24 Assessment of nutritional status from analysis of blood and other tissue samples
In theory, feed analysis and ration calculation is the method of choice to evaluate adequate nutrient intake or to detect nutrient deficiencies or excesses (see Chapter 23). There are, however, certain obstacles in the path of those who want to use this method. Feed analysis may be costly and time consuming and quite often it is difficult or impossible to obtain an accurate nutritional history, which is a condition sine qua non. Owners may just forget or disregard the supplement, which is the culprit in cases of excess, or, in cases of deficiency they may not know that the horse does not eat its supplements. In forensic cases, an inaccurate nutrition history may even be given deliberately. There may be situations where the ration is well known but still additional information on the nutritional status of the horse may be helpful. Also, horse owners and trainers often assume that analysis of a blood sample is a fool proof and highly efficient method for the assessment of a horse’s nutritional status, and ask their veterinarian to perform such analyses. A prime example is that in practice any decline in red cell indices is often misinterpreted to be due to an insufficient iron intake. The hematocrit and blood hemoglobin are commonly used to evaluate iron status in horses; however these parameters does not provide information on iron status. In reality, iron deficiency anemia is very rare in horses and most often arises secondary to severe (external) blood loss (Hinchcliff et al 2008).
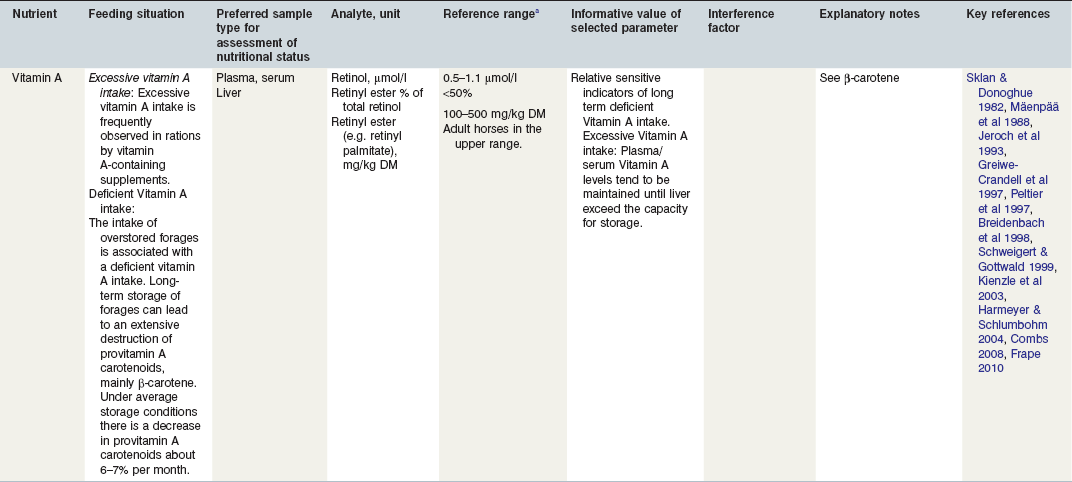
Tables 24-1–24-4 provide the authors’ summary recommendations on sample types and parameters which may be helpful in assessing nutritional status in horses, specifically protein, macrominerals, trace elements, and vitamins (further discussion on the use of urine, hair, hoof horn, or liver samples for nutrient analysis is presented below). This nonexhaustive list also mentions certain parameters that the authors believe are not very useful, but which are nevertheless analysed quite often. The typical practical situations for the respective nutrient excess or deficiency are described. Important interferences and sources of errors are given, as well as reference ranges. According to different methods of analyses, a certain variation is possible, and it is recommended to check with the chosen laboratory. Comments are also made on the informative value of the selected parameter. The informative value of analytical findings can differ when comparing results from a single animal vs. a number of animals in a herd. Thyroid hormone concentrations, for instance, are not a good indicator of iodine nutrition in a single animal as hormone concentrations can be below reference values for reasons unrelated to iodine intake (Wehr et al 2002). On the other hand, low hormone concentrations in a large number of animals from a single herd may indicate iodine deficiency.
Table 24-1 Factors in Nutritional Assessment of Protein
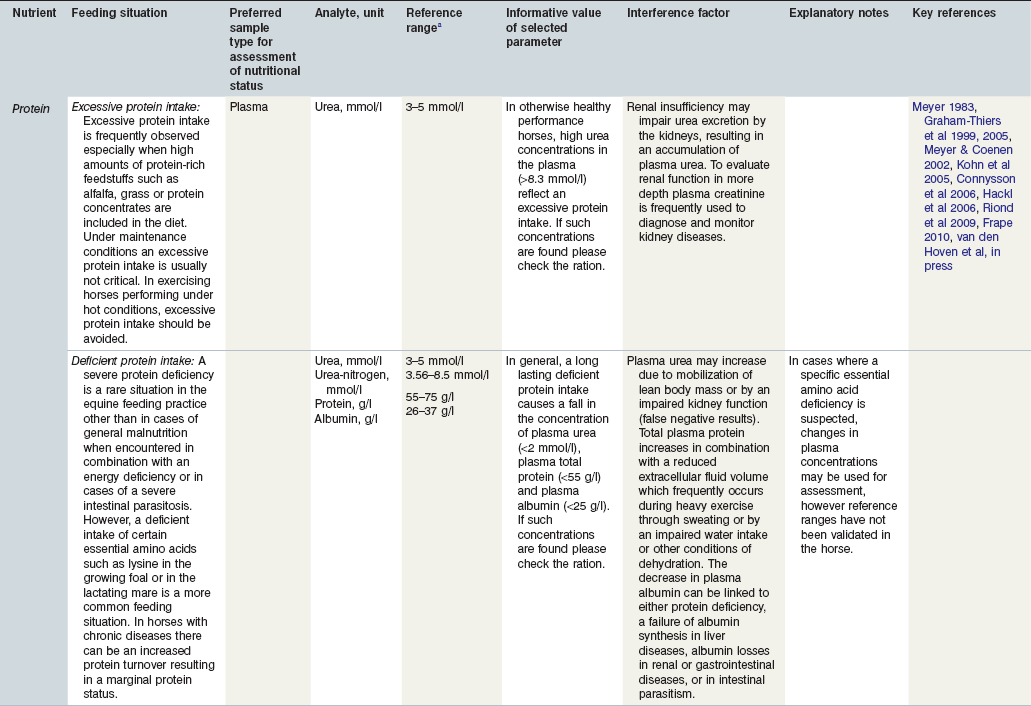
a Reference ranges: Most labs have specific reference ranges which have to be considered.
Table 24-2 Factors in Nutritional Assessment of Minerals
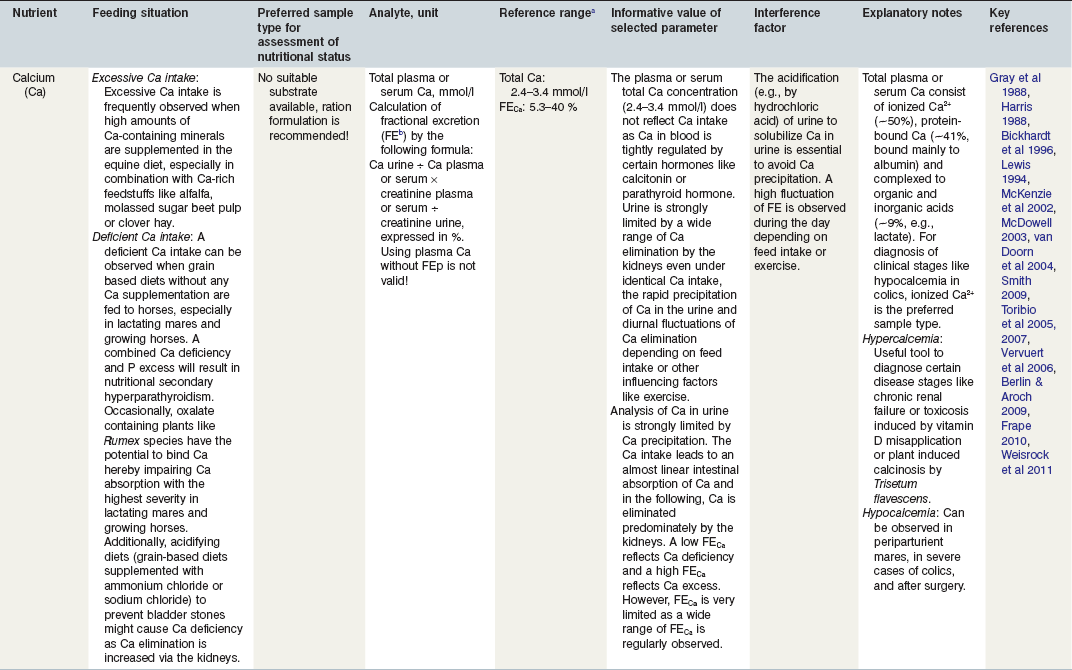
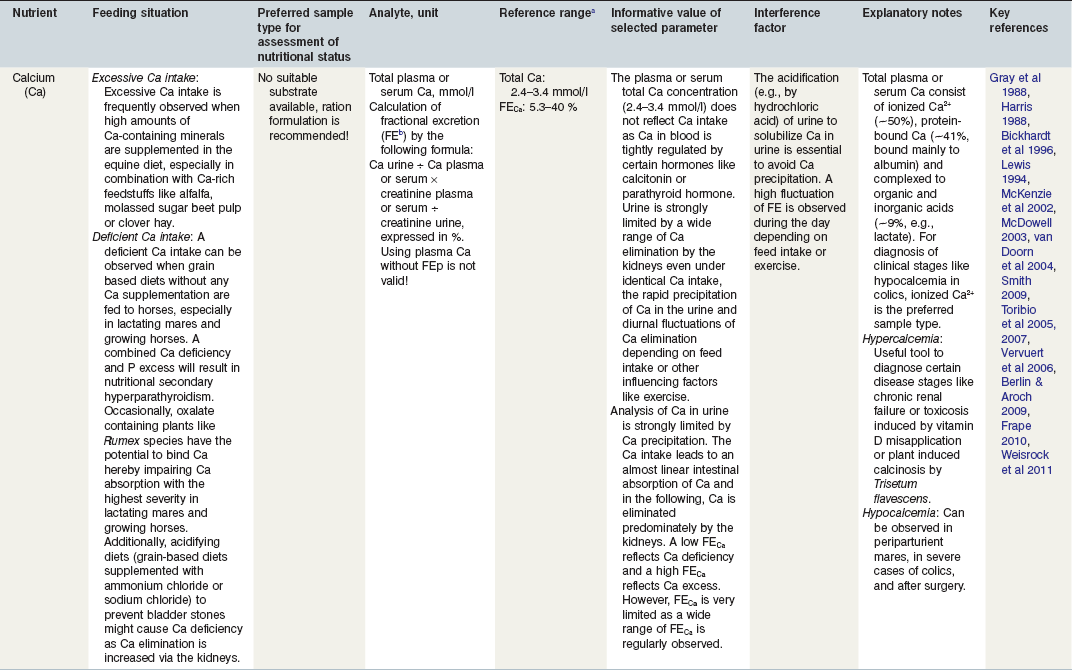
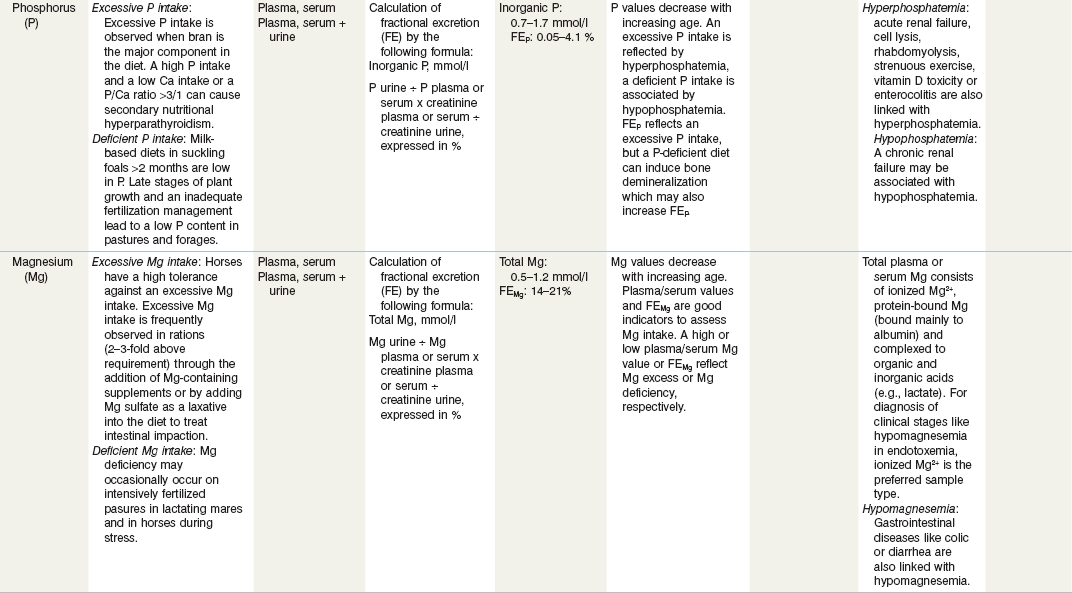

a Referance ranges: Most labs have specific reference ranges which have to be considered.
b FE: Fractional excretion describes the percentage of the respective filtered electrolyte by the kidneys (e.g., Ca, P, Na) which is excreted in the urine. Recommendation for urine sampling: Constant diet for at least 14 days; use freely voided urine sample, preferential 8 h after feed withheld and pre-exercise. Urine creatine >9 mmol/l and urine pH > 7. A standardized protocol according to Harris (1988) is recommended for scientific purposes.
Table 24-3 Nutritional Assessment of Metal Ions
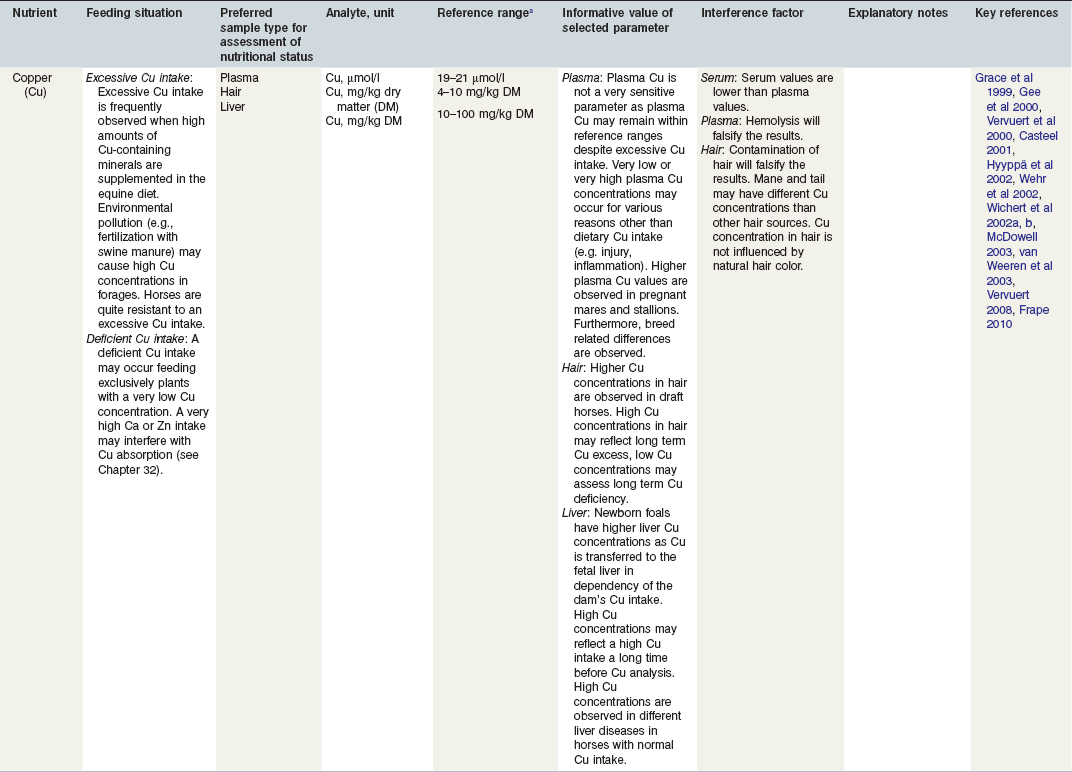
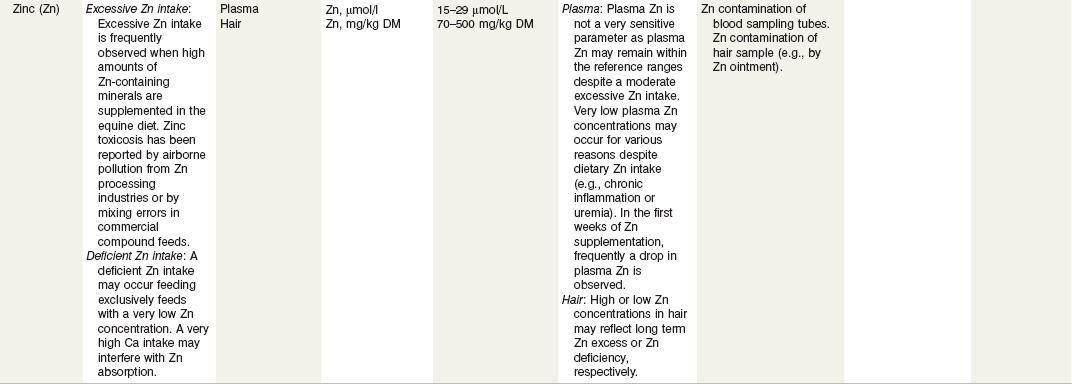
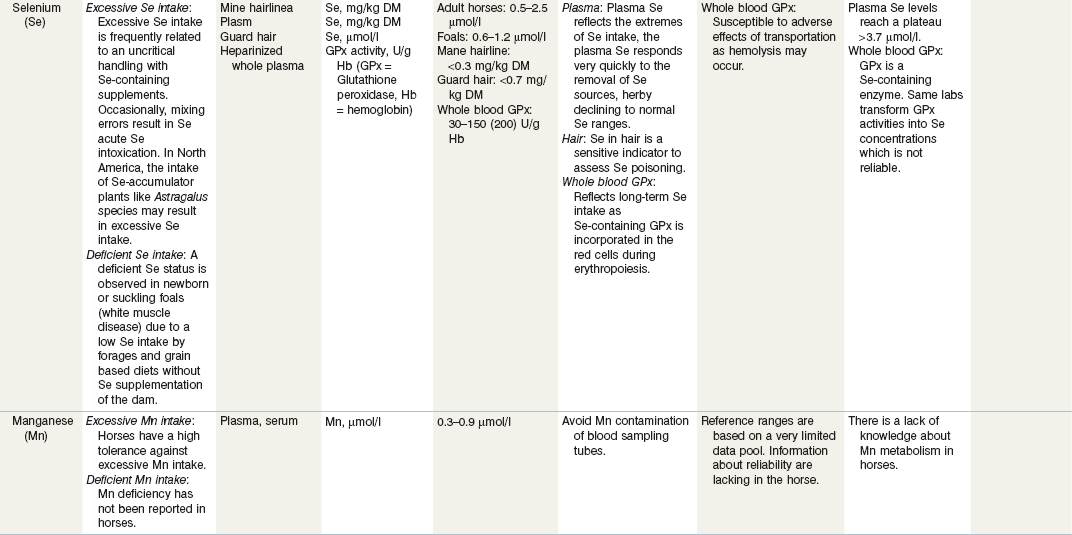

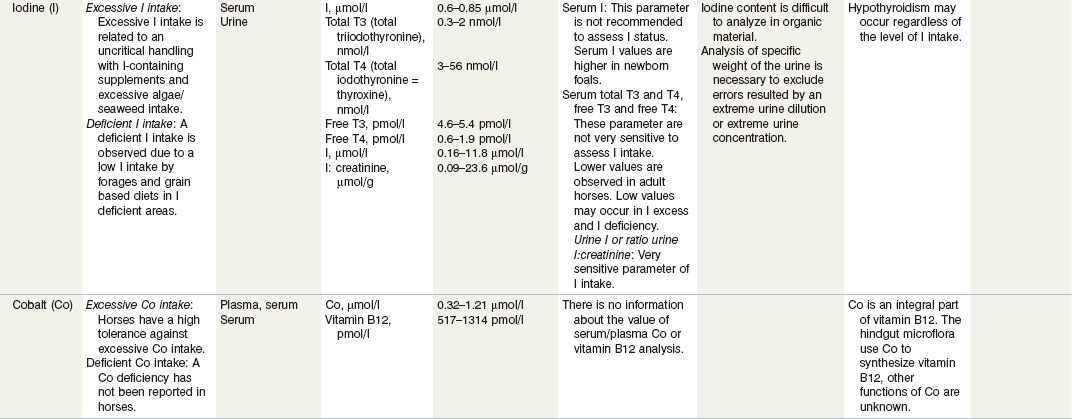
a Reference ranges: Most labs have specific reference ranges which have to be considered.