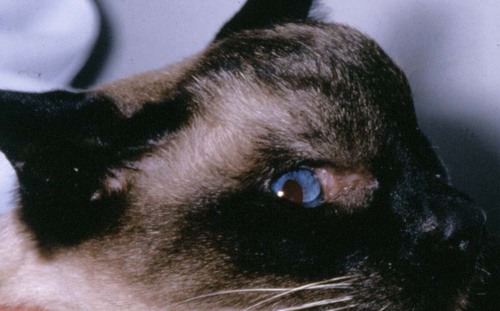
Cryptococcosis is an important fungal infection of humans and animals and the most common systemic mycosis of cats. The infection is thought to be acquired from the environment, and disease transmission between infected subjects has not been reported. Occasional outbreaks of infection in humans and animals, such as in British Columbia, Canada, may result from exposure to common environmental sources.88,140 Because cats are more susceptible to infection than humans, they may represent a sentinel for human exposure.30,31 Infections are almost always present in warm-blooded mammals or birds; however, there have been rare reports of its occurrence in poikilotherms.109 The genus Cryptococcus is now known to contain at least 39 variously encapsulated yeast species, two of which most commonly cause disease—Cryptococcus neoformans and Cryptococcus gattii. C. gattii was formerly known as C. neoformans var. gattii (Web Table 59-1). Cryptococcus bacillisporus is considered an emerging pathogen of immunocompetent humans in temperate regions of North America. Other species, particularly Cryptococcus laurentii and Cryptococcus albidus, rarely cause disease in humans, dogs, or cats70,84 but may be emerging as a result of immunocompromise.80 Cryptococcus magnus was isolated from the external ear canal of a cat with otitis externa,69 and a cat with granulomatous mass involving a thoracic limb,132a and C. laurentii has also been isolated from the external ear canals of dogs.9 The ability of C. neoformans and C. gattii to grow at 37° C may be a factor in their pathogenicity, because other members of the genus grow poorly at this temperature. WEB TABLE 59-1 Major Causative Agents of Cryptococcosis Yeast Form (Cryptococcus spp.)a aOther species of lesser clinical importance include C. laurentii, C. albidus, and C. magnus, see text. C. neoformans and C. gattii are dimorphic, basidiomycetous fungi. The life cycle of the fungus involves vegetative growth as a haploid budding yeast, combined with the ability to undergo a transition to a filamentous form (Filobasidiella species), also known as a teleomorph. The teleomorphs of C. neoformans and C. gattii are F. neoformans and F. bacillisporus, respectively. Two mating types of Cryptococcus spp. exist, α and a, the α-type being vastly more prevalent in environmental and clinical samples.129 Under appropriate conditions and in response to mating pheromones, the two mating types can fuse and adopt a dikaryotic filamentous state, also known as the perfect state. This is followed by production of basidia, which are small, specialized club-shaped structures on which basidiospores form. Cells of the α mating type can also respond to desiccation, nitrogen limitation, and pheromone signals and undergo asexual reproduction via a process known as haploid fruiting, which also involves filamentation and spore formation.46,62,62 This may explain their relative high prevalence. Basidiospores are thought to represent the infectious propagules that give rise to mammalian disease.22a,41,141,153 The mating of Cryptococcus spp. has never been observed in nature, but can be demonstrated under special laboratory conditions and likely occurs in certain natural environments. Unlike other dimorphic fungi, the yeast phase of Cryptococcus is found under routine laboratory conditions and in mammalian host tissues. An unusual temperature-sensitive isolate of C. neoformans, which would not grow at 37° C, produced hyphal elements in a granuloma within the nasal passage of a cat and in culture at 35° C.8 In animal tissues, the yeast form of C. neoformans and C. gattii is round to oval with a variably sized polysaccharide capsule. The capsule provides protection from environmental insults (e.g., desiccation) and the phagocytic response of the host. In tissues, Cryptococcus reproduces by forming one or two daughter cells (buds) that are connected to the parent cell by a narrow isthmus. Buds may break off when small, and thus the cell population varies in size.65 Cryptococcus spp. are distributed worldwide. Various domestic and native mammals may be infected, including cats, dogs, ferrets, horses, camelids, goats, sheep, cattle, dolphins, birds, koalas, and other marsupials.* In contrast to the other systemic mycoses, the prevalence of cryptococcosis in cats exceeds that in dogs, with one study reporting an incidence in cats that was eight-fold that in dogs.150 Historically, five serotypes of Cryptococcus (A, B, C, D, AD) were recognized on the basis of antigenic differences in capsular polysaccharide. C. neoformans comprises serotypes A and D, known as C. neoformans var. grubii and C. neoformans var. neoformans, respectively. Serotypes B and C belong to C. gattii. Based on the results of molecular typing methods, including polymerase chain reaction (PCR) fingerprinting, amplified fragment length polymorphism analysis, and restriction fragment length polymorphism analysis of the orotidine monophosphate pyrophosphorylase (URA5) and phospholipase genes, cryptococcal strains from around the world have been divided into eight molecular types (Web Table 59-2).80,122 Other strains that do not fall within these major groups have also been described, including naturally occurring hybrids between C. gattii and C. neoformans. Differences in epidemiology, pathogenicity, clinical features, and drug susceptibility have been associated with species, varieties, and molecular types. WEB TABLE 59-2 Relationships between Phenotypes and Genotypes of Cryptococcus spp. AFLP, Amplified fragment length polymorphism. Data from Ref. 122. C. neoformans has a worldwide distribution and primarily infects human patients who are immunocompromised, especially patients with acquired immunodeficiency syndrome (AIDS). The environmental niche for C. neoformans is thought to be weathered bird (especially pigeon) guano, in which it mates prolifically.65,123 It can also be found in decaying plant matter in hollows of certain trees.87 C. neoformans passes through the gastrointestinal (GI) tracts of pigeons, but systemic infection of pigeons is rare.98 Pigeon guano provides an alkaline, hyperosmolar environment that is rich in many nitrogen-containing compounds including creatinine that favor cryptococcal growth. Cryptococci may remain viable for at least 2 years in environments such as pigeon lofts, where accumulations of pigeon guano are protected from drying or sunlight. The majority of human cryptococcal infections result from infection with C. neoformans var. grubii. C. neoformans is the most common cryptococcal species isolated from humans, dogs, and cats in southeastern Australia, accounting for more than 70% of infections.96,104,119,125,139 The majority of dogs with cryptococcosis in California are infected with C. neoformans, whereas infection of cats with this species appears to be rare.150 C. gattii has historically been associated with tropical and subtropical climates.42 It is endemic in Australia, New Zealand, Papua New Guinea, Southeast Asia, parts of Latin America, California (especially southern California), Mexico, Hawaii, Central and South Africa, and certain parts of Europe, including Austria, Germany, France, Italy, Greece, and Spain.138,154 Since 1999, infections with C. gattii have been increasingly recognized in humans and domestic and wild animals in North America within British Columbia, especially Vancouver Island, and the Pacific Northwest.† These have been occurring in humans at a rate higher than in areas of the world where C. gattii is endemic.92 Niche areas for C. gattii characterized by low-lying elevations and above freezing daily winter average temperatures have been identified within these zones,30,31 and proximity to areas with soil disturbance was a risk factor for dogs and cats.40 The main molecular types identified in this epidemic have been VGIIa, and to a much lesser extent, VGIIb. In northern California, the majority of cats have been infected with C. gattii VGIII, but molecular subtypes VGIIa and VGIIb have also been detected in dogs and cats from California.150 In contrast, the prevalence of C. gattii infection is lower in cats from southeastern Australia, with most C. gattii isolates belonging to VGI, with VGII predominating in southwestern Australia and the Northern Territory.22,107 C. gattii VGI in Australia is exclusively associated with dead plant material in eucalyptus tree hollows.42,78 VGI is also found in countries where Australian eucalyptus trees have been exported. The ecologic niche of VGII in Australia is less clear. In British Columbia, Canada, the organism is consistently found on living tree bark (Garry oak, maple, cedar, and pine) and in the air, and has also been found in freshwater and seawater.4,30,31,88 In eastern Australia, about 20% to 30% of human cryptococcosis cases are caused by C. gattii,119,125,125 with similar percentages in cats and dogs, whereas in western Australia, approximately 50% of cats and dogs are infected with C. gattii.107 Animals in rural environments tended to be infected with C. gattii, presumably because of increased exposure to eucalyptus material, and all cases recorded in koalas have been attributable to C. gattii, predominantly VGI. In California, C. gattii infections appear to be rare in dogs but common in cats.150 In contrast to C. neoformans, C. gattii primarily infects otherwise healthy individuals,119,139 although infections in immunosuppressed individuals have also been reported.26 Interestingly, koalas serve as a sentinel host for infection, and at the same time, they seem capable of amplifying the number of cryptococci in certain environments.78 Although the natural route of infection acquisition is usually through inhalation, transmission can occur through experimental parenteral inoculation of animals.112,114 Inadvertent transmission of infection in humans has been documented via organ transplantation.1a Localized cutaneous lesions have occurred in naturally infected cats at the site of presumed dermal inoculation (see cutaneous lesions under Cats in the section on Clinical Findings). The exact mode of infection is unproven, but the most likely route is via inhalation of airborne organisms, such as basidiospores or yeast cells desiccated by environmental exposure. Based on the small particle size of infectious propagules, the lung is thought to be the primary site of infection in humans. Shrunken, poorly capsulated cryptococci that are small enough for alveolar deposition have been isolated from pigeon guano and soil. Clinical involvement is uncommon in dogs and cats, although lesions can be detected in thoracic radiographs, thoracic computed tomography (CT) scans, or at necropsy. In cats, dogs, koalas, and psittacine birds, the nasal cavity may be the primary site of infection. C. neoformans var. grubii has been shown to be a transient colonizer of the nasal mucus of cats, dogs, and koalas in Australia,76,105 whereas C. gattii can be isolated in sufficient numbers and with sufficient frequency to be considered part of normal sinonasal flora of koalas in certain environments.76 Studies in koalas have shown that self-limiting, subclinical infection is common, with limited invasion, granuloma formation, and successful eradication or containment of organisms.76 Asymptomatic colonization of the nasal cavity may be followed by the development of mycotic rhinitis in some, but not all, dogs and cats, and others may develop positive cryptococcal antigen tests in the absence of disease.37,38 Nevertheless, some nasal cavity involvement may result from hematogenous dissemination, because intracarotid injection of C. neoformans in cats has been followed by development of lesions on the bridge of the nose. The pathogenesis of cryptococcosis is dependent on the size of the inoculum, the virulence of the cryptococcal strain, and the status of host defenses. Cryptococcus has several virulence factors, including its polysaccharide capsule, melanin, mannitol, laccase, and other enzymes such as phospholipase and superoxide dismutase. The organism has been aptly described as “a sugar coated killer with designer genes.”130 Combined with the ability to grow at mammalian body temperature, the possession of a capsule and the enzymes laccase and phospholipase by C. neoformans and C. gattii likely contribute to enhanced pathogenicity of these species over other Cryptococcus species.27,129 After Cryptococcus infects the host, the capsule enlarges. The capsule is composed of glucuronoxylomannan, which is highly negatively charged. Capsular polysaccharide is continuously shed into the host’s extracellular fluid, including cerebrospinal fluid (CSF) and blood, where it circulates for an extended period. Although the capsule may be chemotactic for neutrophils, the capsule also inhibits phagocytosis through its negative charge and via blockage of the Fc portion of bound antibodies from interacting with the host phagocyte receptors. It also interferes with migration of leukocytes from the bloodstream into infected tissues by causing them to shed selectin. It can also deplete complement and directly inhibits T-cell responses,160 with a shift from a Th1 to a Th2 immune response. Members of the C. neoformans complex may also undergo phenotypic switching, characterized by stable alterations in cell membrane sterol composition and glucuronoxylomannan structure.67 This allows the organism to persist in the host by minimizing the inflammatory response. After infection is established within the lung, the organism is thought to spread hematogenously via macrophages to other sites, particularly the central nervous system (CNS). In the CNS, C. neoformans may cross the capillary endothelium by traveling within deposited infected macrophages or undergo exocytosis within the capillary lumen with transcytosis in which free organisms cross through capillary endothelial cells into the brain parenchyma.23 The incubation period may range from 2 to 13 months or longer.48,49,49 Humans have been shown to be colonized with C. neoformans var. grubii long before they are clinically ill. Exposure and self-limiting infection may occur in some individuals, with reactivation of viable cryptococci in residual granulomatous foci months or years later. Because of this, a travel history to areas where certain types of Cryptococcus are endemic may be available for infected animals and humans residing in nonendemic regions. In animals with nasal cavity involvement, extension of infection may occur into the brain after osteomyelitis of the cribriform plate, into the olfactory bulbs and olfactory tract, or osteomyelitis of the ventral wall of the frontal sinus, with resultant meningoencephalitis. Middle ear involvement has also been documented.6 Spread of infection to the eye, either along the optic nerves from the brain, or as a result of hematogenous dissemination, may result in concurrent cryptococcal optic neuritis and retinitis. The infection typically spreads caudally within the meninges along the floor of the cranial vault. As a result, multiple cranial nerve abnormalities can develop, especially in dogs. Infection may also spread via the CSF to other surfaces of the CNS, such as the dorsal aspects of the brain, and out the foramen magnum along the spinal cord. Cryptococcal antigen has been persistently identified in the CSF within days after experimental parenteral inoculation of cats with C. neoformans.112 Multifocal cutaneous involvement in cats and dogs reflects hematogenous dissemination from the primary site of infection, as do lesions in bone (e.g., digits) or periarticular soft tissues.148 In some cats, infection spreads to the mandibular lymph nodes, presumably via the lymphatics from the nasal cavity. GI cryptococcosis has also been reported, with involvement of segments of bowel, mesenteric lymph nodes, stomach, pancreas, and other intra-abdominal organs.94,97,97 Cryptococcosis can occur in healthy or immunodeficient individuals, although those with T-cell deficiencies are most commonly infected. Although AIDS is the most common underlying immunodeficiency, malignancy, chemotherapy, immunosuppressive drug therapy, diabetes mellitus, or sarcoidosis represent other possible predisposing conditions.139 In metropolitan communities, about 10% of patients with human immunodeficiency virus (HIV)/AIDS are likely to develop cryptococcosis in the absence of highly effective antiretroviral therapy.29 Severe meningitis and disseminated infections are common in immunodeficient patients. When disease does occur in immunocompetent patients, it is often characterized by the presence of granulomatous intracranial or pulmonary mass lesions (cryptococcomas). Because C. gattii tends to infect immunocompetent hosts, cryptococcomas are more common in patients infected with C. gattii compared with those infected with C. neoformans.119,139 In cats and dogs, no gender predisposition exists. The age range of affected cats is broad,104 although young adult cats (2 to 3 years old) appear to be at increased risk. Cryptococcosis is a disease that develops predominantly in young adult dogs, and over 50% of dogs are younger than 4 years at diagnosis.96,107 In one study, the median age of cats was 6 years, and for dogs, 4 years.150 The vast majority of cats with cryptococcosis do not have identifiable underlying immunosuppressive illness. Infections with feline leukemia virus (FeLV) and feline immunodeficiency virus (FIV) have been suggested to predispose cats to cryptococcosis. However, the prevalence of retrovirus infection in cat populations with cryptococcosis does not appear to differ significantly from that in cat populations without cryptococcosis.104,150,150 Cats that are co-infected with FeLV do respond more slowly or fail to respond to treatment and are more likely to have relapses.65,66 Leukocyte and lymphocyte subset numbers in cats with cryptococcosis that had either positive or negative FIV antibody test results were the same. In one study, a positive FIV status did not imply an unfavorable prognosis.155 Many cats with cryptococcosis that also had positive FIV test results are cured of the fungal infection and do not relapse, despite cessation of therapy. Cryptococcosis is occasionally reported in cats receiving immunosuppressive therapy or chemotherapy for malignancy.68,150 The development of cryptococcosis has also been reported shortly before diagnosis of overt malignancy, often lymphoma.93,161 Concurrent opportunistic infections such as toxoplasmosis or aelurostrongylosis have been reported in other cats, suggesting an underlying immunodeficiency.3,90,90 Inadvertent treatment of dogs and cats infected with Cryptococcus spp. with glucocorticoids may be followed by development of disseminated disease, including CNS involvement. Genetic factors may be involved in the predisposition toward development of cryptococcosis. Siamese (Fig. 59-1), birman, and ragdoll cats have been predisposed in Australian studies but were not found to be at risk in California.125,150 Breed predisposition has also varied geographically, with German shepherd dogs, Doberman pinschers, and great Danes being predisposed in Australia, and American cocker spaniel dogs being strongly predisposed in the United States, with an odds ratio of 16.* In dogs, immunosuppressive diseases such as ehrlichiosis have been associated with cryptococcosis. Some dogs, like cats, have evidence of concurrent opportunistic infections such as neosporosis or papillomavirus infection.150 Nevertheless, the majority of dogs have no evidence of obvious immunosuppressive illness.10 Primary cutaneous cryptococcosis has been reported rarely in humans and usually manifests in immunocompetent individuals as a solitary skin lesion in unclothed areas.121 C. neoformans can be found on the skin of healthy humans without infection. Participation in outdoor activities or exposure to bird droppings are risk factors. Cases may occur rarely in cats, possibly after contaminated cat scratch injuries.103 Localized cutaneous involvement has also been reported in ferrets.94 Cryptococcosis is often a chronic infection, causing listlessness and weight loss due to poor appetite. Fever is extremely rare in affected cats. Upper respiratory tract signs are most common and include sneezing; snuffling; and mucopurulent, serous, or hemorrhagic nasal discharge that is unilateral or bilateral. Signs are usually chronic, and in some cases, a polyp-like mass is evident in the nostril (Fig. 59-2). In others, a firm to fluctuant subcutaneous swelling over the bridge of the nose is present (see Figs. 59-1 and 59-2). These swellings may also be located over, and communicate with, the frontal sinus, and palpation of the lesions may reveal crepitus due to subcutaneous emphysema. Cats with nasopharyngeal cryptococcosis develop stertor, inspiratory dyspnea, a tendency to use open-mouth breathing, and occasionally otitis media.63,99 Mandibular lymphadenomegaly may be evident, and ulcerated or proliferative lesions in the oral cavity occasionally develop. CT of the nasal cavity in affected cats may reveal soft tissue and fluid opacification of the nasal cavity or frontal sinus, peripherally contrast enhancing mass lesions of the nasal planum, and associated lysis of the nasal bones or cribriform plate.71 Lower respiratory tract signs are uncommon to rare in cats and include tachypnea or a chronic cough. Thoracic radiographs are often normal, although interstitial to alveolar infiltrates or small nodular lesions may be present. Occasionally, large pulmonary nodules, hilar lymphadenomegaly, a mediastinal granuloma, or pleural effusion are found.65,150 Neurologic signs associated with cryptococcal infection in cats vary depending on the location of lesions in and around the CNS. Common signs include obtundation, changes in temperament and bizarre behavior, hyperesthesia, twitching or tremors, seizures, circling, head pressing, ataxia, paresis, head tilt and other vestibular signs, anosmia, and blindness. These signs may develop individually or in association with other physical findings and may result from the presence of mass like cryptococcomas or meningoencephalomyelitis. In humans, cerebral cryptococcosis is often associated with increased intracranial pressure,119 and the same is likely in cats and dogs. Magnetic resonance imaging findings have been described and include single or multifocal contrast-enhancing mass lesions that tend to be hyperintense on T2-weighted images and isointense on T1-weighted images, as well as changes supportive of meningitis.142 Ocular abnormalities develop in as many as one third of affected cats and almost always are a marker for CNS involvement (Fig. 59-3). Fundic abnormalities may include granulomatous chorioretinitis, exudative retinal detachment, signs consistent with optic neuritis, papilledema, and retinal hemorrhage (see Systemic Mycoses of Dogs and Cats in Chapter 92). Less commonly, anterior uveitis is observed. Severe ocular lesions may result in peripheral blindness with dilated and unresponsive pupils. Cutaneous lesions are common in cats with cryptococcosis, especially solitary or multiple nodular soft tissue masses located on the bridge of the nose (see Fig. 59-2, A). Multifocal skin lesions are the result of hematogenous dissemination; they consist of papules and nodules that are fluctuant to firm and range from one to several centimeters in diameter. Some lesions may ulcerate, leaving a raw surface with a serous exudate (Fig. 59-4). It is possible that some localized cutaneous lesions may result from direct inoculation of propagules (e.g., after a cat scratch; Fig. 59-5). Cats with disseminated disease may also have enlargement of thoracic or abdominal lymph nodes. Renal involvement may be accompanied by isoechoic to hypoechoic mass lesions on ultrasound, with or without associated renal failure. These may involve the renal pelvis. Some cats with renal involvement have no demonstrable sonographic changes.150 Involvement of the spleen, liver, thyroid and salivary glands can also occur. In one study, over 80% of cats had no significant findings based on abdominal ultrasonographic examination. Other reported signs include peripheral lymphadenomegaly (unassociated with skin lesions and often asymmetrical), lameness due to osteomyelitis or cryptococcal arthritis, and swollen digits.65,85 One cat with urinary system involvement presented with lower urinary tract signs.25 In contrast to cats, dogs frequently develop severe disseminated disease. In one study, 80% of dogs had involvement of multiple anatomic locations, and 50% of dogs had involvement of atypical sites (i.e., sites other than the nasal cavity, skin, lungs, lymph nodes, kidney, eye, and CNS).150 Weight loss, lethargy, and inappetence are common nonspecific findings. Dogs may also have signs relating to gastrointestinal or pancreatic involvement. Major organ systems affected by canine cryptococcosis include the CNS, eyes, urinary system, and nasal cavity. Neurologic signs were present in two thirds of the dogs in a study from California.150 CNS signs are typically multifocal, caused by meningitis and progressive meningoencephalomyelitis, and include obtundation, head tilt, nystagmus, anisocoria, facial paralysis, paresis, paraplegia, tetraplegia (usually upper motor neuron), ataxia, circling, seizures, twitching, reluctance to open the mouth, and commonly, cervical hyperesthesia.10,11 Some dogs show signs resembling those of cervical disk protrusion. The most common ocular abnormalities consist of chorioretinitis, signs consistent with optic neuritis, papilledema, and retinal detachment, which may be associated with dilated pupils and blindness. Occasionally, anterior uveitis develops, as does periorbital swelling and cryptococcal conjunctivitis. Magnetic resonance imaging may show changes suggestive of diffuse meningitis and/or multifocal meningoencephalitis, mass lesions with associated edema, and ventricular dilation (Fig. 59-6). Rhinosinusitis from cryptococcosis may be subclinical; thus, its prevalence may be underestimated because respiratory signs are frequently not reported in dogs that show florid neurologic signs.96,150 However, dogs may also have sneezing, mucopurulent nasal discharge, or evidence of an intranasal mass lesion. CT findings include peripherally contrast-enhancing mass lesions within the frontal sinuses, caudal nasal cavity or nasopharynx, or retrobulbar space and changes consistent with osteomyelitis of the skull. As in the cat, involvement of the lower respiratory tract is uncommonly detected antemortem. However, over half of dogs that are subject to necropsy have lower-respiratory involvement.150 When present, changes on thoracic radiographs resemble those described for cats, although nodular lesions are less common, and hilar lymphadenopathy and mycotic pleural effusion may be more common. Ultrasonographic findings include intra-abdominal lymphadenomegaly, multifocal gastric and small intestinal thickening in dogs with GI involvement, changes suggestive of pancreatitis, renal mass lesions, and ascites.150 Less common clinical signs include lameness caused by lytic bone lesions or joint involvement, and peripheral lymphadenomegaly. Box 59-1 summarizes the clinical abnormalities associated with cryptococcosis in dogs and cats. CSF in patients with neurologic signs may have elevated protein concentration (usually between 10 and 140 mg/dL in cats, but may be several hundred or rarely over 1000 mg/dL in dogs). Leukocyte counts may range from 2 to above 1000 cells/µL, with a mean of around 300 cells/µL. A mixed cellular pleocytosis is generally present, with neutrophils or large mononuclear cells generally outnumbering the other cell types. Uncommonly, a marked eosinophilic pleocytosis may occur in dogs.158 In most of the previously mentioned conditions, definitive diagnosis is straightforward and based on obtaining representative tissue specimens for cytology, culture, and occasionally histopathology.34 Suitable specimens may include nasal swabs, nasal washings, needle aspirates from mass lesions or enlarged lymph nodes, bronchoalveolar lavage specimens, pleural fluid, CSF, and urine. In one dog with rectal involvement, the organism was found by cytologic examination of fecal smears.53 A definitive diagnosis of cryptococcosis can be made by culture and identification of the organism by a reputable laboratory. However, it is possible to obtain a high index of suspicion of cryptococcosis by identifying characteristic encapsulated, narrow-based budding yeasts in cytologic smears or histopathology sections. Cytologic examination can be performed on aspirates, body fluids, or impression smears of biopsy specimens. Romanowsky-type stains (Diff Quik, Giemsa, and Wright), new methylene blue, and Gram stain are all satisfactory for making a cytologic diagnosis. Diff Quik is simple and reliable (Fig. 59-7, A). With Gram stain the organism retains the crystal violet, whereas the capsule stains lightly pink with safranin. The organism is often most easily visualized at low power (×10). India ink has been used historically to examine CSF for cryptococci, which appear unstained and silhouetted against a black background, and is still used in the diagnosis of human cryptococcal infections (see Fig. 59-7, B).129 However, considerable experience is required to use this method, because lymphocytes and fat droplets are easily confused with the organism. Yeasts are best demonstrated in CSF using stained cytocentrifuged preparations, combined with culture. Collection of CSF in dogs and cats suspected to have cryptococcosis should only be performed if necessary (see Serologic Testing, later).
Cryptococcosis
Etiology
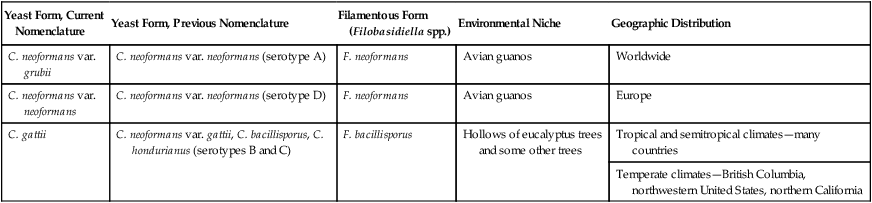
Yeast Form, Current Nomenclature
Yeast Form, Previous Nomenclature
Filamentous Form (Filobasidiella spp.)
Environmental Niche
Geographic Distribution
C. neoformans var. grubii
C. neoformans var. neoformans (serotype A)
F. neoformans
Avian guanos
Worldwide
C. neoformans var. neoformans
C. neoformans var. neoformans (serotype D)
F. neoformans
Avian guanos
Europe
C. gattii
C. neoformans var. gattii, C. bacillisporus, C. hondurianus (serotypes B and C)
F. bacillisporus
Hollows of eucalyptus trees and some other trees
Tropical and semitropical climates—many countries
Temperate climates—British Columbia, northwestern United States, northern California
Epidemiology
Serotype
Species
Molecular Type
AFLP Genotype
A
C. neoformans var. grubii
VNI
1
VNI
1A/1B
B + C
C. gattii
VGI
4
VGII
6
VGIII
5
VGIV
7
D
C. neoformans var. neoformans
VNIV
2
AD
VNIII
3

Pathogenesis
Clinical Findings
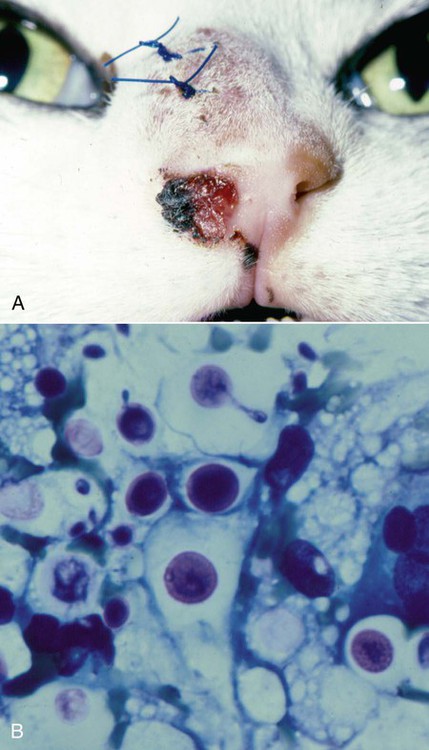
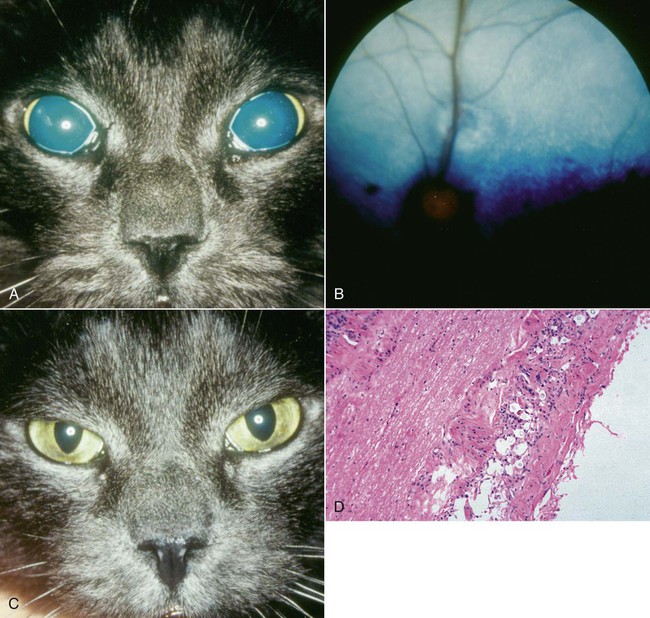
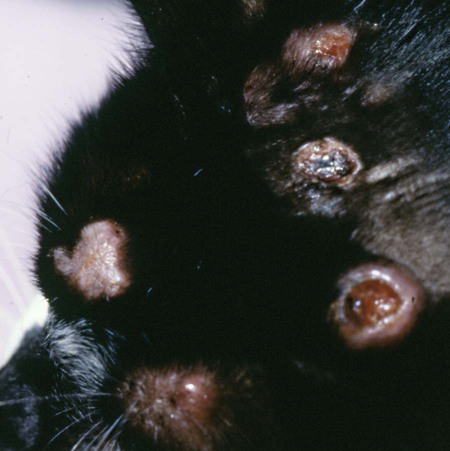
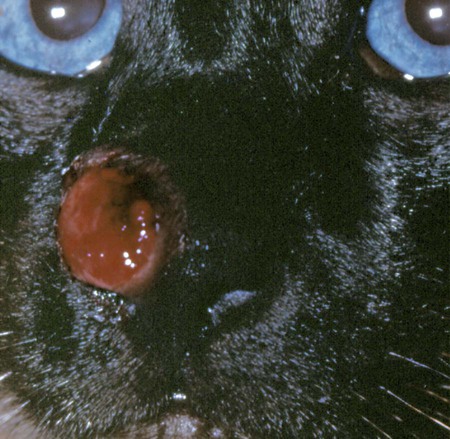
Cats
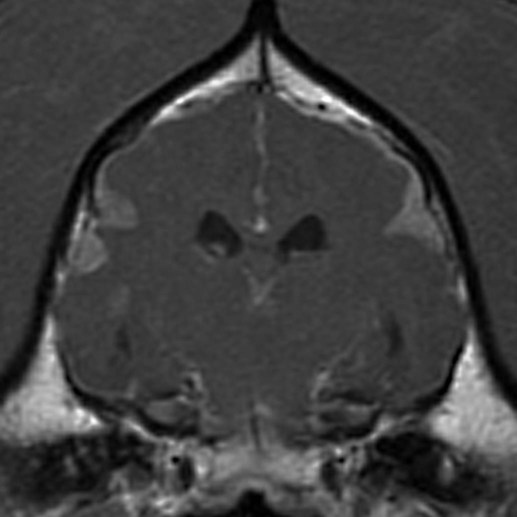
Dogs
Diagnosis
Clinical Laboratory Findings
Specimen Collection
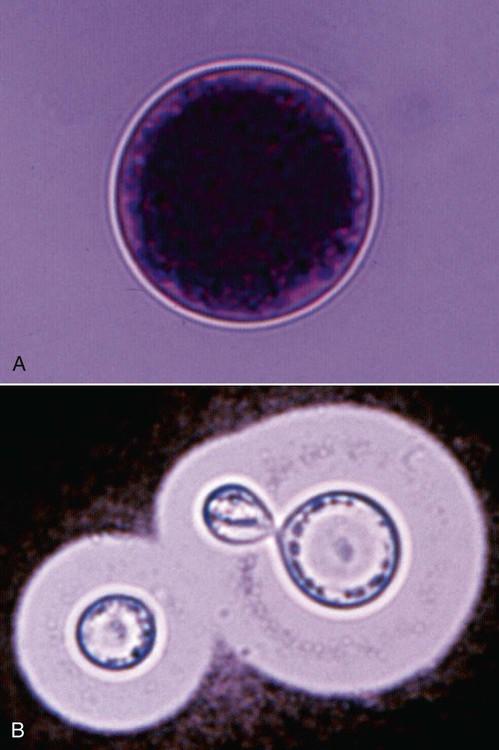
Cytologic Findings
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Cryptococcosis