8 Carbohydrates
The horse evolved primarily as a grazing and browsing, hind-gut fermenting herbivore with a wide range of forage carbohydrates – hydrolyzable to fermentable – as its main source of energy. Pastures provide the main habitat and nutrition for most horses, and the remaining stall-confined horses should have at least one-half of their dry matter intake supplied by conserved pasture. Horse owners supplement a diet of pasture and hay with energy-dense feedstuffs that often contain substantial quantities of cereal grains in order to meet energy demands of performance and to provide a carrier for micronutrients that are marginal or deficient in forages. Common experience has been supported by epidemiological and experimental studies that associate grain concentrates with several digestive and metabolic disorders, including colic (Clarke et al 1990, Tinker et al 1997, Hudson et al 2001), laminitis (Pass et al 1998, Hoffman et al 2007), gastric ulcers (Murray 1994), developmental orthopedic disease (Kronfeld et al 1990, Ralston 1996), insulin resistance (Hoffman et al 2003a, Treiber et al 2005) and polysaccharide storage myopathy (Valentine et al 2001, Ribeiro et al 2004). The abundant starch in grain concentrates has been implicated as the “culprit”, leading to development and marketing of “low starch” concentrates for horses. Corresponding trends in human nutrition towards “low carb diets” have fed wide, consumer support for low starch feeds for horses, perhaps to excess.
While low starch complementary feeds (“concentrates”) provide an alternative energy source that is critical for horses with a history of digestive and metabolic disorders that are sensitive to dietary starch load, these feeds are not a “one fits all” solution, as horses performing high intensity exercise may require some dietary starch in order to appropriately fuel performance. Horses have an opportunity for small intestinal metabolism of starch and simple carbohydrates to glucose, which is more metabolically efficient than hindgut fermentation of fibers to volatile fatty acids (Blaxter 1989, Kronfeld 1996). Compared to fatty acids, glucose (or its stored form glycogen) is aerobically metabolized nearly twice as fast to generate ATP for muscle contraction. As speed and exertion increase to the point of anaerobic work, glucose from glycogen is favored metabolically over fatty acids. Thus, in spite of the evidence that many horses perform well on high-forage, low-starch diets, some may not, and it should be noted that the critical lower limit of starch intake, especially for high intensity performance, is not known.
Carbohydrate nomenclature
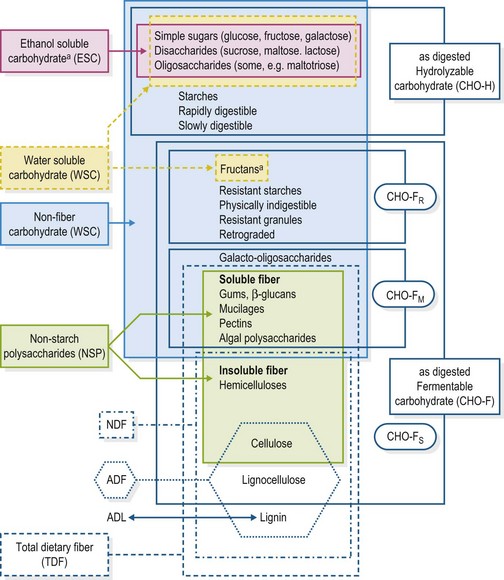
From the perspective of plant physiology, carbohydrates may be divided into three general groups: simple sugars, polymeric storage molecules (e.g., starch, fructans), and structural polysaccharides (e.g., hemicelluloses, cellulose). From the perspective of equine digestive physiology, carbohydrates may be divided into two major groups: those that can be hydrolyzed to simple sugars in the small intestine, and those that cannot be digested by mammalian enzymes, but instead undergo bacterial fermentation to volatile fatty acids. Carbohydrates with α-1,4 glycosidic linkages are subject to enzymatic hydrolysis (although they may also be fermented), while β-1,4 linked molecules must be fermented. Specific descriptions of carbohydrates include the following descriptions and are noted in the scheme of carbohydrate fractions (Fig. 8.1).
Fructans, or oligo- and polyfructosyl sucrose polymers, contain a single sucrose starter molecule attached to a variable number of fructofuranose units (<10 = oligo; >10 = polyfructosyl); polymeric fructans may contain several thousand fructofuranose units. Depending on the polymerization, fructans may be considered either oligosaccharides or polysaccharides. Some fructans are linear with β-2,1 or β-2,6 glycosidic bonds, while others are branched, similar to starch. The two most common fructans in equine forages are inulins, generally linear with β-2,1 glycosidic bonds, and levans, linear with β-2,6 glycosidic bonds. A third type is graminan, a branched fructan containing both β-2,1 and β-2,6 glycosidic bonds, common in wheat and barley straw (Ritsema & Smeekens 2003). All are resistant to mammalian enzyme hydrolysis. Fructans are the main storage carbohydrate in cool season grasses and may comprise 5 to 40% of the dry matter in these grasses, depending on season and sunlight (Longland et al 1999, Cuddeford 2001, Harris & Geor 2009).
Starch is a polysaccharide containing glucose units linked by glycosidic bonds, including linear amylose with α-1,4 linkage and the branched amylopectin with α-1,6 linkages. Starch is produced in all plants but is the primary storage carbohydrate in grains, warm season grasses and legumes. Starch is subject to enzyme hydrolysis in the equine small intestine, but can be rapidly fermented. Like fructans, plant starch content is influenced by season and sunlight (McIntosh et al 2007a, b). Resistant starch is starch that is resistant to small intestinal enzyme hydrolysis, either due to physical or chemical structure.
Hemicellulose includes several polysaccharides present in plant cell walls, most of which are polymers containing xylose, glucose, mannose, and arabinose, also known as xylans, glucomannans and arabinans. Most hemicelluloses are soluble in acid detergent and relatively easily and slowly fermented in the equine hindgut. It is generally approximated by concentrations of neutral detergent fiber (NDF) minus acid-digestible fiber (ADF) (see proximate analysis), and considered to represent insoluble fiber.
Sources of dietary carbohydrate and relevance to equine health
Forages
When the production of sugars exceeds the energy requirements of the plant, they are converted to storage carbohydrates, most commonly starch or fructans. Cool season pasture grasses accumulate mainly fructans, while warm season grasses and legumes accumulate starch. The accumulation of storage carbohydrates in plants is affected by temperature, light intensity and plant growth rate (Longland et al 1999, Hoffman et al 2001; see Chapter 18). While plants that accumulate starch are limited to maximum storage when their chloroplasts are saturated, plants that accumulate fructans have no self-limiting mechanism, so high concentrations may accumulate.
Abrupt changes in fructan concentrations were observed from day to day in rapidly growing pastures and diurnally as plant composition changed from night to day or from shade to sunlight (Longland et al 1999, Longland & Byrd, 2006, McIntosh et al 2007a, b). Fructan concentrations usually rose during the morning, peaked in the afternoon, and declined to a low overnight until the early morning hours. Horses grazing in the afternoon, as compared to morning, may ingest between two to four times as much fructan (Longland et al 1999). See additional notes regarding fructans below and in Chapter 27.
Forages are the main component of equine diets, ranging from providing approximately 50% to 100% of the energy intake. There is currently no specific requirement for fiber (NRC 2007); rather, recommendations are made in order to avoid problems associated with under-provision of fiber, including colic (Clarke et al 1990, Tinker et al 1997), gastric ulcers (Murray 1994, see also Chapter 34), acidosis in the hindgut (Argenzio et al 1974, de Fombelle et al 2001, Medina et al 2002), and stereotypic behaviors such as cribbing (Gilham et al 1994). Plant fibers in horse nutrition are designated either by nomenclature (see above) or proximate analysis components (see below).
It has been generally assumed that voluntary forage intakes of horses sufficiently match their energy requirements; however, numerous studies indicate that intake of various forages and hay is highly variable and influenced by palatability, plant maturity, plant species and composition, time of day in which the hay was harvested (hence sugar/starch/fructan content), grazing patterns, interaction with other horses, and weather (Dulphy et al 1997a, b, MacKay et al 2003). Although NDF concentration has been highly correlated with voluntary intake of forages in cattle, voluntary dry matter intake in horses was not influenced by NDF of forages (Dulphy et al 1997b). See Chapter 3 for more information on factors affecting feed intake.
Grains and grain processing
It is common practice for horse owners to supplement forages with grain-based feeds in order to meet energy demands of performance and to provide a carrier for micronutrients required by horses. Although a wide variety of grains may be fed to horses, the most commonly used are oats, corn and barley. Oats and barley contain less starch and more fiber than corn (NRC 2007), but compared to corn and barley, oat starch appears to be more digestible in the small intestine (Radicke et al 1991, de Fombelle et al 2004).
Starch digestion is impeded when the physical form of the food limits contact with pancreatic amylase. This occurs if the starch is contained within whole grain or waxy seed coats, such as rice or corn, entrapped within rigid cell walls that hinder swelling and dispersion of the starch, such as soybeans, or if the starch is densely packed, which is more typical in human foods, such as pasta, than horse feeds. Such starch has been termed resistant starch by human nutritionists (Eerlingen et al 1993, Englyst et al 1996). Milling and grinding increased susceptibility to hydrolysis in vitro by breaking the seed coats and cell walls, as well as abrading the surface of starch granules at the microscopic level, which is smooth in its natural state (Gallant et al 1992). Preileal digestibility of starch was improved in horses when oats (slightly) or corn were ground, while rolling and breaking did not improve digestibility over that of whole grain (Kienzle et al 1992). Preileal digestibility of whole or crushed corn was 30%; grinding increased preileal digestibility to 51%, and popping increased preileal digestibility of corn to 90% (Meyer et al 1995). Similarly, the glycemic response, hence assumed preileal digestibility, was greatest to least, respectively, after equivalent meals of steam flaked, ground and cracked corn (Hoekstra et al 1999). In more recent studies, processing (whole, ground, steamed, micronized, steam-flaked and popped) had no effect on glycemic and insulinemic responses when using processed oats (Vervuert et al 2003) or corn (Vervuert et al 2004), but glycemic and insulinemic responses to barley feeding were influenced by processing method (Vervuert et al 2007).
Assessment of carbohydrates in feed
Proximate analysis
Standardized methods of proximate analysis (Van Soest 1963, Van Soest et al 1991) relate to plant anatomy and fit reasonably well with the digestive physiology of ruminants, but not as well with the digestive physiology of the horse. A comprehensive scheme was proposed in order to separate carbohydrates into groups for analysis appropriate for horses (Hoffman et al 2001). This scheme, which compares carbohydrate fractions obtained by current systems of analysis with fractions as digested by the horse, has been updated here (Fig. 8.1). The system of analysis for ruminant nutrition separates carbohydrates largely on the basis of plant anatomy into NDF, from plant cell walls, or nonstructural carbohydrate (NSC), mainly cell contents (Van Soest 1963, Van Soest et al 1991). NSC was traditionally calculated by difference, 100 – water – protein – fat – NDF, until a recent movement within academia and the industry to improve the definition of terms associated with the nonstructural or nonfiber carbohydrate portion of feeds. Laboratories now analyze NSC directly, and the calculated by difference fraction is now termed “non-fiber carbohydrate” (NFC) and assumed to contain carbohydrates not found in the NDF fraction of feeds and forages (Anon 2001).
NSC analysis currently refers to the portion of plant carbohydrates analyzed directly, either by extraction in water or ethanol, or by enzyme hydrolysis. Thus, the NSC fraction may be further annotated as water-soluble carbohydrates (WSC), ethanol-soluble carbohydrates (ESC), and starch. The WSC fraction includes simple sugars, disaccharides, oligosaccharides, and some polysaccharides, namely fructans (Smith, 1981, Van Soest 1994). The ESC fraction is a subset of WSC, including mainly sugars, glucose, fructose and sucrose, and low molecular weight fructans but not polymeric fructans. Even with the potential inclusion of low molecular weight fructans, the ESC fraction is the most practical method for estimating simple sugar content. Starch is analyzed by treating residues (previously subjected to extraction with ethanol) to acid or enzyme hydrolysis using α-amylase (Smith 1981, Hall et al 1989). Fructan may be analyzed directly by HPLC (Cairns & Pollock 1988) or colorimetrically (McCleary et al 1997), but the HPLC is rather costly for practical purposes, and the colorimetric method (Megazyme™) appears to substantially underestimate the fructan content of some forages, yielding widely variable results (Longland & Harris 2009). Estimating fructan by subtracting ESC from WSC is perhaps the most practical approach, but since ESC may contain low molecular weight fructans, this method can underestimate actual fructan content (Longland & Harris 2009).
A system of carbohydrate analysis for human nutrition places greater emphasis on plant chemistry and includes non-starch polysaccharides (NSP) resistant to digestion by mammalian enzymes (Englyst et al 1982). Non-starch polysaccharides include soluble fibers (gums, β-glucans, mucilages, pectins) and insoluble fibers (hemicellulose and cellulose), but exclude lignin and lignocellulose, which have some significance in horse nutrition. Lignin and lignocellulose retard the rate of fermentation (Hall 1989) and are present in much larger proportions in horse feeds than in human diets. Additionally, lignocellulose may be degraded to cellulose by fungi and possibly other microbes present in the equine hindgut, so excluding this fraction may limit dietary assessment.
1. a hydrolyzed group (CHO-H) that yields sugars, mainly glucose for metabolism;
2. a rapidly fermented group (CHO-FR) that yields primarily lactate and some propionate, which may be metabolized as 3- or 6-carbon units via glucose, but may cause hindgut dysfunction;
3. a moderately rapid fermented group (CHO-FM) that yields primarily propionate as well as some acetate, which are metabolized largely as 3- or 6-carbon units, mainly via glucose, or 2-carbon unites via acetyl-CoA;
4. a slowly fermented group (CHO-FS) that yields primarily acetate and butyrate, which are metabolized as 2- and 4-carbon units, largely via acetyl-CoA.
Until such an analysis is available, it may be practically approximated in terms of hydrolyzable carbohydrate (simple sugars + starch or practically ESC + starch), rapidly and moderately fermented carbohydrate (the difference between NFC and ESC + starch), and slowly fermented carbohydrate (approximated by NDF), as indicated in Fig. 8.1.
These approximations are based on limited data reported from in vivo studies (de Fombelle et al 2004, Moore-Colyer et al 2002, Longland et al 1997) but are limited by the NFC fraction, which is calculated by difference and accumulates laboratory errors. The NFC fraction contains hydrolyzable, rapidly and moderately fermentable portions, including sugars, starch and fructan as well as gums, mucilages, β-glucans, and pectins, which are not recovered by the NDF method. The NSC hydrolyzable fraction as analyzed directly is not equal to NFC, and accounts for about one-fifth of the NFC in hay, one-third of the NFC in pasture, one-half to two-thirds of the NFC in fiber-rich feeds, and the majority of the NFC in typical “sweet feed” grain-mixes for horses (Hoffman et al 2001). Old laboratory methods calculated NSC by difference using the same equation now used to calculate NFC. While it would be less confusing to drop the use of the by difference terminology altogether, some laboratories still employ the old method and equate NSC and NFC. Thus, care should be taken when interpreting laboratory reports. For example, the NFC method of calculating NSC (old laboratory method) estimates the hydrolyzable carbohydrate fraction of beet pulp around 32% DM, when it is actually closer to 11%.
Glycemic response and glycemic index
The glycemic response is a reflection of plasma glucose and insulin responses to a meal, an in vivo estimate, rather than a chemical analysis of the hydrolyzable carbohydrates in a feed. The glycemic index is a classification of feeds relative to their capacity to raise blood glucose, and is generally expressed as a percentage of the area under the curve response to a standard quantity of a test feed compared to that of a standardized reference: an oral glucose dose or white bread in human nutrition (Jenkins et al 1981, Englyst et al 1996). The glycemic index has been applied primarily in human nutrition for diabetics in order to formulate diets with a low glycemic impact (Wolever & Mehling 2002), but it has not been applied consistently in horse nutrition (Harris & Geor 2009). Meal-related responses of blood glucose and insulin to different diets in horses have been quantified in several reports (Stull & Rodiek 1988, Rodiek et al 1991, Williams et al 2001
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree