Anthelmintic Resistance in Camelid Parasites
Definition of Anthelmintic Resistance
Anthelmintic resistance is defined as genetic mutation in the worm population, which alters susceptibility to drugs being used for treatment.1,2 Frequent exposure to an anthelmintic creates selection pressure, which eliminates susceptible worms from the population. Resistant worms survive treatment and pass their genetic advantage to subsequent generations. Over the past few decades, many factors have contributed to increase in anthelmintic resistance in nematodes that affect grazing animals. Most parasitologists believe that frequent administration of anthelmintics on a whole-herd basis is a major contributing factor.3 This approach fails to preserve refugia. Refugia refers to the proportion of worm population spared selection pressure from exposure to anthelmintics.4 Refugia provide a reservoir of drug-susceptible genes that help dilute out resistant genes in the worm population. Refugia include eggs and larval stages in the environment and also worms in animals that were not treated with the anthelmintic. Parasitic stages within the host that are not affected by an anthelmintic treatment also represent refugia. The evolution of drug resistance is slowed when a large refugia is maintained. Unfortunately, the importance of maintaining refugia has only recently been recognized, and producers are now reaping consequences of decades of nonselective use of anthelmintics in the form of worms that are resistant to multiple classes of anthelmintics.
Multianthelmintic-Resistant Parasites
Emergence of multianthelmintic-resistant worms in small ruminants and camelids is a major concern, as this phenomenon has led to an increasing number of anthelmintic treatment failures.3,5–9 As a result, morbidity and mortality rates have increased in susceptible animals. The trichostrongyle parasites Haemonchus contortus and Trichostrongylus colubriformis are especially capable of rapidly acquiring resistance to anthelmintics. These worms are common inhabitants of small ruminants and camelids in the southeastern United States.3,5,7–9 Most nematode species that affect camelids also can be found in the North American cervid populations.10 Life-threatening haemonchosis has also been reported in many northern states and in Canada, indicating that these worms are not as geographically restricted as once believed (Bob Storey, personal communication 2011). Haemonchus contortus is a virulent, blood-feeding nematode, which damages the third compartmental and abomasal mucosa in camelids and small ruminants, respectively. Heavy infections cause chronic anemia, hypoproteinemia, loss of fecal consistency, and significant weight loss. Severely affected animals may ultimately die from progressive blood loss. Periparturient females, crias in their first grazing season, and animals debilitated from other diseases are most at risk. Mandibular edema (“bottle jaw”) is often observed in severe small ruminant cases but is not a common feature of camelid haemonchosis. T. colubriformis is less virulent than H. contortus and causes morbidity primarily through irritation of the small intestinal mucosa. In contrast to H. contortus, T. colubriformis infections are not associated with anemia.
Several studies conducted in the southeastern United States have provided insights into the magnitude of anthelmintic resistance associated with H. contortus. On the basis of results of in vitro larval developmental assays (LDA) performed on 45 sheep and goat farms, H. contortus isolates were found to be resistant to benzimidazoles, levamisole, and ivermectin on 98%, 54%, and 24% of the farms, respectively.5 Resistance to all three anthelmintics (hence, all three major anthelmintic classes) was evident on 48% of the farms. Total anthelmintic resistance (i.e., resistance to benzimidazoles, levamisole, ivermectin, and moxidectin) was identified on 17% of the small ruminant farms.5 The situation is, apparently, not as dire in camelids, but the same trends are evident. LDAs conducted on 32 camelid farms in the southeastern United States showed that 97% of the farms had multianthelmintic-resistant H. contortus isolates.9 Resistance to benzimidazoles was noted on 100% of farms, and resistance to ivermectin was noted on 88% of farms. However, only 22% of the H. contortus isolates were resistant to levamisole, and 22% were resistant to moxidectin. Total anthelmintic resistance was identified on only one of the 32 camelid farms.9 These findings are sobering because unless an immediate change is made in the way camelid owners use anthelmintics in their herds, the camelid industry will soon find itself in the same straits as the small ruminant industry. Currently, diseases associated with H. contortus infections threaten the viability of the sheep and goat industry in areas where this worm is highly prevalent.3,5,6
Making the Case for Selective Anthelmintic Treatment
In small ruminants, worm burdens are unevenly distributed among animals; approximately 20% to 30% of the animals harbour 80% of the parasite population.3,11 A similar disaggregate distribution of trichostrongyles was noted in camelid herds.12 The majority of camelids in most herds have low parasite burdens, and they will not gain much benefit from anthelmintic treatment. If left untreated, however, these minimally infected animals provide a major source of refugia. Discriminating between camelids that need anthelmintic treatment and those that do not takes a combination of hands-on husbandry and periodic evaluation of quantitative fecal egg counts. This approach is more labor intensive than traditional calendar-based “whole herd” treatment programs, but the benefits of adopting a selective treatment program cannot be over-emphasized.3 Selective use of anthelmintics will prolong the efficacy of currently available anthelmintics, which should be regarded as precious and limited resources. Even though several new anthelmintics are on the horizon, they will be significantly more expensive than those that are currently available. It is highly likely that resistance will continue to evolve more quickly than new anthelmintics can be developed to replace those that are failing. As a result, sensible use of anthelmintics and management practices aimed at breaking the parasitic life cycle will remain essential aspects of current and future parasite control strategies.
Accurate identification of animals that require anthelmintic treatment to maintain health and productivity is the cornerstone of a selective treatment program. Body condition score (BCS) is a useful parameter to monitor camelid herds, as it is a good barometer of overall health. Generally, healthy-appearing, active animals that have optimal to high body condition scores are the least likely to be harboring health-threatening worm burdens. The BCS subjectively grades the amount of muscle and subcutaneous body fat over bony protuberances and categorizes them on a 1-to-9 scale, 1 for emaciation and 9 for obesity. A body condition score of 5 out of 9 is considered ideal, although some healthy individuals naturally maintain condition at a slightly higher or lower score. Camelids should be palpated from the withers to the loin and in the brisket area because fiber may conceal the actual body condition (Figure 6-1). It is important to notice any changes in the BCSs among individuals and the overall herd, especially if the scores are declining. Parasitism and inadequate nutrition are common reasons for decline in BCSs in multiple animals in a herd.
Since anemia is a prominent feature of haemonchosis, pallor of the conjunctiva and mucous membranes is an excellent indicator of the severity of H. contortus infections. Francois Malan noticed the association between anemia and eyelid pallor in parasitized sheep in South Africa.13 He subsequently developed a laminated card that depicted five illustrations of ocular membrane colors. The membrane colors are categorized as follows:
The ocular colors correlate with mean packed cell volumes (PCVs) in parasitized sheep. Studies conducted in the United States on sheep and goats with haemonchosis have demonstrated that the FAMACHA© eye score, PCV, and fecal egg counts are all highly correlated.14,15 Since introduction of the FAMACHA system in 2004, over 15,000 FAMACHA cards have been distributed in the United States, and numerous workshops have been conducted to teach small ruminant producers how to implement the concepts in their herds and flocks (Figure 6-2).16

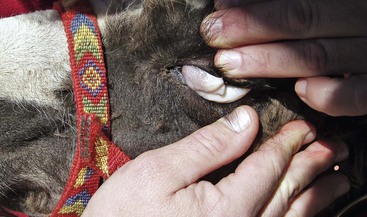
Figure 6-2 Comparison of a llama’s lower conjunctival color to the color blocks on the FAMACHA card. This llama scored as a 1 out of 5 (not anemic).
The FAMACHA system was recently evaluated on 17 llama and alpaca farms that had a high prevalence of H. contortus. PCV was highly correlated to FAMACHA eye scores, fecal egg counts, and BCSs.12 On the basis of these findings, the FAMACHA system has been deemed a useful tool for the management of H. contortus in llamas and alpacas in the area studied. It is most accurate when the prevalence of anemia associated with haemonchosis is high. The FAMACHA system is, however, not intended as a management tool for other types of internal parasites. Ocular examinations need to be conducted in bright natural light, using the FAMACHA card as a color reference. Alpacas and docile llamas may be restrained manually, but the more fractious camelids need to be restrained in a chute for safety purposes. The normal tendency of most handlers is to place a hand on top of the animal’s forehead during ocular examination, but camelids often object to being touched in this area. Camelids are accepting of the scoring process if the examiner stands to the side of the animal and slowly slides one hand up the side of the animal’s face towards its eye. The index finger is used to gently but firmly retropulse the globe through the upper eyelid, and the thumb of the same hand is used to evert the lower eyelid. The FAMACHA card is held beside the eye with the opposite hand. The examiner should expose as much conjunctiva as possible to accurately assess color. The color of the lower eyelid conjunctiva is compared with the FAMACHA card over a 1- to 3-second period and assigned a score. Both lower eyelids are scored, and if variation is noted between the two eyelid scores, the more anemic (higher) score is assigned. Some camelids have heavily pigmented inner eyelids, but usually, a nonpigmented area can be scored. Animals with haemonchosis with scores in categories 4 or 5 should be treated with an effective anthelmintic. Animals with scores in categories 1 or 2 are not anemic and therefore do not need treatment. Animals that receive a FAMACHA score of 3 should be handled according to prevailing circumstances. For instance, young animals and animals in herds or flocks where the majority of the animals have anemia, poor body conditions, or both should be treated, particularly during periods of high H. contortus transmission (warm, moist conditions). The main benefit of the FAMACHA system is that far fewer animals will be treated than when nonselective methods are used, so refugia are maintained. FAMACHA examinations should be conducted every 1 to 2 weeks during periods of high H. contortus transmission and less frequently during times of the year when transmission is typically low. Although haemonchosis is a major cause of chronic blood loss in camelids, other causes of anemia should be considered when pale ocular membranes are observed (Figure 6-3).17 Other etiologies include blood loss from causes other than endoparasitism, ingestion of wilted red maple leaves, chronic disease, naturally occurring iron deficiency, and symptomatic Candidatus Mycoplasma haemolamae (CMhl) infections. Although most CMhl infections are not clinical, red blood cell (RBC) destruction may occur if the animal becomes severely stressed or immunocompromised.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree