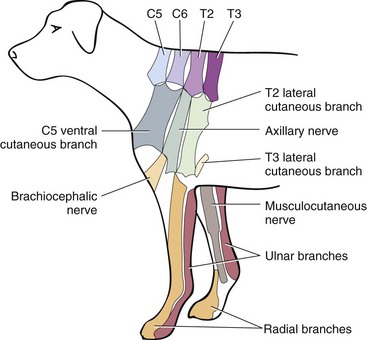
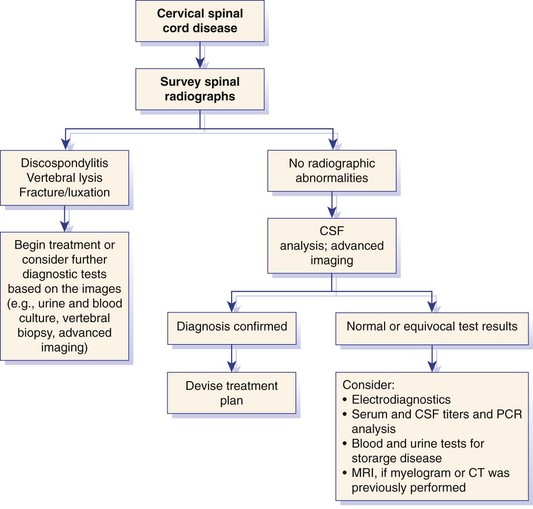
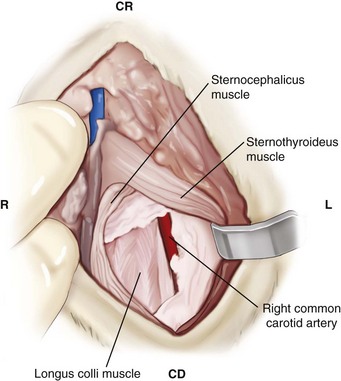
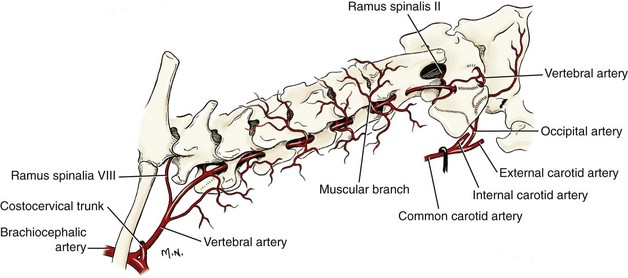
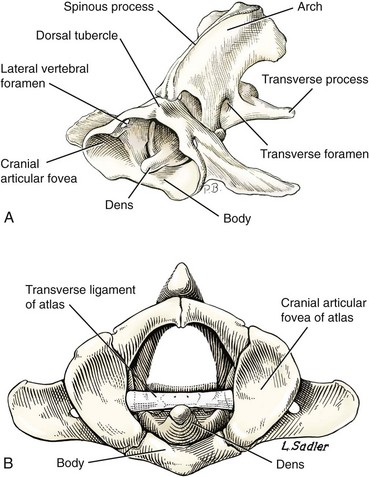
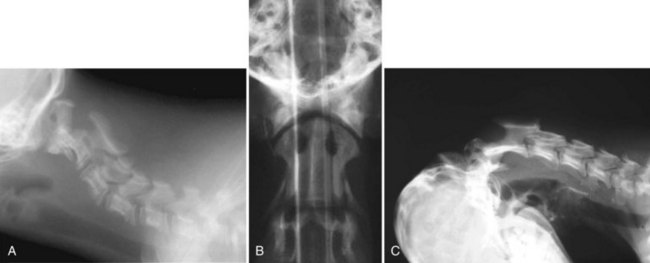
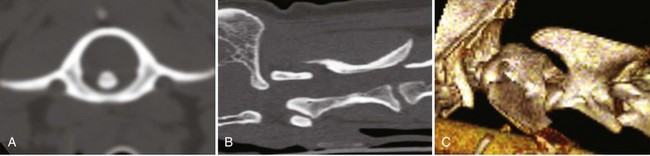
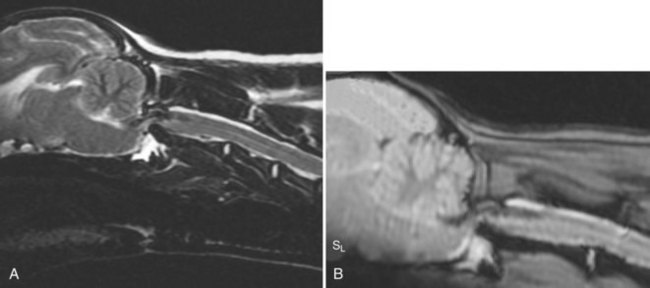
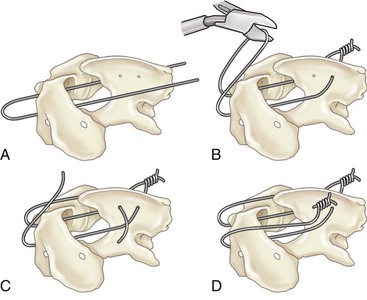
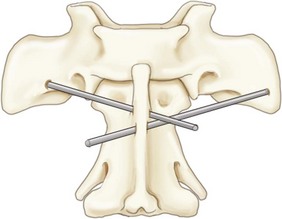
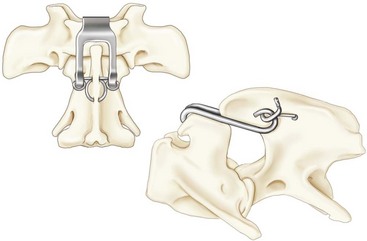
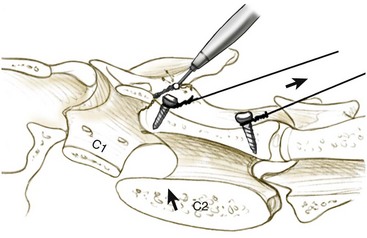
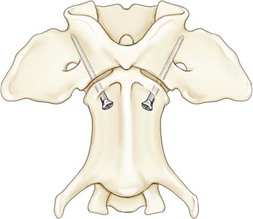
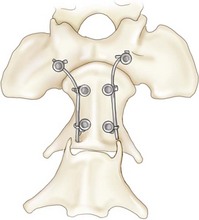
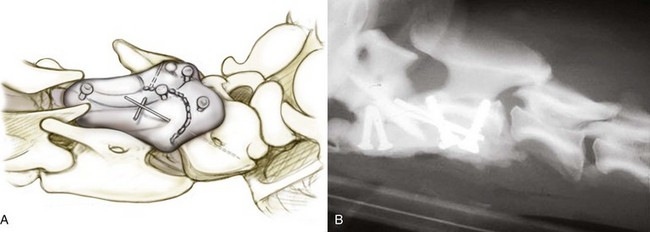
Chapter 31 Observation of an animal’s gait, posture, and mentation, in addition to examination of postural reactions, spinal reflexes, cranial nerve function, and state of consciousness, is necessary to confirm the presence of spinal cord disease; these tests will also assist with accurate lesion localization and in determination of whether a focal, multifocal, or diffuse disease process is responsible for any abnormalities found.141 An animal with spinal cord disease can display clinical signs as described in the following five groups: (1) reduction or loss of voluntary movement, (2) spinal reflex abnormalities, (3) alterations in muscle tone, (4) muscle atrophy, and (5) sensory dysfunction. Use of these five groups can help the clinician to accurately localize the neurologic lesion (Table 31-1). Most spinal cord diseases will result in varying degrees of abnormality of both movement and sensation because the neuroanatomic pathways responsible for these properties are adjacent to each other. Clinical signs of intracranial disease, in addition to the aforementioned signs, suggest a multifocal disease process or a focal brainstem disease.216 Table • 31-1 Clinical Signs Manifested by a Focal Lesion Affecting the Cervical Spinal Cord It is often the case that animals with cervical spinal cord disease will present with more pronounced signs of motor dysfunction in the pelvic limbs; this seems more common in large-breed dogs. The main reason for this observation is that the motor pathways to the pelvic limbs are more peripherally located in the spinal cord than those responsible for thoracic limb motor function. Therefore, particular attention should be paid to the thoracic limb gait when an animal presents with ambulatory paresis that apparently affects only the pelvic limbs, as this could represent mild cervical spinal cord dysfunction. The stride length of the thoracic limbs in an ambulatory tetraparetic animal can sometimes be helpful to further localize the responsible lesion. Lesions affecting the C1-C5 spinal cord segments may result in increased thoracic limb stride length, and those affecting the C6-T2 spinal cord segments may result in reduced stride length. The latter presentation is often accompanied by increased stride length in the pelvic limbs and is sometimes called a “two-engine” gait. Reduced stride length in all four limbs is most likely due to neuromuscular lesion localization or to a diffusely painful condition such as polyarthritis.141 Occasionally, an animal will present with more profound motor dysfunction in the thoracic limbs than in the pelvic limbs. If the pelvic limbs are normal in their function, this indicates a peripheral lesion such as brachial plexus trauma or neuritis. If the pelvic limbs manifest neurologic dysfunction, the disparity is often due to central spinal cord syndrome, whereby the disease process more severely affects the center of the spinal cord (gray matter and lower motor unit cell bodies) than the periphery of the spinal cord (white matter tracts to thoracic and pelvic limbs). The most clinically useful and objective reflexes in the thoracic limbs are the flexor withdrawal reflexes.71 However, although the examination of spinal reflexes can be extremely helpful in narrowing down lesion localization in a tetraparetic animal, 34% of dogs with a single-level cervical disc lesion were incorrectly localized on the basis of their reflex examination.71 Most commonly, these dogs had a decreased withdrawal reflex noted in the thoracic limbs that was associated with a lesion of the C1-C5 spinal cord segments.71 This suggests that a decreased withdrawal reflex does not always indicate a lesion affecting the C6-T2 spinal cord segments. Abnormalities of muscle tone are interpreted in a similar fashion to abnormalities of spinal reflexes. Muscle tone may be depressed (hypotonia), absent (atonia), or exaggerated (hypertonia), dependent on lesion localization.141 Two types of muscle atrophy may occur in association with cervical spinal cord disease. Neurogenic or denervation atrophy of the thoracic limbs is seen with lesions affecting the lower motor units (C6-T2) and is evident within 7 days of disease onset; it is usually severe enough to enable observation of the bony prominences of involved limbs.141 Disuse atrophy of the thoracic limbs indicates a lesion affecting the central nervous system cranial to the C6-T2 segments and takes several weeks of disease before it becomes evident.141 Ataxia or incoordination is usually evident in all dogs with ambulatory tetraparesis due to spinal cord disease. It is confirmed on the basis of gait observation alone and indicates involvement of the sensory tracts of the spinal cord.141 Postural reactions (e.g., proprioceptive positioning) should be tested in all animals with suspected cervical spinal cord disease. Abnormalities of postural reactions are often the first sign of a myelopathy and can assist with confirmation of a neurologic disease as opposed to an orthopedic disease.141 Pain perception is usually classified as normal, depressed (hypesthesia), absent (anesthesia), or exaggerated (hyperesthesia). Two types of pain perception can be assessed in veterinary patients: cutaneous (superficial) pain and deep pain. Cutaneous pain is manifested by response to pricking or pinching the skin, and deep pain perception is manifested by pinching the bones of the digits. Loss of cutaneous sensation may be useful for mapping out specific nerve, nerve root, and spinal cord segment dysfunction, particularly worthwhile with lesions affecting the brachial plexus (Figure 31-1).141 It is unfortunately a subjective test in veterinary medicine, and so the unaffected limbs should be used as controls before a decision can be made on the quality of the animal’s response. Disease of the cervical spine may result in pain, which may be the only clinical sign or may accompany the signs mentioned previously. Numerous structures in the neck may be affected to cause cervical pain; these include the meninges over the spinal cord and nerve roots, the annulus fibrosus of the intervertebral discs, the periosteum over the vertebrae, the joint capsule (especially that of the zygapophyseal joint, which is the diarthrodial joint of the articular processes), the epaxial musculature, and the ligamentous structures surrounding the vertebrae. It should also be noted that intracranial disease may cause a “referred” type of neck pain in circumstances or diseases in which elevated intracranial pressure may result in compression or stretching of the cerebrovasculature, which itself is densely innervated with nociceptors.34 Neck pain may be obvious from the animal’s posture or behavior, but it may be detectable only by careful physical examination. Palpation of the vertebral column to assess for pain can be done at the end of the neurologic examination to avoid distressing the patient; this is performed by pressing on the spinous processes or squeezing the articular or transverse processes, depending on the size and temperament of the animal. Just before firm palpation, the spine should be palpated for curvature, displacement, masses, swelling, and paraspinal muscle atrophy.217 Cervical pain is often manifested by tensing of the cervical muscles and twitching of the ears during palpation.217 Turning of the head and neck with the palm of the hand placed on the side of the neck to evaluate for resistance to movement in addition to flexion and extension of the neck can also be performed; if possible, the pain should be located to cranial, middle, or caudal cervical segments. If there is a history of trauma or the patient is a toy-breed dog, neck movement during the examination should be kept to a minimum to avoid exacerbating an underlying subluxation. Occasionally, an animal may hold a thoracic limb in a partially flexed position, a posture that may be consistent with C1-T2 nerve root or spinal nerve entrapment. It is more common with C6-T2 spinal segment lesions and is termed “root signature.”141 Diseases of the cervical spinal cord can result in loss of both urinary and fecal continence (of an upper motor neuron origin), particularly if cystic lesions are affecting the dorsal portions of the spinal cord, which may disrupt the sensory pathways for defecation30,197; however, upper motor neuron incontinence is more of a problem associated with spinal cord diseases localized caudal to the second thoracic vertebrae.27,30,155 Reflex urination and defecation will remain intact with upper motor neuron lesions. The differential diagnoses that should be considered when a disease is localized to the C1-C5 spinal cord segments or the C6-T2 spinal cord segments are listed in Tables 31-2 and 31-3, respectively. The animal’s signalment, history, and presenting complaint and the results of its neurologic examination are used to determine the most appropriate diagnostic plan (Figure 31-2). At minimum, data should be obtained from multiple sectors—including hematology, serum chemistry, urinalysis, and radiography of cavity areas—for most animals with a cervical spinal cord lesion localization. These data are used to investigate the possibility of systemic disease, such as infection, and to evaluate the patient for potential anesthetic risks. The latter is necessary, as many patients will require anesthesia for further investigation, which may include advanced imaging (myelography, computed tomography [CT], or magnetic resonance imaging [MRI]), cerebrospinal fluid tap, and electromyography. Spinal radiography can be performed without anesthesia in many patients to rule out lysis, bone proliferation, and traumatic damage of the vertebrae. In such cases, even sedation can present a risk, as it may be accompanied by muscle relaxation, which could exacerbate underlying vertebral instability. Ventral Approach to the Cervical Spine A ventral approach to the cervical spine166 may be necessary for a ventral slot, disc fenestration, fracture repair, or atlantoaxial fusion and to address cervical spondylomyelopathy. The reader is referred to other texts for a detailed description of the standard approach.164 A modified ventral approach to the cervical vertebrae via a paramedian dissection has been described.26 After identification of the sternocephalicus and sternohyoideus muscles, the right sternocephalicus muscle is separated from the right sternohyoideus muscle (Figure 31-3). The sternohyoideus muscles are then retracted to the left with the trachea, esophagus, and carotid sheath. Further exposure of the vertebrae after this point is as for the routine approach described previously. This approach helps protect the trachea, right recurrent laryngeal and vagosympathetic nerves, and right carotid sheath, while providing increased exposure of the caudal cervical vertebrae and decreasing the likelihood of hemorrhage from the right caudal thyroid artery.26 The ventral approach to the atlantoaxial junction201 has been described.164 A modification of this approach utilizing a right-sided parasagittal approach to the atlantoaxial junction also has been described. Potential advantages of the modified approach include improved exposure of the joint and avoidance of dissection around the thyroid gland, trachea, and recurrent laryngeal nerve.193 A ventral median incision is made from a point at the level of the caudal third of the mandible to a point at least 5 cm caudal to the base of the larynx. The right sternothyroideus and sternocephalicus muscles and the right carotid sheath are then exposed and separated with blunt dissection, exposing the paired longus colli muscles and the ventral midline of the cervical vertebrae (Figure 31-4). Careful retraction of the right carotid sheath to the left side and separation of the sternothyroideus and sternocephalicus muscles result in a better view of the paired longus colli muscles on the ventral surface of the cervical vertebrae.193 Surgical orientation can be provided by locating the sharp ventral prominence on the caudal aspect of C1; this prominence is the ventral midline location of the C1-C2 interspace. The longus colli muscles are elevated away from the bodies of C1 and C2 as for the standard midline approach, exposing the joint for ventral stabilization. This approach appears to provide an improved ability to drill or drive a wire across the right C1-C2 articulation without the larynx and trachea directly adjacent to the surgical field.193 A lateral approach to the cervical vertebrae and vertebral canal132 is useful for animals with lateral or intraforaminal disc extrusions, as well as ventral or dorsal nerve root tumors, spinal nerve tumors, and other laterally located masses.164 A modified lateral approach to the cervical spine is viable for surgical treatment of cervical myelopathic or radiculopathic lesions when exposure to foraminal and lateralized lesions of the vertebral canal involving the C2-C7 vertebral articulations is desirable.176 Patients are positioned in lateral recumbency with the affected side up and with the nondependent leg retracted caudally. The articular processes of C3-C6 are palpated to localize the operator. A curvilinear skin incision is made from C2 to the cranial margin of the scapula at the level of the cervical zygapophyseal (articular process) joints, which exposes the underlying platysma muscle. The platysma muscle is incised to expose the underlying brachiocephalicus and trapezius muscles. In the cranial cervical region, the splenius and serratus ventralis are exposed by bluntly dividing and retracting the brachiocephalicus muscle in a direction parallel to its individual fibers, using a grid technique (Figure 31-5). Superficial fibers of the serratus ventralis muscle are bluntly divided and retracted, facilitating exposure to the medial fibers of the serratus ventralis muscle, which are subsequently bluntly dissected from the underlying muscles of the longissimus system.176 To approach the C5-C7 vertebral segments, the splenius and serratus ventralis muscles are exposed by separating the brachiocephalicus muscle craniolaterally and the trapezius muscle in a caudodorsolateral direction after insertion of a self-retaining retractor in the fascial plane that naturally divides these muscles (Figure 31-6). The superficial cervical artery and vein, which are located between the brachiocephalicus and trapezius muscles, are then isolated, ligated, and divided. Exposure to the C6-C7 vertebral segment is further facilitated by simultaneous abduction and caudal retraction of the scapula. Retraction of the scapula is performed to expose the C6-C7 vertebral segment without the need to incise the muscular attachments on the cranial border of the scapula. Lesion localization can be performed by palpating the prominent transverse processes of C6, or by palpating the first rib. The appropriate articular processes are exposed by creation of a plane of dissection between the longissimus capitis and complexus muscles.176 The dorsal branch of the spinal nerve leaving the intervertebral foramen needs to be sacrificed and bipolar electrocautery is then used to incise the tendinous attachments of the complexus and multifidus muscles from the articular processes. A periosteal elevator can be used to elevate these muscles dorsally to the level of the spinous processes, and the longissimus capitus muscle is sharply dissected from the transverse processes and reflected ventrally, to fully expose the joints. Removal of the articular processes performed after this point is as described for the standard lateral approach. A dorsal approach to the cervical spine75,102,159 may be necessary to perform a dorsal laminectomy, fracture fixation, and atlantoaxial stabilization. The reader is referred to detailed descriptions of these procedures.164 A lateral approach to the brachial plexus147 was developed for the surgical treatment of brachial plexus avulsion but is also ideal for the exploration of brachial plexus neoplasia. With the dog in lateral recumbency, a long curvilinear incision is made 3 to 4 cm cranially from the midpoint of the cranial border of the scapula to a point located slightly distal to the greater tubercle of the humerus. The platysma and fascia cervicalis are incised, exposing the cleidocervicalis, omotransversarius, and trapezius muscles (Figure 31-7, A). The superficial cervical artery and vein, which emerge between the cleidocervicalis and trapezius muscles, should be ligated. The superficial cervical lymph node, lying medially to these vessels, should then be retracted caudally. The omotransversarius muscle can then be incised near its insertion on the spine of the scapula and retracted cranially. The dissection is continued medially through the deep fascia along the dorsal border of the cleidocervicalis muscle, which is withdrawn ventrally. A self-retaining Gelpi retractor is positioned between the cleidocervicalis and trapezius muscles (Figure 31-7, B). The scapula is withdrawn caudally with a Farabeuf retractor. The extrathoracic part of the brachial plexus can now be exposed and palpated ventrally. The atlas articulates with the skull cranially at the occipital condyles via its modified articular processes, and with the axis caudally. This vertebra lacks a dorsal spinous process; it has large “wing-like” lateral expansions as transverse processes and a reduced body size when compared with the rest of the cervical vertebrae. The cranial articular surface consists of two cotyloid cavities that may meet ventrally. They articulate with the occipital condyles, forming a joint in which the main movements are flexion and extension, allowing up and down movement of the head, and so it is often called the “yes joint”; however, in reality, this joint permits little flexion. The caudal articular surface consists of two shallow glenoid cavities that allow free movement between the atlas and the axis. Movement at this joint is mainly rotational, and therefore the atlantoaxial joint is often called the “no joint.” The dorsal surface of the body of the atlas contains the fovea of the dens, which is concave from side to side and articulates with the dens of the second cervical vertebra. In addition to the large vertebral foramen through which the spinal cord passes, two pairs of foramina are present in the atlas. The transverse foramen is a short canal passing obliquely through the transverse process or wing of the atlas; the lateral vertebral foramen perforates the craniodorsal part of the vertrebral arch.64 Cranial and caudal notches are located at the origin of the transverse processes. The atlantal fossae are depressions ventral to the wings. The vertebral vein and artery traverse the atlantal fossa. The vein extends through the transverse foramen caudally and anastomoses with the internal jugular vein in the ventral condyloid fossa rostrally. A venous branch runs dorsally through the cranial notch in the wing and aids in forming the external vertebral venous plexus. The vertebral artery enters the vertebral canal through the lateral vertebral foramen, after first having run through the transverse foramen of the atlas (Figure 31-8).64 Whereas the adult atlas develops from three bony elements (a pair of neural arches that become the vertebral arch and a body), the axis develops from seven bony elements (a pair of neural arches, three parts of the vertebral body, the dens, and the apical element on the dens). The appearance and fusion times of these elements in both bones vary even within litters; in Beagles, fusion of the suture at the dorsal midline of the atlas is normally complete by 106 days after birth, and fusion of the ventral sutures is complete by 115 days after birth.64 Fusion of all of the sutures of the axis progresses from 30 to 396 days after birth.64 The axis has an elongated dorsal spinous process, which overhangs the cranial and caudal articular surfaces of the vertebral body. The cranial vertebral notches concur on either side with those of the atlas to form the large intervertebral foramina out of which run the second pair of cervical nerves and the intervertebral vessels. The axis also has a cranioventral peg-like projection called the dens or odontoid process. The dens lies within the vertebral foramen of the atlas, held down by the transverse ligament, which prevents its movement into the spinal canal but still allows rotational movement. The dens is also attached to the foramen magnum by the apical ligament and to the occipital condyles by bilateral alar ligaments. A dorsal atlantoaxial ligament joins the dorsal arch of the atlas and the craniodorsal spine of the axis (Figure 31-9). Atlantoaxial subluxation was first reported in dogs in 1967.79 Since that time, several congenital and developmental deformities of the atlantoaxial joint have been documented to cause instability of the vertebral column, predisposing to atlantoaxial subluxation, particularly in young, small-breed dogs.76,130,227,229 Possible congenital or developmental anomalies of the atlantoaxial joint include dysplasia (34% of dogs), hypoplasia or aplasia (46% of dogs), dorsal angulation, and separation of the dens, as well as absence of the transverse ligament.12,76,139,229 Any abnormality of the dens will predispose to instability of the atlantoaxial joint because of its important role in the normal stability of this joint; however, up to approximately 24% of dogs with atlantoaxial subluxation will have a normal dens.12 Recent additions to this list of anomalies associated with atlantoaxial subluxation include incomplete ossification of the atlas and the presence of block vertebrae.130,227 Incomplete ossification of the atlas has been documented in five dogs, with four of the dogs exhibiting concurrent atlantoaxial subluxation associated with relative dorsal displacement of the axis.227 The authors suggested that this vertebral anomaly may be a contributory factor to subsequent vertebral instability because associated deficiencies of the atlantoaxial ligaments or their attachments may allow laxity of the joint.157,227 Block vertebrae have been reported in association with atlantoaxial subluxation in two dogs.130 Block vertebrae are rare but well-known congenital vertebral deformations involving the fusion of two or more vertebrae. This vertebral deformity may occur at any level of the vertebral column and is usually considered an incidental radiographic finding; however, block vertebrae may contribute to clinical signs of spinal cord disease in some animals.7 In the two dogs reported to have concurrent atlantoaxial subluxation, the authors hypothesized that congenital vertebral fusion created a “fulcrum effect” that predisposed the dogs to traumatic atlantoaxial subluxation.130 Small breeds of dogs, including Yorkshire Terriers, Chihuahuas, Miniature Poodles, Pomeranians, and Pekingese, are most often affected by the congenital and developmental anomalies that predispose to atlantoaxial instability and potential subluxation.90,139,215 This occurs mainly because the dens is prone to maldevelopment in miniature breeds because of aberrations in physeal growth plate closure. However, atlantoaxial subluxation due to congenital vertebral anomalies has also been reported in large-breed dogs.101,230 Atlantoaxial subluxation due to congenital vertebral anomalies is very rare in cats.104,192,218 Traumatic atlantoaxial subluxation can occur in dogs of any breed and age. Traumatic atlantoaxial subluxation results from forceful overflexion of the head, which may tear the ligaments or cause a fracture of the dens or dorsal arch of the axis.139 Considerable impact may be required to cause such injuries in a normal atlantoaxial joint, and so many times even traumatic atlantoaxial luxations are associated with an underlying congenital defect and instability of the joint.12,139 Instability of the atlantoaxial joint associated with congenital anomalies or subsequent to trauma allows excessive flexion of the joint and causes spinal cord compression and concussion from the dorsally displaced cranial portion of the axis in relation to the atlas.139,181 Spinal cord trauma secondary to an acute episode of subluxation has the same underlying pathophysiology as that related to acute disc extrusion and acute vertebral fracture luxation. Neck pain is the single most common sign associated with atlantoaxial subluxation; it is seen in most dogs with traumatic lesions and in 30% to 60% of dogs with congenital lesions.12,139,201,215 The associated neurologic deficits are determined by the degree of damage present in the spinal cord following both concussion and residual compression. Neurologic deficits can range from mild postural reaction abnormalities (56%) to tetraplegia (10%); overall, gait dysfunction has been reported in up to 94% of dogs.12,165,201 These deficits can appear asymmetric in addition to appearing worse in the pelvic limbs or the thoracic limbs. In the rare cases that present with tetraplegia, progression of the clinical signs to a state of clinical respiratory compromise and even arrest is possible.139 Severe signs can be present in some dogs in the absence of notable compressive disease; the use of magnetic resonance imaging has confirmed the presence of significant parenchymal pathology, including hemorrhage, in such dogs.113 Abnormalities of posture such as torticollis can be seen in dogs with atlantoaxial subluxation due to concurrent syringohydromyelia or vestibular dysfunction, or associated with the underlying pain.80,111 Occasionally, dogs will present with intracranial signs that are purported to be related to extension of parenchymal pathology into the brainstem, basilar artery compromise,104 concurrent hydrocephalus,28,58 and other incidental diseases, such as hepatic encephalopathy, that are prevalent in toy-breed dogs.183 Seizure activity may be temporally associated with atlantoaxial subluxation; when this occurs, it is likely that the seizures are an inciting cause of subluxation in a dog with a preexisting instability.165 Atlantoaxial subluxation can be diagnosed from survey radiographs of the cervical spine, although extreme care must be taken when restraining and moving dogs in which this disease is suspected. Unsedated views may be attempted and are especially advised if the dog could have a fracture. If the animal is sedated or anesthetized, the head and neck should be supported in slight extension to avoid further spinal cord injury. On lateral radiographs, increased space can be seen between the dorsal lamina of the atlas and the dorsal spinous process of the axis (Figure 31-10, A). In severe cases, malalignment of the bodies of the atlas and axis is clearly visible. Objective parameters that take into account the normal range of motion of the atlantoaxial joint have been reported recently.145 An angle between the atlas and the axis of less than 162 degrees is more predictive of instability than a decrease in atlantoaxial overlap.145 Without objective measurement, some dogs may be incorrectly diagnosed. The presence and size of the dens can be evaluated most accurately on ventrodorsal (VD) views (Figure 31-10, B); this can also be well evaluated on oblique radiographs.36 These views are preferable to open-mouth views, which place the patient at severe risk of spinal cord trauma. If no evidence of subluxation is found on the lateral views, the neck can be carefully flexed to check for instability (the space between the dorsal lamina of the atlas and the dorsal spinous process of the axis should be evaluated) (Figure 31-10, C). It is preferable to do this with fluoroscopy, so that the movement can be monitored to prevent accidental iatrogenic subluxation; this can provide a rapid diagnosis in a conscious dog. However, flexion of the atlantoaxial joint when the dens is dorsally deviated risks severe iatrogenic trauma to the spinal cord. Computed tomography and MRI can add vital information, which helps with decision making regarding treatment of the individual patient.111,113,181 Computed tomography can assist with identification of dens conformation or dens or vertebral fracture presence, as well as with surgical implant placement (Figure 31-11, A and B).111,181 Three-dimensional CT reconstruction of the atlantoaxial joint can add an extra level of understanding to the diagnosis, which can assist with surgical decision making (Figure 31-11, C). An MRI can provide additional information regarding spinal cord pathology such as hemorrhage or edema and syringohydromyelia, which might be important for prognosis (Figure 31-12, A and B).113,181 The goal of conservative treatment is to stabilize the atlantoaxial junction while the ligamentous structures heal.90 Nonsurgical treatment of atlantoaxial subluxation, including strict cage confinement for 6 weeks, analgesia, and a rigid cervical brace, has been successful in some patients; however, nonsurgical or conservative approaches are likely to result in recurrent or progressive clinical signs.58,82,90 The splint must immobilize the atlantoaxial junction, and so the entire wrap must come over the head cranial to the ears and go back to the level of the chest (Figure 31-13). The splint usually is manufactured out of fiberglass cast material, incorporated into a bandage, and applied ventrally, with the neck held in extension as it is secured in place with cast padding and elasticized wrap. The splint must extend from the rostral extent of the mandible to the xiphoid process. The splint should ideally be changed once a week while the dog is sedated or under anesthesia to avoid the development of pressure sores associated with migration of the splint under the bandage. Repeat cervical radiographs can be taken while the splint is in place and preferably after each successive rewrap. Complications associated with the use of a splint and neck wrap include recurrence of disease, corneal ulcers, migration of the splint to become ineffective, moist dermatitis and decubital ulcers, hyperthermia, respiratory compromise (dyspnea, aspiration), anorexia, otitis externa, and accumulation of food between the splint and the mandible.90 The goal of surgical treatment is to stabilize the atlantoaxial joint, thereby preventing further spinal cord damage. Although the surgery will by design reduce the compressive component of the disease, it will not address any underlying parenchymal disease resulting from the concussion associated with this disease. Surgery should be considered in all dogs, as it holds the potential to fuse the atlantoaxial joint permanently and reduces the chance of catastrophic recurrence. The historical indications for a conservative approach, besides systemic health and economic factors, have included clinical signs of cervical pain only, dogs with mild neurologic deficits, and dogs with no radiographic abnormality of the dens.90 Additional indications for a temporary conservative approach may include very young patients that will benefit from further growth before surgical correction and patients with severe neurologic dysfunction that may show signs of improvement, thereby clarifying the prognosis for the owner.113 Both dorsal and ventral surgical stabilization techniques have been described. Dorsal surgical stabilizing techniques have included the use of wire, wire and polymethylmethacrylate (PMMA), braided suture material, a portion of the nuchal ligament, a metallic retractor, and pins.28,58,79,108,115,120,172 Dorsal techniques provide for decompression, reduction, and stabilization of the atlantoaxial joint unless compression of the spinal cord is due to dorsal deviation of the dens, which remains a contraindication for dorsal stabilization of the joint.167 Osseous fusion generally is not achieved with dorsal techniques because they cannot resist movement in directions other than flexion, resulting in continuous movement of the joint; therefore, it may be more likely that failure of fixation materials is associated with recurrence of clinical signs.215 However, dorsal fixation may be preferable in dogs with small body weight (<2 kg), which may prohibit the use of metallic implants ventrally, and in those dogs in which ventral fixation initially fails.12 Additionally, it has been suggested to be the more biomechanically sound approach to fixation of this joint.108 Ventral fixation methods described include bone plating, pins alone, pins with PMMA, transarticular lag screws, and screws and pins with PMMA.165,181,183,201,206 Additional decompression is rarely required once the vertebrae have been realigned, although concurrent hemilaminectomy has been described.215 Odontoidectomy may be required to decompress the spinal cord in some patients with dens abnormalities.215 The ventral approach has the advantage of providing a means for joint fusion and is the approach of choice for the repair of atlantoaxial fractures. Dogs are positioned in sternal recumbency with towels placed between the surgical table and the neck to slightly elevate the cervical vertebral column. A midline skin incision is made from the external occipital protuberance to the level of the fourth cervical vertebrae, and the epaxial muscles are dissected from the dorsal arch of the atlas and the dorsal spine of the axis. Reduction of the joint can be achieved simply by gripping the cranial aspect of the spinous process of the axis with forceps and applying cranioventrally directed pressure.108 Atlantoaxial Wiring.: A routine dorsal approach to C1 and C2 is made. The periosteum is gently reflected from the arch of the atlas, and dissection of the soft tissues is pursued to open up the vertebral canal at the cranial and caudal margins of the arch of C2. Two holes are drilled in the dorsal spine of the axis. Reduction of the luxation is achieved by depressing the axis ventrally, after which a loop of 20 to 24 gauge orthopedic wire is passed cranially underneath the dorsal arch of the atlas and is retrieved from the atlanto-occipital space. The loop needs to be folded back toward the axis, and at this time risk of iatrogenic trauma to the spinal cord accompanies the necessary manipulation of the implant. If successful, the loop is cut so that two sets of free ends can each be threaded through the predrilled holes in the axis and secured together (Figure 31-14). Instead of being used to secure the vertebrae in alignment, the wire can be used to drag a loop of nonmetallic nonabsorbable suture material back caudally under the atlas, and this material can be tied through the predrilled holes instead.28,139 Nuchal Ligament Technique.120: The nuchal ligament is split longitudinally and is transected at its caudal attachment to T1. Both free ends are then brought from cranial to caudal beneath the dorsal arch of the atlas by retracting them one at a time with a looped wire in the same fashion as the dorsal suture technique. The ends of the ligament are tied to each other through a notch created with a rongeur through the spine of the axis or through a predrilled hole, as for the wire and suture techniques. Dorsal Cross-Pinning.108: Once reduction of the joint is achieved, a 0.045-inch Kirschner wire is driven in a ventrolateral direction across each side of the spinous process of the axis to engage and penetrate the caudal half of the wing of the atlas. Correct orientation of the implant can be facilitated by predrilling a smaller diameter hole in the bone and using a wire passer or grooved instrument (such as a bone gouge) as a guide for the wire. The wires are bent and trimmed to leave about 1 cm protruding from the dorsal surface of the spinous process of the axis (Figure 31-15). The pins are then incorporated into the PMMA. Kishigami Atlantoaxial Tension Band.: Although the Kishigami atlantoaxial tension band (Kishigami AATB) was first described 25 years ago,115 it has only recently become commercially available. The Kishigami AATB further reduces the risk of damaging the spinal cord compared with other dorsal techniques because it rests in the epidural space without crossing the arch of the atlas.115 The atlantoaxial joint is exposed by a standard dorsal approach. Two or three small holes are drilled in the dorsal spinous process of the axis. Malleable stainless steel wire (0.6 to 0.8 mm, 20 to 22 gauge) is threaded through the caudal hole of the axis, and both ends are directed through the next hole, intersecting each other. The dorsal atlanto-occipital fascia cranial to the arch of the atlas is then carefully incised to a width equal to that of the cranial hook of the Kishigami AATB, which is carefully inserted over the dorsal arch of the atlas in the epidural space; this is the most intricate of the steps.167 The spinal process of the axis is pushed down to reduce the joint, assumed to be properly reduced when the spinal process of the axis is lying over and in contact with the atlas. The ends of the wire are now threaded outward through the hooks of the side legs of the Kishigami AATB and are bent caudally. Vertebral alignment and position are checked at this point, and any persistent flexion is reduced by adjusting the length of the wire.167 The excess ends of the wire are cut, and the hooks of the legs are squeezed to ensure stable reduction (Figure 31-16). Dogs are positioned in dorsal recumbency with a towel placed on the table underneath their neck to extend the spinal column. The neck can also be placed within a vacuum positioning device to ensure that movement is limited. The thoracic limbs are extended caudally, and tape wraps should secure the limbs and the rostral skull. A routine approach is made to the ventral aspects of C1, C2, and cranial C3. Care should be taken to avoid damage to vital structures such as the recurrent laryngeal nerve and the vascular supply to the thyroid gland. The ventral aspects of C1 and C2 should be cleaned of muscle, and the joint capsule should be exposed as far laterally as possible. The joint capsule can be incised and the articular cartilage scarified from the surface of both caudal end plates. This can encourage fusion of the joint, which is the primary long-term goal of fixation; this can be assisted by the use of autogenous cancellous bone grafts obtained from the humerus.215 To secure the joint and vertebrae in appropriate alignment while implants are inserted, thereby protecting the spinal cord from repeated concussion, it has been recommended to insert a 1.5 or 2.0 mm cortical bone screw into the caudal part of the body of C2. This screw serves as an attachment point for the orthopedic wire, which is twisted around the head of the screw. The wire is pulled in a caudoventral direction, elevating the bone ventrally toward the operator (Figure 31-17).165 The length of the screw placed in C2 is best estimated from CT or MRI scans. The vertebral body of the axis may be temporarily stabilized with micro Halsted forceps and positioned in the middle of the lateral section of the body, if preferred to the technique described previously.109 Maintaining bone stability by inserting an instrument into the C1-C2 intervertebral space is another option but is dangerous because of the risk of trauma to the spinal cord. Odontoidectomy can be performed through a ventral approach and may be necessary if fracture or nonunion of the dens has occurred.201 Transarticular Lag Screws or Pins. (Figure 31-18): Once the vertebrae have been aligned and are temporarily secured by the previously mentioned methods, a 1.5 mm hole for a 1.5 mm cortical lag screw is drilled in the cranial section of C2, beginning on one side of the vertebral body just behind the bony cranial wall.58 Subsequently, the C1 hole for the screw is drilled using a 1.1 mm drill; the drill is directed craniolaterally, approximately 30 degrees from midline, to reach the largest portion of the bony lateral part of the atlas. The drill hole is tapped with a 1.5 mm tap.109 A similar technique using Kirschner wires or pins (0.045 inch or 0.062 inch) has been described.201 The optimum angle for the pin is obtained by directing each pin toward the medial border of the respective alar notch and maintaining the tip of the pin as far ventral as possible. Pins and PMMA.: Kirschner wires or acrylic fixation pins (0.035 to 0.062 inch) can be placed in both C1 and C2 ventrally, with subsequent incorporation within PMMA.183,193 Following bone grafting and vertebral realignment, the pins are directed perpendicular to the median and transverse planes into each of the pedicles of the atlas with a pin driver. Transarticular pins are also placed as described previously. Pins are then placed into the caudal body of the axis at an approximately 30 degree angle to the transverse plain. All pins are cut 1 to 2 cm ventral to the bone surface, and the ends of the pins are bent before all are embedded in PMMA. Following prolonged saline irrigation, the longus colli muscle can be closed, but this often is not possible. Placement of a biologic membrane (e.g., Vet BioSISt, SurgiVet, Smith’s Medical North America, Waukesha, WI) over the fixation apparatus may encourage local muscle healing and may protect local tissues (e.g., esophagus, trachea) from fibrous adherence. Screws and PMMA.: Cortical bone screws can be placed into the medial aspect of each wing of the atlas caudal to the transverse foramen in a craniolateral direction. Screw sizes should be chosen based on the size of the dog, but 1.5 to 2.0 mm diameters and 10 to 20 mm lengths are usually appropriate.181 A third screw can sometimes be placed in the middle of the ventral body of C1, depending on the size of the patient; this screw can be inserted at 90 degrees to the bone surface and should not break through the second cortical surface.165 This portion of the atlas frequently is entirely cortical in nature. Two ventral C2 screw patterns have been described. In the first pattern, four cortical screws are placed into the body; one screw is placed in the middle of the caudal aspect of each of the cranial articular surfaces of C2 at the insertion point of the longus colli muscle. The screws are directed craniolaterally at 30 to 40 degrees. The second pair of screws is placed at the base of the transverse processes of C2 or C3 and is directed laterally at 30 to 40 degrees to the midline. With this technique, Steinmann pins or Kirschner wires can be used to bridge the screws; the pins or wires are positioned parallel to midline and are secured by orthopedic wire to the screws (Figure 31-19).181 This entire apparatus can be encased in PMMA; as for all ventral techniques, the minimum amount of PMMA necessary to cover all the screws should be used, as it can cause significant compression of surrounding soft tissues. A second pattern uses just two cortical screws, one of which can be the screw that facilitates secure realignment, as described earlier in the technique overview section. The second screw can be placed in the cranial half of the C2 vertebral body.165 This latter technique additionally differs from the first screw and PMMA technique in that transarticular 0.035 to 0.062 inch threaded or nonthreaded Kirschner wires are used to secure the atlantoaxial joint, subsequently incorporating all of the screws and the wires into the PMMA (Figure 31-20, A and B).165 Complication rates have been reported to be as high as 71% for dorsal fixation techniques and 53% for ventral fixation techniques.139,215 Surgical manipulation of the joint is done with the risk of further injury to the spinal cord at the site caused by repeated concussion. This has been reported to be associated with both dorsal and ventral techniques.215 Iatrogenic damage to the spinal cord is also possible as the result of erroneous implant placement. As with all ventral cervical spine surgeries, the risk of laryngeal paresis is due to retraction-related trauma to the recurrent laryngeal nerve. Additionally, compression of the trachea by the implants and by PMMA has been reported to cause tracheal necrosis.215 If this does not cause dyspnea, it may cause an excitement-related cough in some dogs, which can also result from prolonged traction of the trachea during surgery. It is unfortunate that any respiratory pathology often compounds an already present brachycephalic airway syndrome or collapsing trachea in many patients because of the nature of the dog breeds predisposed to atlantoaxial subluxation. In severe cases, dyspnea may need to be managed with a temporary tracheostomy tube. Fatal respiratory arrest is rare, but central nervous system trauma experienced during surgery may be responsible.58,215 Death following perioperative aspiration pneumonia has been documented in some dogs.165 If external coaptation is used before or after surgery, the dog must be monitored extremely closely for the first few hours because there is a risk of respiratory compromise if the bandage material has been placed too tightly. Migration or fracture of the implants can occur in ventral and dorsal surgeries, although it seems to most commonly affect transarticular Kirschner wires and can occur months after the procedure; however, it tends to occur within the first 3 weeks after surgery.12,111,183,215 Transarticular lag screws can also be subject to failure, especially during the implantation procedure, as the small volume of axis bone available for screw engagement and the “target” area for screw positioning do not permit any error.165 Improper pin placement with inadequate bone purchase is the main cause of implant migration.215 This can be an incidental finding at recheck radiographic examinations, or it may become apparent following onset of recurrent clinical signs, or with the appearance of the implant surfacing through the skin or even the mouth.183 Dorsal repairs made with wire can also be associated with breakage of the implant.215 The fixation failure rate may be as high as 25% with dorsal fixation techniques and 18% with ventral fixation techniques.139 Depending on the length of time post surgery when the failure occurred, another surgery may not be necessary, as the joint may have already experienced osseous fusion. The dorsal arch of the atlas can fracture following a dorsal repair because of the weakness of the thin dorsal bone.172,215 Ventral repairs can result in fractures of the cranioventral aspect of the C2 body when a transarticular repair is undertaken, because of improper implant positioning or vertebral immaturity, or both.109 Episodic pain has been reported in dogs for several months to life post surgery and is often a reason for euthanasia of these patients.165,183 This can occur as the result of failure of the surgery to decompress the spinal cord and realign the vertebrae, or it may be due to fixation failure; neurologic status may be improved in some dogs with persistent pain.
Cervical Spine
Clinical Signs and Neurologic Examination of Animals with Cervical Spinal Cord Disease
C1-C5 SPINAL CORD SEGMENT LOCALIZATION
C6-T2 SPINAL CORD SEGMENT LOCALIZATION
Mentation/behavior
Normal; may be difficult to assess if the condition is painful
Normal; may be difficult to assess if the condition is painful
Cranial nerve function
Very rare positional strabismus and/or facial hypesthesia with C1-C3 lesions
Normal
Motor dysfunction
Tetraparesis, tetraplegia, hemiparesis, hemiplegia
Tetraparesis, tetraplegia, hemiparesis, hemiplegia, monoparesis, monoplegia
Spinal reflexes
Intact (normal to increased) in all four limbs
Reduced to absent ipsilaterally or bilaterally in the thoracic limbs; intact (normal to increased) in pelvic limbs; reduction or loss of cutaneous trunci reflex with C8, T1 lesions
Muscle tone
Normal to increased in all four limbs
Reduced to absent in thoracic limbs; normal to increased in pelvic limbs
Muscle atrophy
Disuse atrophy in all four limbs
Neurogenic atrophy in ipsilateral or bilateral thoracic limbs; disuse atrophy in pelvic limbs
Sensory dysfunction
Proprioceptive ataxia affecting all four limbs equally. Postural reactions decreased to absent in all four limbs or in ipsilateral thoracic and pelvic limbs. Deep pain may be absent in tetraplegic animals, but this is very rare.
Proprioceptive ataxia affecting the pelvic limbs more, with the thoracic limbs showing usually mild ataxia.
Postural reactions decreased to absent in all four limbs, in ipsilateral thoracic and pelvic limbs, or in just the ipsilateral thoracic limb with a nerve root or spinal nerve lesion. Cutaneous sensation may be decreased in regions innervated by involved spinal segments and/or their nerves. Deep pain may be absent in animals with nerve root or spinal nerve lesions. Deep pain may be absent in tetraplegic animals, but this is very rare.
Horner’s syndrome
Rarely present
Possible with lesions affecting T1-T3 nerve roots
Voluntary Movement
Spinal Reflexes
Muscle Tone
Muscle Atrophy
Sensory Dysfunction
Neck Pain
Incontinence
Differential Diagnosis of Cervical Spinal Cord Disease
Diagnostic Approach to Cervical Spinal Cord Disease
Surgical Approaches to the Cervical Spine
Ventral Approach to the Atlantoaxial Junction
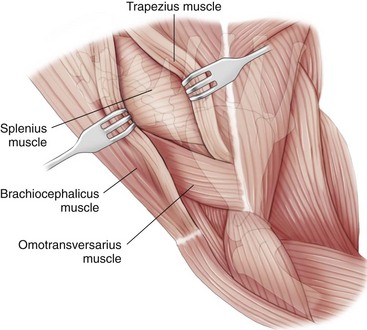
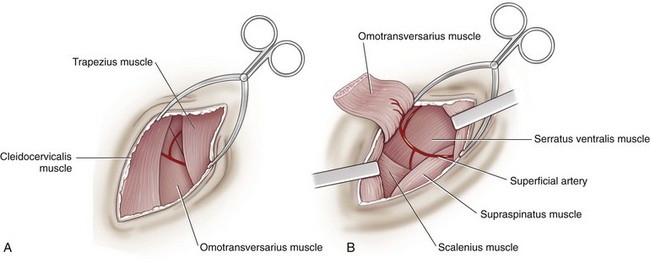
Lateral Approach to the Cervical Spine
Dorsal Approach to the Cervical Spine
Lateral Approach to the Brachial Plexus
Atlantoaxial Instability
Epidemiology and Pathophysiology
Clinical Signs
Specific Diagnosis
Conservative Treatment
Indications for Surgery
Specific Surgical Techniques
Dorsal Techniques
Ventral Techniques
Surgical Complications
Neurologic Deterioration
Respiratory System Compromise
Implant Failure
Fracture of the Atlas or Axis
Recurrent Pain
< div class='tao-gold-member'>
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree