Chapter 18 Collapse and syncope
DEFINITIONS
The term “syncope” describes a transient and abrupt loss of consciousness and postural tone associated with inadequate cerebral blood flow often due to a cardiovascular anomaly: either a sudden reduction in cardiac output or a loss of vasomotor tone resulting in peripheral vasodilatation. In both circumstances, the result is an abrupt fall in blood pressure. Therefore, horses with collapsing episodes of cardiovascular origin are often undergoing syncope. However, occasionally a disturbance of cerebral nutrient blood flow composition may be the cause of syncopal attacks (e.g. hypoxia, hypercapnia, hypoglycaemia).1
INTRODUCTION
The difficulty in establishing a diagnosis for many cases of nonfatal collapse is reflected by the lack of relevant studies and reports. For comparison, there are a number of reports of studies associated with investigating sudden death in the horse,2–16 which is due in part to the relative ease in establishing a diagnosis at necropsy. Nevertheless, even with extensive post-mortem examination, the cause of death is not determined in approximately 30% of horses that die suddenly. However, it is important to consider that the cause of collapse in one horse may be the cause of sudden death in another, or that a number of collapsing episodes may occur in an individual before a fatal attack, or that a collapsed horse may subsequently die. In a review of 69 cases of sudden and unexpected death, in 24 of the cases, collapse and subsequently death were observed; in 6 of those 24 horses, death was due to trauma. Cardiovascular reasons were implicated as the cause of death in nine horses: six died from internal haemorrhage, one had aortic valvular disease, one had ruptured chordae tendineae of the mitral valve and one had a verminous granuloma in the coronary artery. In the remaining nine, seven of which had died at exercise, a post-mortem examination failed to reveal pathological changes and a functional cardiac disorder was postulated.15
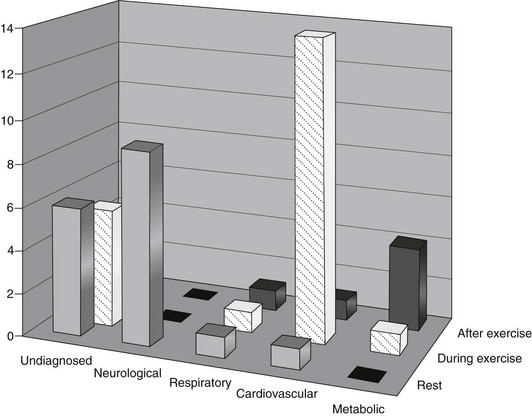
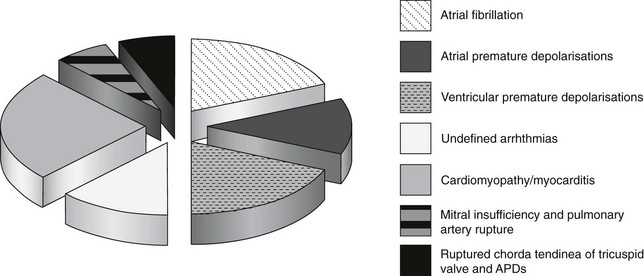
Circulatory causes of nonfatal collapse in the horse occur commonly: 8.5% of horses referred for a second opinion for evaluation of the cardiovascular system at a referral centre had actually collapsed, and a further 11% had a history of inco-ordination, stumbling or unsteadiness and/or recumbency.17 Figure 18.1 details the final diagnosis for 44 horses presented with a history of collapse after referral for cardiac investigation at a veterinary hospital. The cardiovascular causes of collapse in this group are illustrated in Figure 18.2.
CARDIOVASCULAR AND CIRCULATORY CAUSES OF COLLAPSE
Syncope
Horses that have suffered one or more episodes of acute, nonfatal collapse may be suspected of suffering from syncope. Usually there is little or no warning of collapse and, because of cerebral hypoxia, a temporary, quiet, comatose state ensues.18 The animal may go on to recover or progress to die, in which case other signs of cardiac failure usually become evident.
An alert state is maintained through multiple sensory inputs to the reticular activating system in the rostral brain stem and subsequently to the thalamus and cerebral cortex where consciousness is maintained18: in human beings it has been estimated that cerebral oxygen delivery of approximately 3.5 mL oxygen/100 g tissue each minute is required for consciousness.19,20 A reduction of cerebral oxygen delivery below this level for periods of 10 seconds or more is associated with loss of consciousness and postural tone.21 Cerebral blood flow is usually carefully maintained at a relatively constant level of 50–60 mL/100 g of tissue/minute by autoregulation: a drop to half this level may precipitate a syncopal attack. Cerebral blood flow is influenced by PaCO2, PaO2 and pH; however, autonomic control mechanisms (that profoundly affect blood flow in the rest of the body) appear to be capable of only fairly small changes in brain blood flow because of relatively sparse innervation.22
Acute haemorrhage
Acute haemorrhage leading to collapse occurs with the rupture of major vessels such as the aorta23,24 (Fig. 18.3), the pulmonary artery24 (Fig. 18.4), the anterior mesenteric artery and the uterine arteries.25 Shock and rapid death can follow profuse haemorrhage into the pleural or abdominal cavities. Collapse and haemorrhagic shock may also occur following rupture of vessels associated with the guttural pouch in disorders such as empyema and mycosis or with severe exercise-induced pulmonary haemorrhage.11
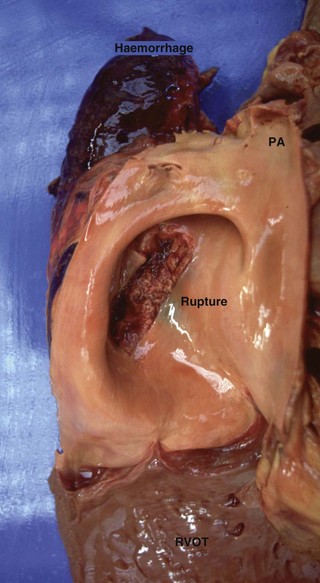
While rupture of any major vessel is possible, if the horse survives the episode, the specific site may not be identified. However, certain vessels appear to be ruptured more commonly because of pre-existing disease. Rupture of the pulmonary artery leading to syncope or sudden death has been reported in association with pulmonary hypertension caused by patent ductus arteriosus26 and severe mitral insufficiency27 (see Fig. 18.4).
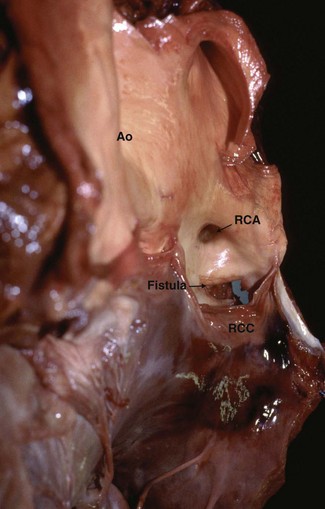
Aorto-cardiac fistula is a particularly well-documented cause of vessel rupture, occurring most commonly in stallions: in one report, four of the eight cases collapsed and died within minutes of covering.28 On post-mortem examination, tears of the aortic ring were identified extending from the right coronary sinus into the right ventricle similar to that shown in Figure 18.3. Some cases die suddenly29 but horses can survive formation of an aorto-cardiac fistula for periods varying up to 12 months.30–33 A characteristic low-pitched continuous murmur may be auscultated over the right thorax and monomorphic ventricular tachycardia is often present due to disruption of the conduction tissue by a dissecting haematoma33 that can be visualized by echocardiography. Some horses appear to have abdominal pain but this may in fact be thoracic pain that can be difficult to distinguish from that arising from the abdomen.30,33 The pathogenesis remains unclear: aortic root rupture may be due to congenital abnormalities, cardiac degenerative changes, associated with chronic aortic insufficiency or occur because of pre-existing sinus of Valsalva aneurysm.30–34 (![]() ACF)
ACF)
Collapse and death may occur as a result of the rupture of arteries at or near parturition: the utero-ovarian, middle uterine and external iliac arteries are most commonly affected. This is also a common cause of abdominal pain and haemoperitoneum in the periparturient brood mare. Pre-existing degenerative changes are thought to be responsible, in combination with the stresses associated with late gestation and parturition.25
Neoplasia should be considered as a possible underlying cause of pleural or abdominal haemorrhage. The rupture of a thoracic haemangiosarcoma with corresponding collapse and death from haemorrhagic shock has been reported35 as has sudden death, caused by thoracic lymphosarcoma.2 Collapse and sudden death in a Thoroughbred stallion during breeding have been associated with intraseptal haemorrhage and atrioventricular node disruption caused by a malignant melanoma.14
Acute cardiac failure
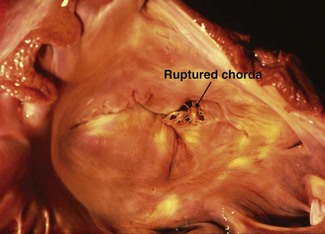
Inadequacy of the pumping mechanism of the heart can have profound effects on arterial blood pressure and hence cerebral blood flow. The most notable spontaneous example is probably mitral valve chorda tendinea rupture leading to severe valve incompetence, regurgitation into the left atrium during systole, pulmonary congestion and oedema7 (Fig. 18.5, see Chapter 19). (![]() RCT) These horses may collapse with severe dyspnoea and cyanosis.36 A loud pansystolic murmur is audible over the left thorax with often a palpable thrill. If the horse survives, clinical signs may improve; however, generally the murmur remains, loudest over the mitral value, and a loud third heart sound may be audible and there may be caudal extension of the cardiac area of auscultation associated with left atrial dilatation. This may be followed by the progression to cardiac failure, possibly with further episodes of collapse, often exacerbated by exercise.7,27,37
RCT) These horses may collapse with severe dyspnoea and cyanosis.36 A loud pansystolic murmur is audible over the left thorax with often a palpable thrill. If the horse survives, clinical signs may improve; however, generally the murmur remains, loudest over the mitral value, and a loud third heart sound may be audible and there may be caudal extension of the cardiac area of auscultation associated with left atrial dilatation. This may be followed by the progression to cardiac failure, possibly with further episodes of collapse, often exacerbated by exercise.7,27,37
Dysrhythmias
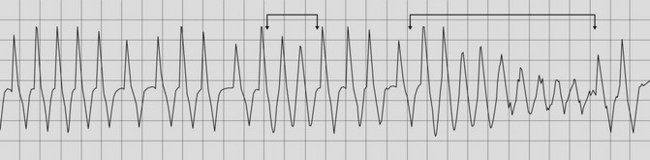
Cardiac rhythm disturbances are amongst the more common causes of collapse, particularly in horses that collapse during exercise (Fig. 18.6). Collapsing episodes occur as a result of failure to maintain cardiac output, caused by the inability to regulate either heart rate or stroke volume in periods of either brady- or tachyarrythmia.38 A normal horse will increase cardiac output at submaximal rates (less than 210 bpm) primarily by tachycardia; at maximum heart rates of between 210 and about 240 bpm increasing cardiac output is achieved with increased stroke volume39 (see Chapter 3). With tachyarrythmias such as atrial fibrillation40,41 and ventricular tachycardia, heart rates in excess of 240 bpm may be reached. In these circumstances there may be limited perfusion of the myocardium during diastole, leading to poor contractility and compromised cardiac output as a result of inadequate ventricular filling. Disease processes that alter myocardial contractility (e.g. focal ischaemia) will compound this effect, which may become apparent even at submaximal heart rates. (![]() AF, RCT)
AF, RCT)
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree