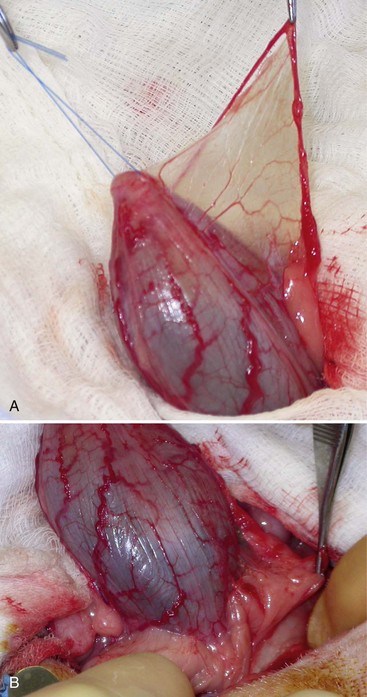
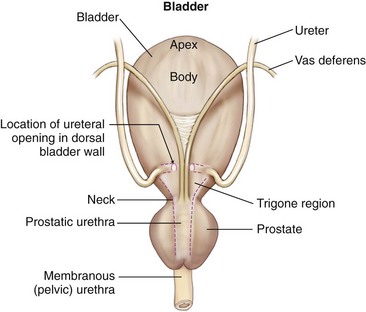
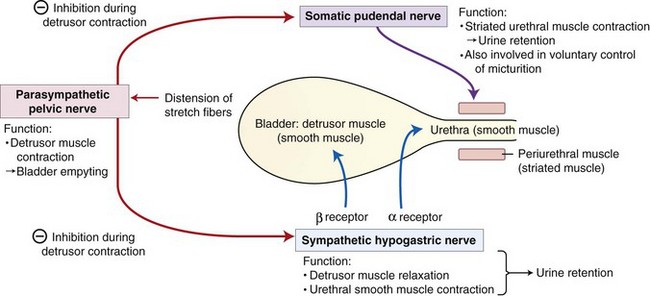
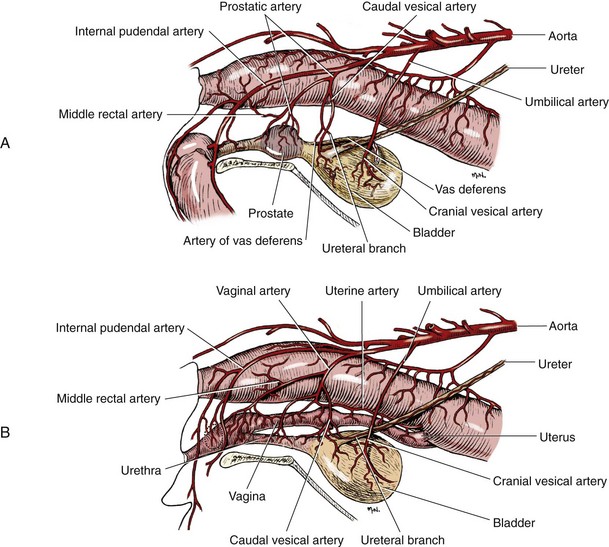
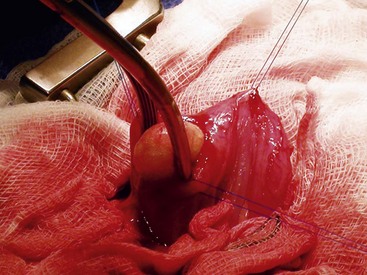
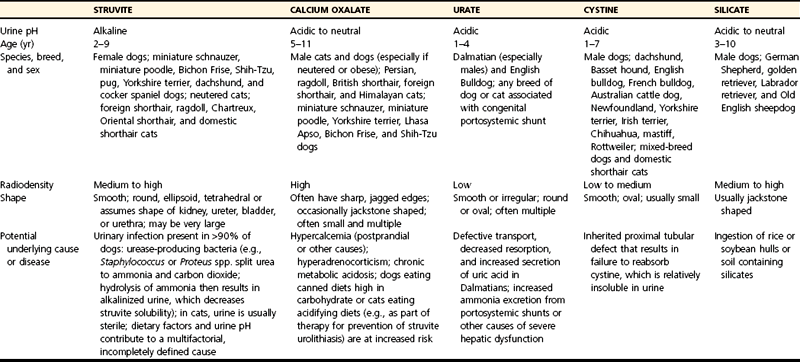
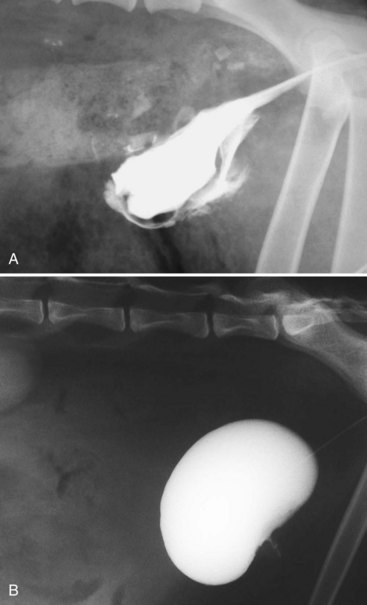
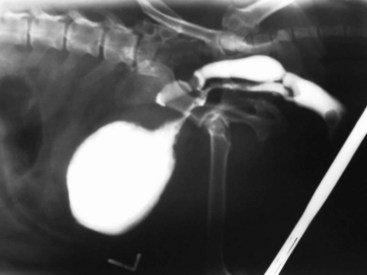
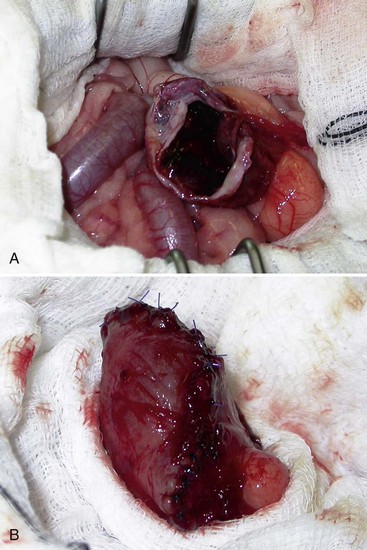
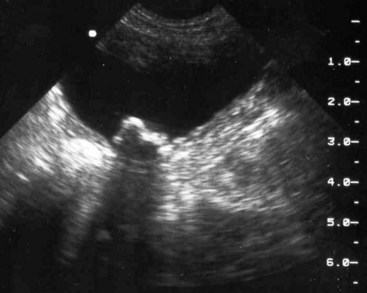
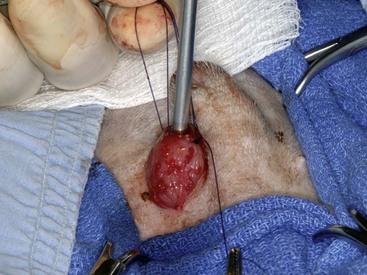
Chapter 116 The bladder lies within the peritoneal cavity and is attached to the abdominal wall via loose, double-layer peritoneal ligaments. The ventral median ligament, which can be cut before cystotomy, is a very thin structure connecting the bladder to the linea alba and pelvic symphysis (Figure 116-1, A). In fetuses, this ligament contains the urachus. The lateral ligaments of the bladder attach to the pelvic walls and contain fat along with the distal portion of the ureter and umbilical artery on each side1 (see Figure 116-1, B). The lateral ligaments should be identified and avoided during dissection around the bladder to prevent iatrogenic damage to the ureters. The bladder is a reservoir for urine and varies in size, depending on the volume it contains. The position of the bladder is also variable, lying just cranial to or within the pelvic canal when empty in dogs and extending cranially along the ventral abdominal wall as it distends.2 The fully distended bladder of a normal dog may be partially located within the pelvic canal. In cats, the bladder remains within the caudal abdomen, even when empty.2 In male animals, the deferent ducts pass dorsal to the bladder; in females, the cervix and body of the uterus are adjacent to the dorsal bladder.1 The bladder has an apex, body, and neck (Figure 116-2). Each ureter tunnels obliquely for a short distance through the dorsolateral bladder wall before opening into the bladder through a narrow, oval orifice. The trigone is the internal region between the ureteral openings in the dorsal bladder wall and the proximal urethral opening at the bladder neck (see Figure 116-2). The urothelium of the bladder consists of mucosa composed of transitional epithelial cells, and submucosa, which contains connective tissue. The normal bladder has a relatively thin wall but becomes thickened when diseased. The urothelium is covered by a layer of smooth muscle (detrusor muscle). The serosa is the outermost layer of the bladder. The detrusor muscle has oblique, interdigitating muscle fibers that are continuous with the smooth muscle of the urethra, so there is no anatomically distinct internal sphincter at the vesicourethral junction.1 Except for during micturition, the smooth muscle at the vesicourethral junction is maintained in a steady state of contraction by sympathetic stimulation of α-adrenergic receptors in the bladder neck and proximal urethra via the hypogastric nerves (Figure 116-3).3 Sympathetic hypogastric nerve stimulation of β-adrenergic receptors in the bladder wall simultaneously relaxes the detrusor muscle, allowing storage of urine (see Figure 116-3).3 The preganglionic branches of the hypogastric sympathetic nerves originate from the thoracolumbar spinal cord. When the bladder nears its capacity, stretch receptors are activated by distention of the detrusor muscle, resulting in parasympathetic pelvic nerve stimulation that initiates reflex detrusor muscle contraction and subsequent urination (see Figure 116-3).4 Activation of the stretch receptors in the bladder wall also depresses sympathetic outflow and acts on the brainstem to cause relaxation of smooth and striated urethral musculature, permitting urination (see Figure 116-3).4 The parasympathetic pelvic nerves originate in the sacral spinal cord (S1-S3) and pontine micturition center. Integration in the brainstem is necessary for the detrusor reflex to be coordinated and sustained long enough for bladder evacuation.4 Not all detrusor muscle cells have direct innervation: transmission of neuromuscular impulses also occurs between one innervated detrusor muscle cell and many adjacent muscle cells.5 Prolonged bladder distension or bladder fibrosis results in loss of this excitation-contraction coupling; subsequent atony of the detrusor muscle results in urine retention and overflow.4 The pudendal nerve originates in the sacral spinal cord (S1-S3) and provides somatic innervation to the periurethral striated muscle at the bladder neck (external urethral sphincter). The external urethral striated muscle, similar to the smooth muscle in this region, remains in a state of steady contraction, contributing to active urethral resistance during urine storage, and is inhibited during reflex urination (see Figure 116-3).3 Additionally, voluntary control can initiate or inhibit the detrusor reflex so that micturition occurs at an appropriate time and place. Voluntary control of micturition occurs via the somatic pudendal nerve innervation of striated urethral musculature and direct cortical innervation of the pontine micturition center.4 All of the innervation to the bladder merges together at the pelvic plexus before entering along the dorsal surface in the region of the bladder neck. There are multiple synaptic connections between the nerves at this level, which facilitates the complex coordination of innervation that is required for maintenance of continence and micturition.6 Similar to the nerves, the vascular supply also enters the bladder dorsally. The caudal vesical artery is the major arterial supply to the bladder. It arises from the uterine (females) or prostatic (males) branch of the internal pudendal artery (Figure 116-4).6 The cranial vesical artery is the terminal ending of the umbilical artery; if it remains patent that far in adult dogs, it also supplies the cranial bladder (see Figure 116-4).6 Bladder venous drainage is into the internal pudendal veins, and lymphatic drainage is via the hypogastric and sublumbar lymph nodes. Wound Healing, Suture Material, and Suture Patterns The normal bladder heals quickly, with mucosal defects healing in 5 days and full-thickness defects regaining 100% of normal tissue strength in 14 to 21 days7. Therefore, many absorbable, synthetic suture materials may be suitable choices for bladder closure. Monofilament suture material is recommended because it causes less tissue drag in delicate bladder tissue than multifilament suture and fewer bacteria adhere to monofilament suture compared with multifilament suture. Nonabsorbable sutures or staples are not required and could predispose to cystic calculi formation.8,9 In one study, a relatively high rate of suture-associated cystoliths was documented as a cause of recurrent urolithiasis (9.4% for dogs and 4% for cats), although the suture materials involved were unknown because of the retrospective nature of the study.8 In an experimental study, calculi were commonly found in rabbits after cystotomy repair with nonabsorbable suture material but not in cats or dogs.9 One theory is that the bladder mucosa and any exposed sutures or staples are quickly reepithelialized in the days after cystotomy, therefore decreasing the risk of suture-associated urolithiasis.10,11 Clinical use of staples for bladder closure has not been reported in cats or dogs, but an experimental study used a linear stapler for cystotomy closure in 52 dogs without penetration of the mucosa by the staples in the majority of staple lines at postmortem examination.10 A study evaluating the tensile strengths of four monofilament suture materials after immersion in canine urine with and without bacteria concluded that polydioxanone and polyglyconate are acceptable choices for bladder closure in sterile neutral and Escherichia coli–contaminated urine but that poliglecaprone 25 may not have sufficient tensile strength during the critical phase of bladder healing.12 This study also found that all four suture materials disintegrated before day 7 in Proteus mirabilis–inoculated urine, prompting a recommendation to minimize exposure of suture that degrades via hydrolysis during bladder surgery in cases in which urine contains Proteus spp.12 Another earlier study supports the finding that synthetic, absorbable suture materials quickly lose tensile strength in P. mirabilis–inoculated urine but found that polydioxanone was superior to polyglyconate in sterile and E. coli–inoculated urine.13 Bladder neoplasia or trauma could also have an adverse effect on bladder healing, and consideration should be given to selecting one of the longer lasting absorbable monofilament sutures in such situations. The recommended suture size for use in viscera in small animal surgery ranges from 1 to 2 metric (3-0 to 5-0), with the final choice for bladder surgery depending on the size of the animal, thickness of the bladder wall, and strength of the suture material selected. A curved, swaged-on needle with a taper point is recommended for use in delicate tissue such as the bladder. Many suture patterns have been reported in the veterinary literature for cystotomy or cystectomy closure, but there is no consistent evidence as to which performs best overall. Whichever pattern is used, accurate needle placement through the strength-holding submucosal layer of the bladder wall is critical. Ideally, the suture pattern should not penetrate the mucosa to avoid producing a potential nidus for calculi formation8,9 or exposing the suture to infected urine, which could result in premature loss of tensile strength.12,13 This may be difficult to achieve in thin-walled bladders (especially given that engagement of the submucosa with every suture is essential). An experimental study in dogs suggested that a continuous, extramucosal, running horizontal mattress suture closure was ideal because it was stronger and associated with less urine leakage, inflammation, and adhesions than a two-layer closure incorporating the mucosa.14 A single-layer, simple, continuous appositional closure is quick, provides accurate apposition of the strength-holding submucosal layer and is suitable for most cystotomy closures. A study comparing single-layer, simple, interrupted appositional and two-layer, inverting, continuous suture patterns found no difference in strength in the first 24 hours.15 Interrupted appositional sutures may be useful when closing irregularly shaped bladder defects after bladder resection. Augmentation of the closure by omentalization is simple and routinely performed by many surgeons. If there is concern about urine leakage postoperatively in a thin-walled bladder, a second layer inverting continuous suture pattern (Cushing or Lembert) may be used. Alternatively, in a thickened, diseased bladder, a serosal patch may be used to augment a cystotomy closure. Surgery that involves entry into the bladder in the absence of infected urine is classified as clean contaminated. The infection rate for this class of surgery in small animal veterinary patients is approximately 5%, and the administration of prophylactic antibiotics does not reduce this rate unless the duration of surgery is prolonged.16 In the absence of controlled studies investigating risk factors for postoperative infection after lower urinary tract surgery, prophylactic antibiotics may be considered when there is urinary tract obstruction, urine leakage, traumatized tissues, or renal failure. Most urinary tract pathogens originate from the surface of the skin or the gut and ascend from the urethral orifice (e.g., E. coli, Proteus spp., Staphylococcus intermedius)17. A broad-spectrum, bacteriocidal, non-nephrotoxic antibiotic, such as amoxicillin clavulanate, or a third-generation cephalosporin is recommended for surgical prophylaxis. The antibiotic should be given intravenously (IV) at induction of anesthesia, repeated every 2 hours during surgery, and discontinued at the end of surgery.18 If a urinary tract infection is present, a course of postoperative antibiotics is indicated based on the results of culture and sensitivity testing. Antibiotics may be withheld until intraoperative cultures have been taken. Suitable empirical antibiotic choices for common urinary tract pathogens include amoxicillin clavulanate, third-generation cephalosporins, and enrofloxacin.17 If the bladder is full, then it may be emptied before surgery using a urethral catheter. Alternatively, it may be emptied intraoperatively using a needle and syringe or by suction after initiation of a cystotomy incision. Gentle manipulation of the tissues during bladder surgery is very important because the bladder urothelium quickly becomes edematous and congested in response to surgical handling. Stay sutures are the most practical and least traumatic way of manipulating the bladder (Figure 116-5). Use of moistened swabs is less traumatic than dry swabs. Suction is essential to remove urine, increase visualization, and reduce the need for repeated use of swabs on the surgical field. The tissues should be kept moist during surgery, and the use of diathermy on the bladder should be avoided when possible because it tends to produce charring, which will delay healing. Fine mosquito forceps applied to the tip of a vessel and ice-cold saline irrigation are less traumatic alternatives for hemostasis. Use of atraumatic (e.g., DeBakey) forceps will minimize tissue crushing. A scalpel incision or an incision made with high-quality, sharp scissors (e.g., Metzenbaum) will also minimize tissue trauma. A surgical assistant, if available, helps to ensure the tissues are retracted atraumatically and reduces surgical time. A pair of abdominal retractors (e.g., Gosset) is necessary to provide appropriate access to the bladder at exploratory laparotomy. Cystocentesis is by far the most accurate method of obtaining urine for bacterial culture and cytologic evaluation. Investigation and treatment of concurrent urinary tract infection before or after bladder surgery are important to reduce the risk of complications and maximize the likelihood of a successful outcome. Transitional cells are a normal finding, but neoplastic cells may also be identified on cytology. The pH, crystal type, and presence of urinary tract infection may help guide the prediction of calculi type (Table 116-1). Cystocentesis is useful to decompress a distended, obstructed bladder that cannot be catheterized to help stabilize an animal before further investigation and treatment. It is performed easily and safely in most dogs and cats with a 22- or 24-gauge needle and a syringe. If necessary, cystocentesis can be performed with ultrasound guidance. Bladder ultrasonography is the most sensitive method of determining the location and size of the bladder, as well as identifying calculi and bladder wall lesions, thickening, or abnormalities (Figure 116-6). The origin and extension of bladder masses can be estimated, and ultrasonography can be used to facilitate catheter biopsies of the mass. Ultrasonography is also used to guide cystocentesis or abdominocentesis when uroabdomen is suspected. Figure 116-6 Ultrasound image of a bladder highlighting a calculus resting in the lumen with acoustic shadowing below it. Contrast radiography is often used to detect or define the extent of bladder or other urinary tract disease. The location of urinary tract rupture must always be identified using contrast radiography before surgery. A positive contrast cystogram result should reveal the presence of bladder rupture (Figure 116-7, A), identify radiolucent calculi, and outline any bladder masses. Small bladder tears may be difficult to visualize on the initial cystogram, and if there is an index of suspicion, a repeat cystogram or serial radiographs may be required to demonstrate the presence of free contrast material within the peritoneal cavity (Figure 116-7, B). A retrograde urethrocystogram, rather than a cystogram, is selected when the lower urinary tract rupture location is not known. A double-contrast cystogram produces enhanced mucosal detail and is recommended for highlighting bladder wall lesions, intraluminal masses, and cystic calculi. For extensive bladder neoplasia, an IV urogram or a retrograde urethrocystogram may also be required to establish whether there is involvement of one or both ureters or extension into the urethra (Figure 116-8). There are few drawbacks to performing contrast radiography, but urinary tract rupture is possible in animals with advanced neoplasia or nonviable bladder tissue secondary to prolonged complete urethral obstruction. Computed tomography (CT) or magnetic resonance imaging (MRI) of the bladder is not routinely performed in animals. CT (including excretory urography) has been evaluated in healthy dogs,19,20 dogs undergoing laparoscopically implanted radiotherapy for the treatment of bladder neoplasia,21 and dogs with suspected ureteral ectopia.22 Postcontrast MRI and CT studies both demonstrate a “layering effect” in which a dependent layer of contrast-opacified urine forms beneath a layer of noncontrast–opacified urine.23,24 Tissue from bladder and urethral masses is easy to obtain noninvasively by applying suction using a syringe via a urinary catheter that has been advanced to lie at the level of the mass.25 Using ultrasound guidance is recommended because it will verify the position of the mass next to the catheter tip, and the ultrasound transducer can be used to carefully push the bladder mass against the catheter tip. This technique was performed in 12 dogs with minimal complications and resulted in a histologic diagnosis in 10 of the 12 dogs.25 Cystoscopy provides a direct, magnified view of the mucosal surfaces of the bladder and urethra. It is a useful adjunct to other routine diagnostic investigations of the bladder and may also permit further interventional diagnostic or therapeutic procedures. Cystoscopy can be performed in both dogs and cats using a rigid cystoscope in females and prepubic percutaneous or flexible cystoscopy in males.26,27 Laparoscopic-assisted cystoscopy can be performed in any animal, and the cystotomy incision can be used to remove urinary calculi or to biopsy or resect a bladder lesion.28,29 During laparoscopic-assisted cystoscopy, traction sutures are used to secure the bladder to the abdominal wall before cystotomy and examination of the bladder with the cystoscope (Figure 116-9). Cystoscopy requires general anesthesia as well as training in use of the equipment and interpretation of the image. Details of the equipment, patient preparation, and examples of the images that can be obtained have been published.26,27 Cystoscopy is a relatively safe procedure, but urinary tract infection or perforation is possible.26,27 Figure 116-9 Picture of laparoscopic-assisted cystotomy. (Courtesy Dr. Karen M. Tobias, University of Tennessee, Knoxville, TN.) Lesions or conditions that may be identified using cystoscopy include lateralization of hematuria of renal origin, ectopic ureters, bladder or urethral neoplasia, bladder diverticulum, cystitis or urethritis, polypoid cystitis, bladder or urethral rupture, urethral stricture (before and after balloon dilatation), and urolithiasis.26,27 Cystoscopy may identify cystic calculi too small to be detected by other methods and is useful in the assessment of calculi size with regards to suitability for voiding hydropropulsion. The bladder can then be reexamined after hydropropulsion to check for residual calculi. For an experienced operator, ectopic ureters are more accurately diagnosed with cystoscopy than contrast radiography.30 Interventional techniques that may be performed using the aid of a cystoscope include retrieval of calculi through the cystoscope using a basket or an Ellik evacuator,68 biopsy of bladder tissue or masses through the cystoscope (although these biopsies are very small),27 removal of cystic calculi or biopsy or resection of bladder masses using laparoscopic-assisted cystoscopy,28,29 lithotripsy of bladder calculi,31,32 laser ablation of ectopic ureters,33 and urethral submucosal injection of collagen to treat urethral sphincter mechanism incompetence.34 Nonsurgical biopsy of bladder masses is particularly useful when attempting to differentiate transitional cell carcinoma from inflammatory polyps. Laser lithotripsy is a rapidly developing veterinary field (see section on urolithiasis). Indications for cystotomy include removal of calculi, repair of bladder trauma, biopsy or resection of bladder masses, biopsy and culture of the bladder wall in severe cystitis cases, repair of ectopic ureters, and inspection or catheterization of the ureters in the investigation of idiopathic renal hematuria. A midline laparotomy incision is made from the umbilicus to the pubis (curving parapreputially through the skin and subcutaneous tissue in male dogs). The bladder is identified and isolated from the rest of the abdomen with moistened laparotomy swabs, and a stay suture is placed in the apex (see Figure 116-1, A). There is no difference in the likelihood of urine leakage, adhesions, or calculi formation between dorsal or ventral cystotomy approaches, but a ventral cystotomy is easier to perform, provides good visualization of the ureteral openings, and reduces the risk of iatrogenic damage to the ureteral openings.35,36 The cystotomy incision is performed approximately in the midline, but the surgeon should choose the least vascular region. A stab incision is made into the bladder using a scalpel blade, and residual urine and blood are removed using suction. The incision is extended cranially or caudally (or both) as needed, and further stay sutures are placed at the edges of the cystotomy incision (see Figure 116-5). The bladder lining is inspected, and any abnormal areas are biopsied. The easiest place to excise a full-thickness bladder biopsy is at the edge of the cystotomy incision; samples may be submitted for microbiology as well as histopathology. Routine closure of the linea alba, subcutaneous tissue, and skin is performed, including reapposition of the preputialis muscle in male dogs. The major complication of cystotomy is uroabdomen, which manifests as depression, vomiting, or other clinical signs secondary to the progressive metabolic changes occurring (see section on bladder rupture). Dehydration, metabolic acidosis, and hyperkalemia must be corrected before reoperation to identify and correct the reason for cystotomy failure. Indications for partial cystectomy include excision of a patent urachus, bladder diverticulum, discrete bladder neoplasia or polyp, and bladder necrosis (Figure 116-10). If a large amount of bladder has been excised, a temporary cystostomy tube or an indwelling urethral catheter may be placed to maintain bladder decompression in the postoperative period. If the cystectomy includes one or both ureteral orifices, then a ureter can be cut and reimplanted into a different location in the bladder wall. The same conclusions for closure of cystotomy wounds (see above) can be applied to cystectomy incisions, although large or awkwardly shaped cystectomy incisions may benefit from placement of at least some interrupted sutures rather than one continuous suture pattern. The neurovascular supply to the bladder enters dorsally in the trigone region, so it is simpler and carries much less risk of urinary incontinence if a cystectomy can be performed without involving this area. In human surgery, more than 75% of the bladder may be excised (excluding the trigone) and closed over a 5-mL Foley catheter with development of a 300-mL bladder capacity within 3 months.37 Bladder capacity increases after partial cystectomy by a combination of mucosal regeneration, synthesis, and remodelling of scar tissue; hypertrophy and proliferation of smooth muscle; and distension of the remaining bladder wall.37 Leaving the trigone intact is important for bladder mucosal regeneration because the regenerating cells arise from the epithelium of the terminal ureters and urethra.38 It has been suggested that, similar to the situation in humans, dogs may also regain normal bladder function in the months after a large partial cystectomy.39 However, the exact percentage of bladder that may be excised in dogs without resulting in long-term pollakiuria or incontinence is unknown. The outcomes of a control group of experimental dogs undergoing a 35% to 40% cystectomy revealed that all had regained their baseline bladder capacity by 10 months.40 However, in a more than 90% cystectomy dog model (trigone left intact), the mean bladder capacity of the control dogs was still 72% decreased from baseline at 9 months, and all dogs had increased urinary frequency.41 In a clinical veterinary study, 40% to 70% of the bladder was excised in 11 dogs for the treatment of bladder neoplasia, including unilateral ureteral stoma excision with ureter reimplantation in four dogs and bilateral ureteral stoma excision with ureteral reimplantation in one dog.42 None of these dogs was incontinent postoperatively, but two dogs had persistent pollakiuria. En bloc removal of the bladder neck and proximal urethra with preservation of the dorsal neurovascular pedicles has been described in two dogs and did not result in urinary incontinence or bladder wall necrosis.43 Single case reports of more than 90% bladder necrosis successfully managed with a seromuscular colonic augmentation procedure (the external wall of the colon became the cranial wall of bladder)44 and an ileocystoplasty procedure45 have also been published. A dog with proximal urethral and trigone necrosis has been managed successfully using a rectus abdominis muscle flap.46 Total cystectomy with urinary diversion has been performed for the treatment of transitional cell carcinoma in dogs47 but is no longer recommended because of the high postoperative morbidity. Although enterocystoplasty techniques have traditionally been the mainstay of bladder augmentation in human surgery, various materials have been used as a scaffold for bladder regeneration after cystectomy, including small intestinal submucosa. Porcine small intestinal submucosa has been used successfully in experimental dogs in a 40% cystectomy model but not in a 90% cystectomy model, suggesting that the extent of the bladder injury is important, with too extensive a cystectomy resulting in significant inflammation and a lack of rapid neovascularization that produces graft failure.40,41 The latest development in human surgery is tissue engineering to produce new bladder tissue using autologous cells, which has been used successfully to augment bladder defects in experimental dogs.48
Bladder
Anatomy
Innervation
Blood Supply and Lymphatic Drainage
Bladder Surgery Principles
Antibiosis
Operating Technique
Diagnostic Techniques
Diagnostic Imaging
Catheter Biopsy
Cystoscopy
Surgical Techniques
Cystectomy
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree