11 Feeding stallions and broodmares
Stallions
Requirements
The nutrient requirements of breeding stallions are assumed to be similar to those of other adult horses, with allowances for level of activity. Level of activity should be considered the sum of breeding frequency and voluntary (and/or imposed) physical activity. Popular thoroughbred stallions may breed more than 150 mares in a season; often mounting 2 or 3 mares a day during the peak periods (Umphenour et al 2011). Conversely, some stallions may only breed a few mares in an entire year. Popular stallions used in artificial insemination programs may mount a phantom once a day, or more commonly, three or four times per week. An assessment of physical activity should include any imposed exercise (riding, driving, etc.) as well as voluntary exercise. Individual temperament can greatly influence level of voluntary physical activity. For example, some stallions may run or walk along a paddock fence during turnout periods while others may graze or rest quietly. Housing can also impact voluntary activity as stallions maintained primarily in stalls will often have a lower level of activity than stallions kept in pastures, particularly if they are used in a pasture-breeding system.
The effect of body condition on stallion libido or semen quality has not received much attention, but in humans, both very low and very high body mass index is associated with increased risk for infertility (Nguyen et al 2007). Obese men have been reported to have increased incidence of oligozoospermia, increased incidence of low progressively motile sperm and increased risk of sperm DNA fragmentation (Hammoud et al 2008a, b). The effects of obesity on fertility in men may be mediated by changes in several reproductive hormones including testosterone, estrogen and inhibin (Hammoud et al 2008a). It is also possible that changes to antioxidant status are related to the changes in semen quality observed in obesity-related infertility.
Based on the observations in humans, it would seem prudent to utilize a feeding program that prevents stallions from becoming too thin or too fat. Henneke and coworkers (1983) developed a 9-point body condition scoring system in which “moderate” body condition was associated with a score of 5. Many performance horses are maintained at condition scores between 4 and 6, while optimal reproductive efficiency in mares occurs at condition scores above 5. Therefore, a target condition score range for breeding stallions could be between 5 and 6.
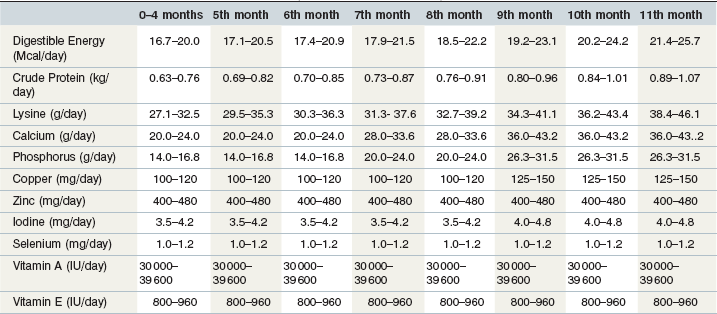
Energy intake is the primary nutritional variable affecting body condition. Mature, inactive stallions with docile temperaments may have digestible energy requirements that are similar to any other mature horse at maintenance. However, very active breeding stallions probably have higher energy requirements. Siciliano and coworkers (1993) reported that commercial thoroughbred stallions (~ 600 kg) received about 26 Mcal of digestible energy per day during early spring (March). This estimate of digestible energy intake was subject to several sources of error (intake was calculated from farm-reported amounts of hay and concentrate offered to the stallions and did not include any pasture consumed during 3 hours of turn-out each day). Nonetheless these observations suggest that the energy requirements of active breeding stallions are substantially above maintenance and may be similar to the requirements of horses in light or moderate work (20 to 40% above average maintenance). Table 11-1 provides daily recommended nutrient intakes for inactive and active breeding stallions.
Table 11-1 Recommended Daily Nutrient Intakes for Breeding and Nonbreeding Stallions (600 kg) with Various Levels of Voluntary Activity
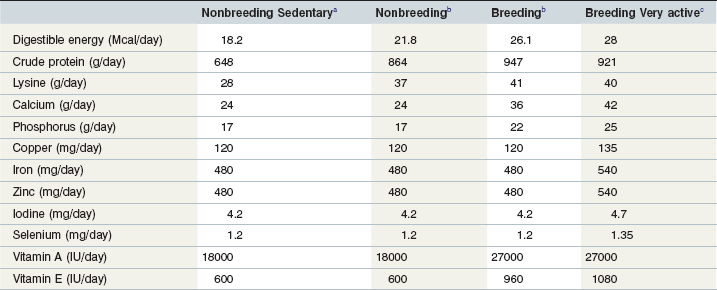
a Source: NRC (2007): Adult horse, minimal voluntary activity, no work (600 kg).
b Source: NRC (2007): Average nonbreeding and breeding stallions (600 kg).
c Source: NRC (2007): Adult horse in moderate work (600 kg). This category would apply to breeding stallions with very high activity levels, either from breeding or from self-imposed exercise.
Feeding programs
Table 11-2 provides several examples of diets that could be fed to stallions at different levels of activity. These examples should be considered as starting points and should be implemented in concert with a regular body condition scoring or weighing program. Dietary adjustments should be made whenever a stallion begins to become too thin or too fat. Adjustments to feeding programs should be made gradually. In the case of a stallion that is too fat, the goal should be to reduce calorie intake while maintaining adequate intake of protein, vitamins and minerals. Inactive stallions may be able to maintain body weight on a diet consisting of good quality hay or pasture and a small amount of a fortified supplement. Pasture availability and quality should always be considered when the diet is evaluated. For example, as pasture becomes available in the spring, or as weather conditions permit longer periods of turnout in good quality pastures, the amount of other feeds in the diet should be adjusted appropriately to prevent the stallion from becoming too fat.
Table 11-2 Example Daily Rations for Nonbreeding and Breeding Stallions (600 kg) with Various Levels of Voluntary Activity
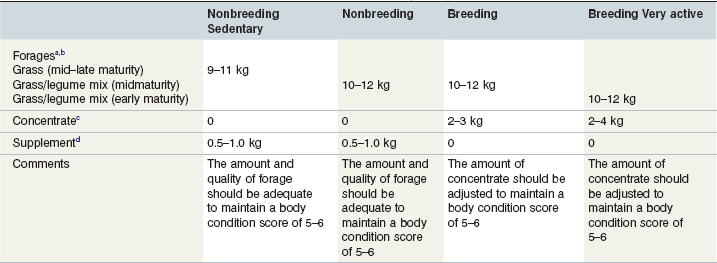
a Based on a late maturity grass hay containing (90% dry matter basis) 1.6–1.7 Mcal DE/kg, 7–11% CP, 0.3–0.4% Ca, 0.25–0.3% P, 6–9 ppm Cu; a mid-maturity grass/legume mix containing 1.8–2.0 Mcal DE/kg, 11–14% CP, 0.6–0.9% Ca, 0.25–0.35% P, 6–9 ppm Cu; and an early maturity legume/grass mix containing 2.0–2.1 Mcal DE/kg, 14–16% CP, 0.6–0.9% Ca, 0.25–0.35% P, 6–9 ppm Cu.
b Hay can be replaced with equivalent pasture.
c Based on concentrates containing (as fed basis) 3.2 Mcal DE/kg; 12–14% CP; 0.5% Ca; 0.3% P; 30 ppm Cu; 0.3 ppm Se.; 0.5 ppm I. Daily concentrate intakes should be divided into meals of less than 1.5 kg. When total daily concentrate intake is less than 2 kg/day, 1 kg of supplement may be used instead to provide adequate mineral intakes.
d Based on supplements containing (as fed basis) 14–30% CP; 2–4% Ca; 1–2% P; 100–150 ppm Cu; 0.6–1.0 ppm Se; 1–2 ppm I. A higher protein supplement should be selected if the hay contains less than 10% crude protein.
Semen quality
Despite the importance of semen quality to reproductive performance, very few studies have examined the effects of diet on sperm concentration, morphology or motility in fresh, cooled or frozen stallion semen. Sperm are relatively high in lipid content and changes in the lipid composition of the diet may have implications for sperm physiology (Wathes et al 2007). The addition of n-3 fatty acids to the diet has been shown to alter the fatty acid content of sperm in several species, but effects on sperm characteristics are variable (Cerolini et al 2006, Gliozzi et al 2009). The effects of fatty acid supplements on stallions have also been inconsistent. Supplementing the diet of stallions with 8 to 11 g of n-3 fatty acids from either fish oil or from a combination of flax seed and algae did not affect semen quality (Grady et al 2009). However, other researchers found that a supplement containing n-3 fatty acids altered semen fatty acid composition and resulted in improved 48 h motility in cooled semen and in frozen semen (Brinsko et al 2005). The authors noted that the supplement appeared to be of most benefit to stallions that started the study with relatively low progressive motility in cooled semen. Stallions that received a supplement containing 30 g of docosahexanoic acid (DHA) for 80 days had increased sperm number, increased motility and reduced percentage of abnormal sperm (Elhordoy et al 2008). These authors also reported that the stallions that exhibited the greatest improvements had the poorest semen quality at the beginning of the experiment.
Interaction between sperm and reactive oxygen species (ROS) appears to facilitate capacitation but exposure to excessive ROS may damage sperm DNA and reduce sperm viability (Ball 2008). To combat ROS, semen contains a variety of endogenous antioxidants, several of which may be susceptible to dietary manipulation. Short term supplementation of fertile pony stallions with an antioxidant supplement containing 300 mg vitamin E, 300 mg vitamin C, 4000 mg L-carnitine and 12 mg of folic acid had no effect on total sperm, percentage total motility or percentage progressive motility. Motility following 24 h storage was not affected, but there was a small decrease in the percentage of sperm with morphological defects (Deichsel et al 2008). Daily supplementation of stallions with 5000 IU of D,L α-tocopheryl acetate did not affect total sperm or percentage progressive motility in fresh semen but effects on sperm viability after storage were not investigated (Rich et al 1983). Although these studies demonstrated limited benefit from antioxidant supplements, it has been suggested that antioxidant therapy in men may be beneficial only in selected cases of infertility (Zini et al 2009). Vitamin E supplementation (3000 IU D-α-tocopherol/day) of stallions with poor post-thaw progressive sperm motility did not affect total or progressive motility in raw, 24 h cooled or frozen semen (Gee et al 2008). However, vitamin E supplementation improved motility in cooled semen after 48 h in that study. As noted above, the response to n-3 fatty acid supplementation also appeared to be most pronounced in stallions with low sperm motility in cooled semen. These results suggest that the relationship of dietary lipids and dietary antioxidants to semen quality in subfertile stallions should be explored further. The efficacy of n-3 fatty acid and antioxidant supplementation in the stallion should be considered in the context of the basal diet. In the studies mentioned above that observed benefits to supplementation, the basal diets consisted predominantly of stored feeds that would have been low in natural sources of vitamin E, vitamin A, β-carotene and n-3 fatty acids. The effect of n-3 supplementation or antioxidant supplementation could be expected to be less pronounced in stallions that have access to good quality pasture which is an excellent source of these compounds.
The relationship of semen quality to a variety of other nutrients (vitamin C, β-carotene, zinc, selenium, B-vitamins) has been studied in humans and other animals (Audet et al 2004, Eskenazi et al 2005, Ebisch et al 2007, Colagar et al 2009). Severe deficiencies are likely to detrimentally affect sperm production and/or semen quality but the response to supplementation above requirements is often variable. Inconsistent results may arise from differences in experimental conditions such as the levels of nutrients in the basal diets or length of the supplementation period.
Key Points –
Stallions
• The nutrient requirements of stallions are similar to other horses with adjustments for level of activity.
• Feeding programs should focus on balanced diets fed in amounts to maintain a body condition between 5 and 6.
• A variety of nutritional supplements have been fed to stallions in an attempt to affect semen quality but results are inconsistent.
• Stallions with normal fertility appear to be less likely to respond to nutritional supplements than subfertile stallions.
Pregnant mares
Requirements
The daily nutrient intakes of pregnant mares should meet the maintenance requirement of the mare and the nutrient needs for the synthesis and maintenance of the products of conception. Gestational weight gain is expected to be 12–16% of a mare’s initial body weight, most of which is attributed to the fetus and placental tissues. Since 1978, most feeding standards for pregnant mares have relied on the estimates of nutrient deposition in the fetus that were published by Meyer and Ahlswede in 1978. Their data (Table 11-3) were obtained from compositional analyses of stillborn and aborted fetuses presented to a veterinary college. A few other studies have also reported fetal weights from aborted fetuses but weights and compositional analyses from normal fetuses in various stages of development are not available.
Table 11-3 Chemical Composition of the Equine Fetus at 7, 8, 9, 10, and 11 Months of Gestationa
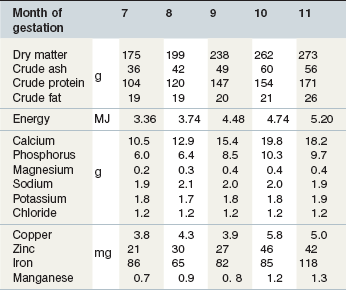
Source: Meyer and Ahlswede (1978).
Most fetal weight gain occurs during the third trimester of gestation but the nutrient needs of pregnant mares begin to increase before then (Table 11-4). The vitamin E and vitamin A intakes of pregnant mares are elevated above maintenance throughout pregnancy (NRC 2007). It is suggested that calcium and phosphorus requirements do not increase until the 7th month of gestation (NRC 2007) even though the available data on fetal composition suggest that calcium and phosphorus have been deposited into the fetus before then. The normal maintenance intakes may be sufficient to meet calcium and phosphorus needs before the 7th month of gestation or the current recommendations for calcium and phosphorus may be somewhat low. In the 6th revised edition of the Nutrient Requirements of Horses, the recommended intakes of copper and iodine were increased for mares in late gestation. However the recommended intakes for several other nutrients (iron, manganese, zinc, selenium) were not changed. An increase in the recommended copper intake was based on the studies that suggested copper supplementation of pregnant mares might influence the copper status of foals and their subsequent susceptibility to developmental cartilage lesions (NRC 2007). The change in the iodine requirement was based on a review of a published study that had been misquoted in an earlier publication (NRC 1989) and the recommendations of Donoghue et al (1990).
Several studies have examined the effect of vitamin and mineral supplementation of the pregnant mare on the passive transfer of immunoglobulins to neonatal foals. There does not appear to be a relationship between maternal vitamin E status and colostral immunoglobulin G (IgG) concentration or foal serum IgG when mares have adequate vitamin E status at foaling (Siciliano et al 2009). However, in mares that were kept in dry lots prior to parturition vitamin E supplementation at twice the recommended concentration resulted in increased colostral IgG concentrations and increased serum IgG concentrations in foals (Hoffman et al 1999). Colostral IgG concentration was increased when mares were given a marine source supplement containing DHA compared to a corn oil supplement. However, foal IgG concentrations were not affected (Kruglik et al 2006). In another study colostral IgG concentrations were lower in mares receiving a marine based n-3 fatty acid supplement (Stelzleni et al 2006), whereas a linseed-based n-3 fatty acid supplement did not alter colostral immunoglobulin concentrations in comparison to a rapeseed-based oleic acid supplement (Duvaux-Ponter et al 2004).
Selenium supplementation above the requirement has been reported to increase serum IgG concentrations (Janicki et al 2000), but in another study selenium supplementation did not affect colostral or foal IgG concentrations (Thorson et al 2010). In addition, time to expulsion of the placenta was not affected by selenium supplementation in either study. Replacing sulfated sources of copper, zinc, manganese, and cobalt with amino acid complexes resulted in increased colostral IgA concentrations but did not affect colostral or foal IgG concentrations (Vickers et al 2009). Clearly the effects of diet on passive transfer are variable. Some of the variation may relate to characteristics of the basal diet that are not always considered during the design of the study such as availability of natural sources of vitamins or minerals from the forage and even the level of nutrient intake by the mare. Pagan and Hintz (1986) observed that energy intake was inversely correlated with the concentration of several milk components. Thorson et al (2010) found that Brix percentage and colostral IgG concentrations were higher in mares that received pasture only compared to mares that received pasture and concentrate. Therefore it is possible that well-fed mares produce more dilute milk or colostrums and comparisons among various treatments should be adjusted to a dry matter basis or another component.
The amount of protein and energy needed to fuel the products of conception increase above maintenance in the 5th month of gestation and continue to increase throughout the gestation period. Feeding excess energy in late gestation will increase body weight and condition but does not result in larger birth weights (Kubiak et al 1988). Similarly, moderate energy restriction in late gestation does not affect birth weight (Banach & Evans 1985, Thorson et al 2010) but it may result in prolonged gestation (Hines et al 1987).
Less than 40% of fetal weight gain occurs before the 9th month of gestation but appreciable increases in body weight have been reported during early or mid-gestation in mares maintained in both research and commercial settings (Lawrence et al 1992, Cassill et al 2009). Increases in condition score occur simultaneously with weight gain in early and mid-lactation (Lawrence et al 1992). The optimal body condition score for a pregnant mare at any stage of gestation has not been established. One group attempted to study the effect of moderate and high rates of weight gain in early and mid-gestation on pregnant mares (Wilsher & Allen 2006, Ousey et al 2008). The study was confounded by an outbreak of Streptococcus equi in the mares at about 100–140 days of gestation, so the authors inadvertently studied the effect of illness and marked weight loss (approximately 8–11%) on pregnant mares. When the authors compared the data obtained from all mares that had been sick to historical data on normal mares they concluded that the placental efficiency of the infected mares had been reduced (Wilsher & Allen 2006). Interestingly, mares that had gained more weight prior to getting sick appeared to sustain fewer long-term effects of illness and weight loss on measures of placental efficiency. In addition the foals from the fatter mares appeared to have more normal responses to an insulin sensitivity test administered at 2–4 days of age. The mares that had been fed for high weight gain had a body condition score of 7 at the onset of illness, compared to a body condition score of 5 for the mares that had been fed for moderate weight gain. The authors suggested that feeding mares to a higher condition score could have mitigated the effects of the acute nutritional deprivation that occurred during the infection with Streptococcus equi (Ousey et al 2008
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree