventricular or ventricular. Various forms exist including premature depolarizations, tachycardia, fibrillation and pre-excitations syndromes. Isolated supraventricular (SVPD) and ventricular (VPD) tachydysrhythmias are found in low numbers in horses without organic heart disease, particularly in prolonged resting ECG recordings (see Chapter 10), but these “normal” dysrhythmias are less common than second-degree AV block and other bradydysrhythmias. Persistent and frequent tachydysrhythmias are more commonly associated with cardiac and noncardiac pathology. Ambulatory ECG monitoring is very useful in documenting the frequency of dysrhythmic episodes in horses with intermittent tachydysrhythmias (see Chapter 10) and is invaluable in monitoring and assessing the effects of any treatment.
Supraventricular premature depolarizations and tachycardia( AF, SFP)
AF, SFP)
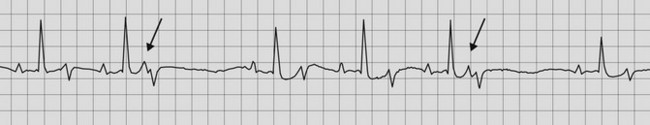
SVPD originate in the atria before SA nodal discharge.21 There is often an underlying regular rhythm and the SVPD may or may not be conducted to the ventricles. On auscultation, beats occurring earlier than normal are detected. Electrocardiography reveals a normally configured QRS–T complex, occurring prematurely. A bizarre P wave may be visible or may be hidden in the preceding T wave (Figs. 13.5 and 13.6). Supraventricular or atrial tachycardia is defined as more than four SVPD occurring in sequence.21 Horses with frequent SVPD or supraventricular tachycardia are often able to maintain a normal ventricular response rate as some of the SVPD are blocked at the level of the AV node (see Fig. 13.6).
Infrequent SVPD can be detected in normal horses. However, frequent SVPD and atrial tachycardia may be indicative of myocardial disease (Fig. 13.7), or occur in association with atrial enlargement due to AV valvular disease or congenital heart disease. If an obvious predisposing cause is identified, treatment should be directed at that. In some horses, SVPD resolve following a period of rest and therapy with corticosteroids2 (see Table 7.1). Specific antidysrhythmic therapy is rarely necessary provided that the ventricular rate is not affected by SVPD. Digoxin31,32 or diltiazem33,34 can be used to control the ventricular rate with rapid supraventricular tachycardia (see Table 7.1). Digoxin has a very narrow therapeutic to toxic range and therefore horses should be monitored for any signs of digoxin toxicity. Serum or plasma samples should be obtained for digoxin concentrations after several days of oral therapy to see if adjustments in the dosage are necessary (see Chapter 7). Phenytoin may also be helpful in suppressing SVPD in some cases (see Table 7.1). Horses with persistent, numerous SVPD, no performance limitations, and minimal identifiable underlying cardiac disease, can continue to be used for ridden activities as it is unlikely that these horses will collapse or represent a danger to a rider. However, numerous SVPD are a risk factor for the development of atrial fibrillation (see below) and thus, the potential for future performance-limiting problems must be considered, particularly when advising potential purchasers.
Atrial fibrillation( AF, VMD)
AF, VMD)
Atrial fibrillation (AF) is a common arrhythmia in horses.35–38 The estimated minimum frequency (i.e. no. of episodes per race starts) of AF in Japanese racehorses is 0.03% with an estimated minimum prevalence (i.e. no. of horses with AF per no. of racehorses) of AF among Japanese racehorses being 0.29%.38 A similar frequency has been reported in the UK.39 In the vast majority of horses that develop AF during racing the dysrhythmia is paroxysmal.38,39 There do not appear to be any gender predispositions but AF is more prevalent in racehorses >4 years of age compared to 2-year-olds. Horses racing on turf were more likely to develop AF than those racing on dirt in one study.38 AF is also very common in large draft breeds40 and Standardbreds have been over-represented compared to hospital populations in some studies.35–37
AF is due to re-entry (see Chapter 6). Shortening of the effective refractory period, atrial inhomogeneity and SVPD set the stage for the development of AF and normal horses are predisposed to the development of AF in the absence of structural heart disease due to their high resting vagal tone and large atrial mass. Most horses with AF have little or no underlying cardiac disease and in this situation, the term “lone AF” is applied. However, AF can also be related to atrial enlargement and horses with congestive heart failure due to congenital or acquired cardiac disease frequently develop AF. Transient potassium depletion, such as can occur with the administration of furosemide or with the loss of large amounts of potassium in the sweat of exercising horses, is also thought to be a predisposing factor in the development of AF.
Presenting complaints for horses with AF include poor performance in horses engaged in athletic activities, tachypnoea, dyspnoea, exercise-induced pulmonary haemorrhage, myopathy and colic. AF can also be an incidental finding during a routine examination, particularly in horses that do not routinely perform vigorous exercise. Horses with lone AF usually have normal resting heart rates (<44 bpm), although the rhythm is irregularly irregular and no S4 is produced. The intensity of the peripheral arterial pulses is also irregularly irregular. Pulse deficits may be present, particularly in horses with two conducted beats occurring in rapid succession. Increased resting heart rates (>50 bpm) and loud cardiac murmurs and signs consistent with congestive heart failure should alert the clinician to the possibility that significant underlying heart disease is present. Clinical signs of left-sided heart failure (pulmonary oedema, coughing, tachypnoea) and/or right-sided heart failure (generalized venous distention, jugular pulsations and peripheral oedema) may be present (see Chapter 16).
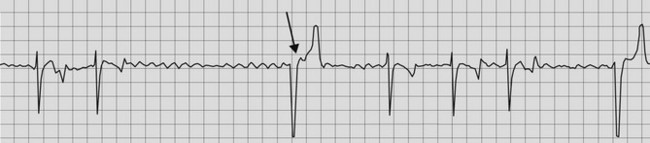
The ECG reveals irregularly irregular R–R intervals, no P waves and normal appearing QRS complexes (Fig. 13.8). Rapid baseline fibrillation “f” waves are usually present, which may be small (fine) or large (coarse). In approximately 10% of horses with AF, QRS complexes originating from the ventricle will also be detected (see Fig. 13.8) while with congestive heart failure, tachycardia is confirmed.
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree