Chapter 11 Diarrhea
Definition
Diarrhea is an increase in the frequency, fluidity, or volume of feces1 that is best characterized by duration (acute versus chronic), pathophysiologic mechanism, and anatomic location. Diarrhea is considered acute if it lasts for less than 14 days, and chronic when it persists for more than 14 days. Acute, self-limiting diarrhea is a relatively common problem in dogs and cats and usually requires minimal diagnostic testing and therapy. In contrast to most animals with acute diarrhea, chronic diarrhea can be particularly challenging to diagnose, because most animals will not respond to empirical therapies, necessitating a well-formulated and cost-effective diagnostic and therapeutic plan. Specific therapeutic modalities are based upon a definitive diagnosis or histologic characterization of intestinal biopsies.
Pathophysiology and Mechanisms
Osmotic Diarrhea
Osmotic diarrhea is caused by unusually large amounts of poorly absorbable osmotically active solutes in the intestinal lumen. Osmotic diarrhea occurs with malabsorptive disorders where nutrients are maldigested or malabsorbed, remain within the intestinal lumen, and osmotically attract water. Exocrine pancreatic insufficiency is an example of an osmotic diarrheal disorder. Retention of nutrients can lead to alterations in intestinal microflora and fermentation of carbohydrates, further increasing numbers of osmotically active particles. The fecal water output in osmotic diarrhea is directly related to fecal output of the solute or solutes that are exerting an osmotic gradient across intestinal mucosa. Electrolyte absorption is unaffected by these osmotically active substances, and fecal water typically contains very little unabsorbed sodium or potassium.2 This is the basis for the calculation of the “fecal osmotic gap.” In this calculation, the difference between luminal osmolality (equal to body fluid osmolality, approximately 290 mOsm/kg, because the colon cannot maintain an osmotic gradient against plasma) and osmolality of luminal contents contributed by fecal electrolytes is estimated. The contribution of fecal electrolytes is calculated as twice the sum of sodium and potassium ions to account for the anions that accompany these cations. A fecal osmotic gap greater than 50 mOsm/kg suggests osmotic diarrhea.2 One of the major hallmarks of osmotic diarrhea is that diarrhea resolves when the patient stops ingesting the poorly absorbable solute.
Secretory Diarrhea
Secretory diarrhea is caused by abnormal ion transport in intestinal epithelial cells. The most common cause of secretory diarrhea in dogs and cats are abnormal mediators resulting in changes in intracellular cyclic adenosine monophosphate (cAMP), cyclic guanosine monophosphate (cGMP), calcium, and/or protein kinases, which in turn cause a decrease in neutral sodium chloride absorption or an increase in chloride secretion.3 Such mediators include endogenous enteric hormones or neuropeptides, inflammatory cell products, bacterial enterotoxins, laxatives, fatty acids, and bile acids (see Chapters 1 and 57 for more detail). Secretory diarrhea has two important distinguishing features; first, fecal osmolality can be accounted for by sodium, potassium, and their accompanying anions, and thus the osmotic gap is small; and second, the diarrhea usually persists despite fasting because the diarrhea is caused by abnormalities in ion transport that have nothing to do with food. Enteropathogenic Escherichia coli and IBD are examples of secretory diarrheas.
Deranged Motility
Experimental studies in dogs show that abnormal ileal and colonic motility patterns may contribute to clinical symptomatology of IBD.4,5 The two major motor abnormalities in intestinal inflammation are suppression of phasic contractions, including migrating motor complexes, and stimulation of giant migrating contractions (GMCs), the powerful ultrapropulsive contractions that usually propagate uninterruptedly form the point of their origin in the small intestine to the terminal ileum and often into the colon.4 Stimulation of GMCs in fasting and fed states produces ultrarapid transit of intestinal and pancreaticobiliary secretions and undigested food into the colon to increase its osmotic load with resultant diarrhea.4 Platelet-activating factor (PAF) may be one of the inflammatory response mediators that stimulates GMCs,5 and it is synthesized and released from several immunocytes, including polymorphonuclear (PMN) leukocytes, monocytes, macrophages, mast cells, and eosinophils.5
Differential Diagnosis
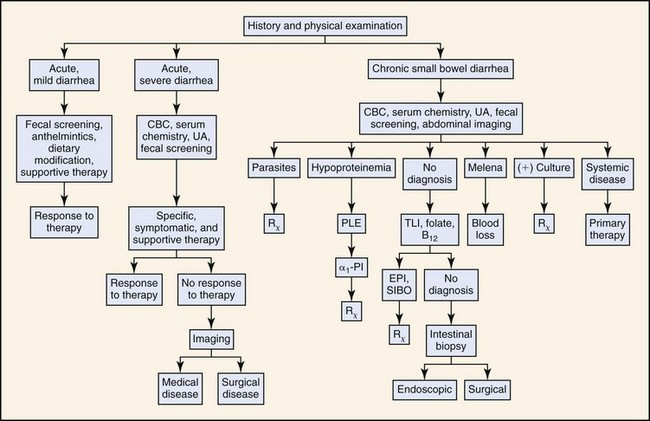
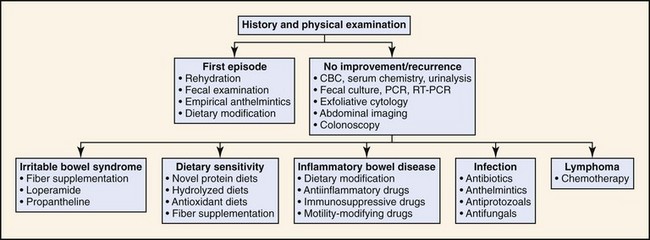
Box 11-1 lists differential diagnoses for acute and chronic diarrhea in dogs and cats, as well as those disorders that are potentially life-threatening. Many cases of chronic diarrhea can manifest initially as acute diarrhea. Localization of the disease process into “small bowel” versus “large bowel” has some limitations, as many diarrheal diseases with primary manifestations of one compartment (large bowel or small bowel) may have diffuse gastrointestinal (GI) involvement (large and small bowel). This point is underscored by a study in 40 dogs suggesting that routine collection of ileal biopsies is warranted in dogs with colonopathy, and that routinely sampling of duodenum and ileum increases the diagnostic yield compared to biopsy of one anatomic site.6 Regardless, the initial differentiation into small and large bowel components helps to further clarify the medical investigation (see Figs. 11-1 and 11-2).
Box 11-1
Differential Diagnoses for Acute and Chronic Diarrhea
Evaluation of the Patient
History
History and physical examination often indicates the anatomic localization and severity of the disease process, and it helps prioritize differential diagnoses. History should ensure that systemic causes of diarrhea are not overlooked. A comprehensive history should also identify important predisposing factors (e.g., exposure to parasites, infectious agents, drugs, toxins). It is equally important to fully characterize the nature of diarrhea and appearance of feces (Table 11-1). For dogs with a history of tenesmus, it is pivotal to determine whether signs are secondary to colitis or to a discrete mass or polyp in the colorectal region. The latter is often associated with a change in the appearance of the stool (“ribbon-like,” “pencil-thin”) in the absence of a marked increase in frequency of bowel movements or increased fecal mucus as seen with colitis. Likewise, absence of clinical signs of diarrhea does not rule out severe underlying intestinal disease; dogs with protein-losing enteropathy (PLE) may have anorexia and weight loss without associated vomiting and diarrhea. Failure to consider the role of diet or dietary supplements in precipitating or alleviating the diarrhea can cause delayed diagnosis or improper dietary recommendations. Box 11-2 outlines specific questions that should be addressed.
Table 11-1 Differentiation of Small Versus Large Bowel Diarrhea*
| Differentiation of small versus large bowel diarrhea based upon clinical signs and the physical appearance of feces. | ||
| Sign | Small Bowel | Large Bowel |
| Frequency of defecation | Normal to mildly increased | Markedly increased |
| Fecal volume | Normal to increased | Decreased |
| Fecal mucus | Usually absent | Often present |
| Fecal blood | Melena | Hematochezia |
| Tenesmus | Absent | Often present |
| Urgency | Absent | Often present |
| Vomiting | May be present | May be present |
| Steatorrhea | May be present | Absent |
| Dyschezia | Absent | Often present |
| Weight loss | Common | Uncommon |
* Caution should be heeded with the oversimplistic attempts at compartmentalizing the diarrhea into a small bowel versus a large bowel compartment, as many diarrheal diseases can have diffuse involvement histologically.
Box 11-2
Questions that Should be Asked in Patients with Diarrhea
• What is the clinical course or onset of the diarrhea (congenital or acquired; abrupt or gradual in onset; continuous or intermittent)?
• What is the duration of signs?
• What are the physical characteristics of the diarrhea?
• Are there any alleviating or exacerbating factors for the diarrhea such as dietary changes, antibiotic administration, stress, recent travel, or recent kenneling?
• What is the animal’s past medical history, and is this diarrhea episode a new problem or a recurrent one? If recurrent, how was the diarrhea managed previously, and what was the outcome?
• What is the animal’s anthelmintic and vaccination history?
• Are any other pets in the household similarly affected?
• Is the diarrhea associated with any other systemic (e.g., polyuria/polydipsia) or gastrointestinal signs such as weight loss, vomiting, and anorexia?
Laboratory Evaluation and Tests
For animals with acute, mild diarrhea that appear relatively healthy on physical exam and are deemed likely to have a self-limiting gastroenteropathy, a minimum database consisting of centrifugation fecal flotation using zinc sulfate (specific gravity of 1.18 to 1.2) complemented with a fecal enzyme-linked immunosorbent assay (ELISA) or immunofluorescence test for Giardia is typically adequate for assessment of parasitic disease. In addition, measurement of hematocrit and total protein are helpful to assess hydration status. Fecal cytology in diarrheic dogs is a low-yield diagnostic test because finding of “safety pin”-shaped endospores consistent with Clostridium perfringens are of no diagnostic value. Their detection does not correlate with the presence of C. perfringens enterotoxin, the putative virulence factor associated with diarrhea.7,8 Likewise, detecting “spiral-shaped” bacteria assuming the appearance of “seagulls” is insufficient for a diagnosis of Campylobacter-associated diarrhea because spiral-shaped bacteria resembling Campylobacter spp. are commonly identified in feces from healthy, nondiarrheic dogs and cats. Isolation of the microaerophilic organism utilizing selective culture media is a far more sensitive diagnostic tool compared with stained fecal smears.9 In contrast to direct fecal cytology, exfoliative rectal cytology is a useful diagnostic test in dogs and cats with signs of colitis, and is best indicated for diagnosis of specific enteropathogens such as Histoplasma spp., Pythium insidiosum, or Prototheca,10 or for colonic neoplasms such as lymphoma and carcinoma.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree