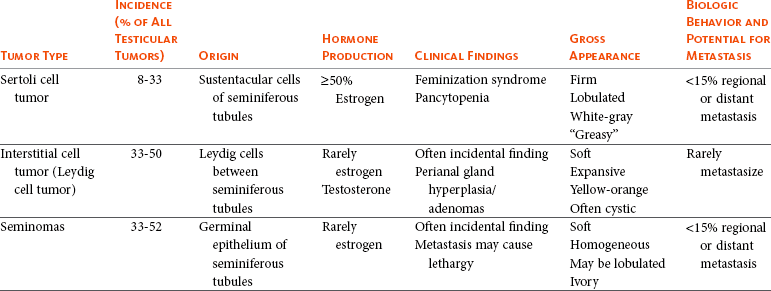
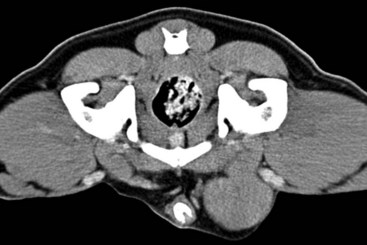
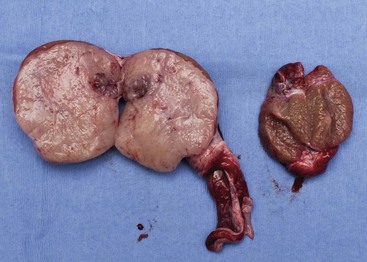
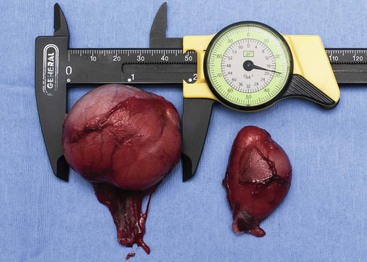
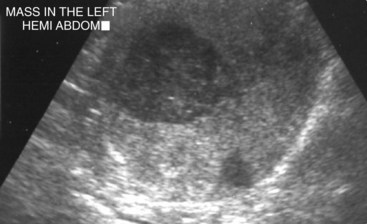
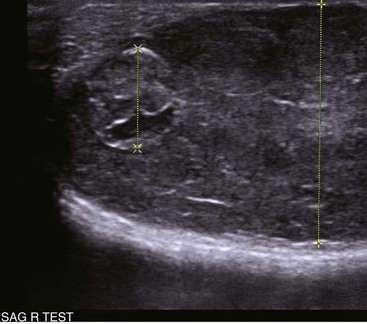
28 Testicular tumors are the most common tumors of the canine male genitalia and account for approximately 90% of all cancers in the male reproductive tract.1–4 In the intact male dog, the testis is overall the second most common anatomic site for tumor development, with an overall prevalence ranging between 6% and 27%.1,3–7 Many of these reports are case series and involve dogs submitted for routine necropsy and/or castration for cryptorchidism, making comparisons between study prevalence data difficult. However, a recent population-based study conducted in Norway, where elective castration is rare, reported a similar prevalence of 7% for testicular tumors.8 The rate of development of testicular cancer in humans has increased in some populations over time and across successive birth cohorts, and a similar phenomenon has been suggested in dogs.7,9–13 A recent population-based study did not find increased rates of testicular tumors among intact dogs; however, only an 8-year period was evaluated.8 Testicular tumors are most often diagnosed in geriatric male dogs with a median age of approximately 10 years.1,3,4,14,15 The three most common testicular tumors arise from distinct testicular subsets: sustentacular cells of Sertoli, the spermatic germinal epithelium, and the interstitial cells of Leydig, giving rise to Sertoli cell tumors, seminomas, and interstitial cell tumors, respectively (Table 28-1).2 The World Health Organization (WHO) classification of tumors of domestic animals differentiates the major types of testicular tumors in dogs as sex cord stromal tumors (Sertoli cell tumors, interstitial cell tumors), germ cell tumors (seminoma, teratoma), and mixed germ cell sex cord stromal tumors.16 Sertoli cell tumors, interstitial cell tumors, and seminomas have historically developed with equal frequency, although recent studies have suggested that the prevalence of Sertoli cell tumors is lower (8% to 16%).* In one study evaluating lifetime occurrence of neoplasia in German shepherds and Belgian Malinois, seminoma occurred most frequently.17 Human testicular tumors are often divided into seminoma and nonseminoma, with the former further subdivided as classical seminoma (SE), atypical seminoma, and spermatocytic seminoma (SS) according to WHO, and some effort has been made to apply this to canine tumors.9,13,19–23 Sertoli cell tumors and seminomas occur with higher frequency in cryptorchid testes.3,24,25 Rarely, other cell lineages can give rise to testicular tumors such as hemangiomas, granulosa cell tumors, teratomas, sarcomas, embryonal carcinomas, gonadoblastomas, lymphomas, schwannoma, mesothelioma, and rete testis mucinous adenocarcinomas.26–30 Many dogs diagnosed with testicular cancer have more than one primary tumor.3,6,18,31 In three separate studies evaluating relatively large numbers of dogs with testicular tumors, between 4% and 20% of dogs had more than one type of testicular tumor.3,8,32 Several factors may influence the development of testicular tumors in the dog, including cryptorchidism, age, breed, and carcinogen exposure. There is a significant association between cryptorchidism and the development of Sertoli cell tumors and seminomas but not interstitial cell tumors.4,15,25 An early prospective epidemiologic study compared the incidence of testicular tumors in cryptorchid dogs to age- and breed-matched control dogs.15 None of the control dogs developed testicular tumors during the study, in which the average duration of monitoring was 2 years. The incidence of testicular neoplasia in the cryptorchid dogs was 12.7 per 1000 dog-years at risk, whereas for cryptorchid dogs older than 6 years, the incidence increased to 68.1 per 1000 dog-years at risk. Inguinal cryptorchidism may further increase the risk of testicular tumor development compared to abdominal cryptorchidism (Figure 28-1).4,15,25 In cryptorchid dogs, tumors more frequently develop in the right testicle; however, this is probably due to the fact that the right testicle is more likely to be retained.4,15,32 One study of cryptorchid dogs found that chronologic age was a risk factor for development of a primary testicular tumor and dogs older than 10 years were more likely to develop tumors compared to dogs younger than 6 years.15 A recent study indicated that the detection rate of testicular tumors in dogs younger than 10 years was significantly associated with cryptorchidism, with over 60% of cryptorchid testicular tumors identified in middle-aged dogs (6 to 10 years).4 Several breeds have been reported to have increased risk of developing primary testicular tumors, including the Boxer, German shepherd, Afghan hound, Weimaraner, Shetland sheepdog, Collie, and Maltese.* The flat-coated retriever, Rottweiler, Bouvier de Flandres, and Leonbergers may have a reduced risk of developing testicular tumors, although low numbers of the latter two breeds were evaluated.8 Two studies evaluating military working dogs suggested evidence of environmental carcinogen exposure during the Vietnam War.35,36 Pathologic changes in the testicles such as hemorrhage, epididymitis, orchitis, sperm granuloma, testicular degeneration, and seminoma were noted, although the causative factor of these could not be definitively determined. These epidemiologic studies postulated that exposure to phenoxy herbicide, dioxin, or tetracycline may have promoted the development of testicular tumors.35,36 Sertoli cell tumors arise from the sustentacular cells of seminiferous tubules, and seminomas arise from the germinal epithelium of seminiferous tubules. Interstitial cell tumors arise from Leydig cells located between seminiferous tubules. All three tumors have relatively distinct gross appearance but require histopathology for definitive diagnosis. Sertoli cell tumors are firm, lobulated, white-to-gray in appearance, and characterized as “greasy” on palpation.37 Seminomas tend to be homogeneous, soft, and occasionally lobulated and have an ivory appearance when sectioned (Figure 28-2).37 Interstitial cell tumors are soft, expansive, and yellow-to-orange in color when sectioned and often contain cysts with serous or serosanguineous fluid.37 The molecular and cellular biology of primary testicular tumors has been investigated. Proliferation markers (proliferating-cell nuclear antigen [PCNA], Ki67, and argyrophilic nucleolar organizer regions [AgNORs]) and TERT expression (the catalytic reverse transcriptase subunit of telomerase) have been interrogated as indicators of degree of malignancy, local progression, and metastasis with discordant results.38–43 Investigators sought to relate TERT to proliferation indices and p53 expression; however, due to the fact that PCNA and TERT were expressed in all testicular tumors, this limited their prognostic potential.40 However, aggressive testicular tumors did tend to express high levels of all markers examined: TERT, p53, PCNA, and Ki67.40 Proliferative activity in seminomas was assessed using AgNORs in a separate study, in which mean AgNOR scores were higher in invasive or diffuse tumors compared to well-differentiated intraductal seminomas.43 Results suggest that testicular tumors develop a proliferative advantage as they become less differentiated, although larger studies should be performed before proliferative indices can be definitively relied on for prognostication in canine testicular tumors. Cyclins, which are intracellular proteins that form complexes with cyclin-dependent kinases to regulate cell-cycle checkpoints, have also been evaluated in normal and neoplastic testes; however, their significance is yet to be determined.44 The neoplastic and stromal cellular environment plays an important role in tumor invasion and progression, and a few studies have attempted to investigate changes in canine testicular tumors. Laminin is an extracellular matrix protein that plays a role in anchoring cells to the basement membrane. As tumors became more invasive, laminin expression became fragmented or lost in Sertoli cell tumors and seminomas and correlated to increasing proliferative activity as assessed by PCNA scoring, Ki67 index, and mitotic index.38 Connexin 43 is the predominant gap junction protein of the testis that plays a role in phenotypic differentiation, cell pattern formation, and morphogenesis, and altered expression patterns may contribute to tumorigenesis and progression.45–49 Similar to other work, differential alterations in connexin 34 expression occur in canine testicular tumors, and its expression may aid in differentiating neoplastic Sertoli cells from seminomas.50 Mutations of the p53 tumor suppressor gene are a common genetic alteration in both human and canine malignancies, and increased p53 expression has been associated with tumor progression.51–55 A recent evaluation of testicular tumors indicated that nuclear p53 immunoreactivity was detected in 15 of 20 seminomas (75%), 6 of 12 Sertoli cell tumors, and all 3 interstitial cell tumors evaluated.40 Interestingly, expression intensity was stronger in diffuse type Sertoli cell tumors and seminomas.40,54 Results suggest that p53 may be an indicator of tumor aggression; however, further studies should be done to corroborate this. Similar to p53 and proliferation indices, angiogenesis plays an important role in cancer progression and metastasis. Vascular endothelial growth factor (VEGF) expression and microvessel density (MVD) were higher in seminomas compared to normal testes in one study.56 Additionally, both VEGF expression and MVD were higher in diffuse seminomas compared to more well-differentiated intratubular seminomas, potentially providing a histologic indicator of malignant behavior.56 The KIT protein or CD117 is a transmembrane protein for a tyrosine kinase receptor encoded by the proto-oncogene c-kit, which, when bound to its ligand stem cell factor (SCF), is essential to the development, proliferation, and maturation of several cell types, including germ cells.57–59 Primordial germinal cells express KIT and migrate to interact with Sertoli cells that express SCF to guide the differentiation of the primordial cells into gonocytes.60 Interstitial cells of Leydig also express KIT and, when stimulated by SCF, are stimulated by Sertoli cells to produce testosterone.57,60,61 The expression of KIT is maintained by spermatogonia until differentiation into spermatocytes, making it a useful marker to define primordial germinal cells and early germinal cells.60,62 In human seminomas, immunohistochemical labeling for KIT and placental alkaline phosphatase (PLAP) is used to distinguish SE and SS because SE should express both and SS should be negative.63,64 Normal and neoplastic canine testes were recently characterized for KIT expression, and results were consistent with humans and rodents in that spermatogonia and Leydig cells were KIT positive, whereas Sertoli cells were negative.62 Interestingly, canine testicular tumors in this study maintained the same KIT expression as their cell of origin, with interstitial tumors consistently expressing KIT, similar to human tumors. Similar to human seminomas, canine seminomas appeared to be differentiated into SE and SS on the basis of KIT and PLAP staining.22,62 Further studies investigating the behavior of SE and SS may support the dog as a relevant model for human disease, permitting investigation into additional roles of KIT-inhibitors.65–67 Most primary testicular tumors in the dog are characterized by local invasion and rarely metastasize. Regional or distant metastasis occurs in less than 15% of dogs diagnosed with Sertoli cell tumors or seminomas.6,32,68–74 Due to the malignant histologic appearance but low malignant behavior, canine seminoma has been compared to human spermatocytic seminoma, thereby spawning interest in using canine seminoma as a relevant comparative model.21 Recent work has tried to further classify canine seminoma into classic and spermatocytic seminomas in an attempt to help determine metastatic potential, but no clinical conclusions may be drawn as yet.19,21,22 Interstitial cell tumors very rarely metastasize.6 Sites of metastasis may include regional lymph nodes, eyes, brain, lungs, kidney, spleen, liver, adrenal glands, pancreas, skin, and peritoneum.6,32,68–74 Primary testicular tumors can also cause imbalances in hormone levels, regardless of the degree of local invasion and presence or absence of metastasis. Sertoli cell tumors can cause signs of feminization, and over 50% of affected dogs display signs of estrogen production.14,25,32,69 Seminomas and interstitial cell tumors are rarely associated with feminization.75–77 Excess estrogen can cause signs such as bilateral symmetric alopecia, cutaneous hyperpigmentation, epidermal thinning, squamous metaplasia of the prostatic epithelium, gynecomastia, galactorrhea, attraction of other males, preputial atrophy, atrophy of the nonneoplastic testicle, and bone marrow suppression.32 Sertoli cell tumors that develop in retained testicles are more likely to produce signs of hyperestrogenism; however, 17% of dogs with scrotal Sertoli cell tumors developed feminization.14,25,32,69 Plasma hormone concentrations from dogs with primary testicular tumors have been investigated in order to better understand their contribution to tumor type and clinical signs.77–80 Estradiol-17β concentrations were higher in dogs with Sertoli cell tumors compared to normal and were significantly higher in dogs with feminization syndrome secondary to Sertoli cell tumors. Testosterone and testosterone/estradiol ratios are lower in dogs with Sertoli cell tumors when compared to healthy control dogs.76 Plasma estradiol concentrations were lower in seminomas compared to normal dogs in one study but were not significantly altered from normal in another study.76,77 It has been suggested that clinical signs of feminization due to Sertoli cell tumors best correlated to testosterone/estradiol ratio reductions rather than to absolute increases in estradiol.76 Expression of inhibins (inhibins α, β, βα), 3β-hydroxysteroid dehydrogenases, and insulin-like growth factors (IGF-1 and IGF-2) has also been assessed, and further study may shed light on their utility as diagnostic or prognostic markers.77–80 A recent study suggested IGF gene expression was altered in canine testicular tumors compared to normal testicular tissue. A unique pattern of expression, as determined by reverse-transcriptase polymerase chain reaction (RT-PCR) of IGFs, their receptors, and their binding proteins, was observed in interstitial cell tumors compared to Sertoli cell tumors and seminomas.80 The overall changes in gene expression were small, however, and it remains unclear how significant the IGF signaling system is in canine testicular neoplasia. Most dogs with testicular tumors are asymptomatic, and a testicular mass is discovered as an incidental finding. However, clinical signs may be attributable to the primary tumor, to the presence of metastasis, or to paraneoplastic syndromes such as hyperestrogenism. Additionally, breeding dogs may present with fertility problems. Diagnosis is usually made via palpation of an enlarged testicle or a testicular mass during routine physical examination, abdominal ultrasound, or necropsy. Atrophy of the remaining normal testicle is common (Figure 28-3). Tumors in cryptorchid dogs may cause a regional mass effect within the caudal abdominal cavity or inguinal region (Figure 28-4). Excess estrogen may cause signs of feminization and is the most common paraneoplastic syndrome associated with canine testicular tumors. As stated previously, seminomas and interstitial cell tumors are rarely associated with feminization, whereas over 50% of dogs with Sertoli cell tumors show signs of hyperestrogenism.14,32,69,75–77 Common clinical signs include bilateral symmetric alopecia and hyperpigmentation, pendulous prepuce, gynecomastia, galactorrhea, atrophy of the penis, and squamous metaplasia of the prostate.32 The most deleterious effect of hyperestrogenism is bone marrow suppression, which may be irreversible and life threatening. Early effects of estrogen on the bone marrow include a transient increase in granulopoiesis with peripheral neutrophilia followed by progressive neutropenia, thrombocytopenia, and nonregenerative anemia.75,81 Severe pancytopenia from bone marrow hypoplasia and blood dyscrasias can be fatal, and clinical signs can range from hemorrhage secondary to thrombocytopenia, anemia, and febrile neutropenia.75,81 Less common reported signs associated with testicular neoplasia include lethargy (Sertoli cell tumors), presence of concurrent prostatic cyst or abscess, hematuria, hemoperitoneum, spermatic cord torsion, hypertrophic osteopathy, and perianal gland hyperplasia/adenomas (interstitial cell tumors).3,32,69,82–85 There are several reports of a syndrome in middle-aged miniature Schnauzers of Sertoli cell tumors occurring in cryptorchid male pseudohermaphrodites.86–88 A recent report also described the presence of both a Sertoli cell tumor and an interstitial cell tumor in a mixed breed male pseudohermaphrodite dog.89 Physical examination of intact male dogs, particularly older dogs, should always include palpation of the testicles for masses and/or asymmetry. A thorough rectal palpation should be performed to evaluate the prostate gland, regional lymph nodes, and perianal region. In dogs with clinical signs of hormone imbalance, serum testosterone and estradiol-17β can be measured along with testosterone : estradiol ratio.76,77 It is important to note that not all dogs with signs of feminization have absolute increases in estradiol-17β, and clinical signs may be more closely linked to altered androgen : estrogen ratios.76 Definitive diagnosis is achieved by histopathologic evaluation, although the presence of a testicular mass and cytology may be supportive of testicular neoplasia. Because most dogs with testicular tumors are older and therefore have a high likelihood of another primary tumor (up to 50%) or concurrent diseases, complete staging prior to surgery is generally recommended. Preoperative staging typically includes a complete blood count (CBC) to evaluate for hematologic abnormalities, chemistry profile, urinalysis, abdominal ultrasound, and three-view thoracic radiographs. A coagulation profile may be warranted in dogs with anemia and signs of hemorrhage. Abdominal ultrasound may serve multiple purposes: it can aid in identification of undescended testicles in the abdominal cavity or inguinal canal, assessment of regional lymph nodes, assessment of prostatic changes, and evaluation of common sites of metastasis such as spleen and liver (Figure 28-5). Testicular ultrasonography may aid in differentiating neoplastic processes from orchitis, testicular torsion, and epididymitis; however, changes are not specific enough to identify tumor type.90–92 Ultrasound-guided fine-needle aspiration (FNA) may support a suspicion of neoplasia prior to orchiectomy, particularly in breeding animals.90,93 For owners with financial constraints, minimum staging should consist of a CBC, chemistry profile, and urinalysis. Castration may be performed prior to full staging for some cases, with the decision to do a full work-up following histopathologic evaluation because it is appropriate therapy for most testicular tumors. Histopathologic diagnosis is generally straightforward; however, occasionally, immunohistochemistry (IHC) using vimentin, cytokeratin, desmin, and possibly KIT and inhibin may be indicated to identify the underlying cell of origin.26,78,94–97 As most primary canine testicular tumors are characterized by local infiltration with low potential for metastasis, orchiectomy with scrotal ablation is the treatment of choice and is often curative. Bilateral orchiectomy is the treatment of choice for testicular tumors, given that up to 50% of dogs have bilateral tumors and only 12% are clinically detectable in the contralateral testicle.15 In valuable breeding dogs, unilateral orchiectomy can be considered with continued monitoring afterward.98,99 Exploratory laparotomy is indicated in cryptorchid dogs, in which case the regional lymph nodes can be visually assessed and biopsied if indicated. In dogs with signs of hyperestrogenism secondary to the primary tumor, clinical signs typically resolve within 1 to 3 months following castration, unless metastatic lesions provide persistent estrogen release.68,69,74 Recurrence of feminization following castration may be associated with the presence of metastasis.69 Serum hormone levels may be monitored following castration and may correlate to resolution of clinical signs.76,100 Dogs with bone marrow hypoplasia secondary to estrogen toxicity require close monitoring perioperatively and postoperatively for complications requiring medical intervention with blood products and/or antibiotics. These dogs carry a guarded prognosis due to the high morbidity and mortality associated with neutropenia and hemorrhage.75,81 Dogs with aplastic anemia warrant a poor prognosis.75 Primary testicular tumors occasionally metastasize to regional lymph nodes and other distant sites, and therapy other than surgery may be warranted in these dogs. Optimal management employing chemotherapy, radiation therapy (RT), and novel targeted therapies is currently unknown. Cisplatin, actinomycin-D, chlorambucil, mithramycin, and bleomycin have been used; however, too few dogs were treated and evaluated to formulate conclusions regarding efficacy.70,71,73,101 Cisplatin was evaluated in three dogs with aggressive testicular tumors, with survival ranging from 5 months to greater than 31 months.70 RT was successfully used in four dogs with metastatic seminoma confined to the regional lymph nodes using total doses ranging from 17 to 40 Gy with 137Cesium teletherapy.102 In all four cases, tumors regressed and survival times ranged from 6 to 37 months; importantly, none of the dogs died of seminoma. Numbers were small, and further studies are warranted to evaluate the role of external-beam RT in managing metastatic seminomas because seminomas are considered extremely radioresponsive.103 In the United States, testicular cancer is the most common cancer in men 15 to 44 years old but is one of the most curable cancers with early diagnosis.104 Recent studies have shown an increase in testicular cancer over the past 40 years, suggesting that an individual’s risk is a function of the era in which he was born.104,105 Causal factors such as genetic predisposition, maternal estrogen exposure, occupational hazards, dietary factors, smoking habits, and birthplace have been evaluated, but, to date, the most established risk factor remains cryptorchidism.23,104,106–114 Most cancers in men are germ cell tumors and are divided into seminomas and nonseminomas, with the former comprising 50% of tumors in this group.13,20,23 Seminomas are further classified into classical, atypical, or spermatocytic seminoma, although management does not vary considerably between types.20 Standard staging in human seminomas consists of physical examination;, radiographic studies; determination of serum markers, including alpha-fetoprotein (AFP), human chorionic gonadotropin (hCG), and lactate dehydrogenase; and histopathologic assessment. Spermatocytic seminoma is a rare variant of germ cell neoplasia that is most commonly seen in older men and carries a low risk of metastasis, suggesting similar behavior to most canine seminomas.21,23 Treatment generally includes surgery for stage I seminomas, and/or RT and chemotherapy for individuals with advanced stage disease.23,103,115–117 Cisplatin-based chemotherapy protocols are generally employed for patients with greater than stage I disease, with cure rates in the range of 70% to 80% despite advanced tumor burdens.23,103,118 The dog has been proposed as a model for studies evaluating the development of testicular tumors.18,21,22 Further interrogation of the molecular pathogenesis, classification system, and behavior of canine tumors may yield further support for use of spontaneously occurring testicular tumors as good comparative models for human disease. Feline testicular tumors are rare, although Sertoli cell tumor, seminoma, interstitial cell tumor, and teratoma have been reported.34,119–125 The biologic behavior of testicular neoplasia in the cat is unclear due to the sparse literature available. Metastasis of a Sertoli cell tumor to the liver and spleen has been reported, and teratoma metastasis to the omentum was observed in another report.122,125 Optimal therapy other than orchiectomy is not known. Prostatic tumors are relatively uncommon in dogs and have a low prevalence at less than 1% (0.2% to 0.6%).126–130 In a collection of over 17,000 confirmed neoplasms of the dog collected from veterinary schools in North America, only 11 prostate carcinomas were identified (0.06%).33 In a separate study that evaluated lifetime occurrence of neoplasia in predominantly intact German shepherd and Belgian Malinois working dogs, over 30% developed at least one cancer; however, only 2 of the 104 primary tumors were prostate adenocarcinomas.17 Despite this low incidence, the dog is one of the few domestic species to develop spontaneous prostate cancer, thus sparking interest in the dog as a comparative model for prostate cancer in men.129,131,132 In three retrospective reviews of dogs with prostatic disease, between 7% and 16% were diagnosed with prostatic adenocarcinoma.133–135 One study of 177 dogs found that prostatic adenocarcinoma was the most common disease in neutered dogs, whereas bacterial prostatitis and prostatic cysts were more common in intact male dogs.134 Elderly dogs are more commonly diagnosed with prostatic carcinoma, with a median age at diagnosis of 10 years.129,135,136 The underlying etiology of canine prostatic cancer is unknown; however, high-grade prostatic intraepithelial neoplasia (PIN or HGPIN), which is believed to be a precursor of human prostate carcinoma, has been detected in dogs without evidence of prostatic disease and in those with existing prostatic carcinoma.137–139 The occurrence of PIN in dogs with concurrent carcinoma varies from 7% to 72%, although in two large studies of dogs without histologic evidence of prostatic carcinoma, the occurrence was low at 0% to 3%.128,137–139 Most tumors of the canine prostate are carcinomas, and the majority are adenocarcinomas. It is believed that prostate tumors in the dog arise from a urothelial or ductular origin rather than acinar because most canine tumors are androgen independent.140–145 Other types of carcinomas, including transitional cell carcinoma (TCC) arising from the prostatic ducts, mixed carcinomas, and squamous cell carcinomas, can occur. Classifying carcinomas on the basis of subtype is somewhat subjective, and there is no standard for definitive diagnosis of canine prostate tumors as there is with humans.139,146,147 Fibrosarcoma, leiomyosarcoma, osteosarcoma, lymphoma, and hemangiosarcoma have also been reported to affect the prostate.128,148–153 Benign tumors of the prostate are rare, although there is a single case report of a dog with a benign prostatic adenoma or nodular hyperplasia.154 TCC of the prostatic urethra will frequently invade the prostate, and it may be difficult to distinguish primary TCC from secondary invasion of a urethral tumor. Both intact and castrated dogs develop prostate carcinomas, although multiple studies have suggested there is an increased risk of prostatic adenocarcinoma in castrated male dogs compared to intact male dogs with an odds ratio of approximately 2.3 : 4.3.126,135,136,140,143 More aggressive tumors may develop in castrated males with a higher risk of metastasis.136 The reason for this difference is unclear, although it is possible that castrated dogs live longer than intact dogs and are thus predisposed to developing age-related cancers.135,143 It is also possible that androgens provide a protective effect on prostatic tissue or that, on castration, the relative estrogen effect aids in neoplastic transformation.143,155–157 A recent study observed that prostatic adenocarcinoma may occur relatively more frequently in intact male dogs, further complicating this issue.128,139,143 Although several studies have suggested that androgens may not be required for initiation or progression of adenocarcinoma of the canine prostate, further controlled studies should be done to definitively determine the role of androgens and the impact of early or late castration.140,142–145,158 Breeds that may be at increased risk of developing prostate carcinomas include the Bouvier des Flandres, Doberman Pinscher, Shetland sheepdog, Scottish terrier, beagle, miniature poodle, German shorthaired pointer, Airedale terrier, and Norwegian elkhound.135,143 The Shetland sheepdog and Scottish terrier remained at increased risk even when TCC was excluded in one study.143 Mixed breed dogs have also been reported as at increased risk for prostate carcinoma, regardless of neutering status, suggesting environmental influences may play a significant role in tumor development.143 The American cocker spaniel, miniature poodle, and dachshund may be at decreased risk for developing prostate cancer.143 At the time of diagnosis, most canine prostatic tumors are characterized by local invasion with a high propensity for regional and distant metastasis. In one postmortem study that retrospectively evaluated 76 dogs, 80% of those with prostate carcinoma had evidence of measurable metastatic disease, with lung and lymph node being the most common sites.128 Importantly, similar to high-grade prostatic carcinoma in men, canine prostatic carcinomas have a tendency to metastasize to bone and 22% to 42% of canine patients develop skeletal metastasis, predominantly to the lumbar vertebrae and pelvis.128,144,159,160 Younger dogs may be at increased risk for metastasis, although the role of castration status in this group is unclear.128,161 Longitudinal studies in dogs with evidence of PIN are not available, and it is unclear if prostate carcinoma can behave in a slowly progressive fashion in early phases of development. It is presumed that most dogs are diagnosed at an advanced stage of disease due to the high metastatic rate; however, the true behavior from time of onset is not definitively known and prostate carcinoma may behave differently in intact and castrated dogs.135,136 The underlying cause of prostate tumors is unknown, and it is possible that both genetic and environmental factors contribute to tumor development. As previously mentioned, HGPIN is considered a precursor of human prostate carcinoma and occurs under the influence of androgenic stimulation in those patients at risk for carcinoma.162 Although PIN has been detected in dogs with existing prostatic carcinoma, it has also been detected in dogs without evidence of prostatic disease, making its role in the dog less clear.137–139 HGPIN as a predictor of carcinoma occurrence is likely not as reliable in the dog as it is in men.138,155 It is not known with certainty if low- and intermediate-grade PIN occurs in dogs, although a recent immunohistochemical study suggested the presence of low-grade PIN.163 Investigators evaluated five prostates from middle-aged to older intact dogs containing lesions of PIN and compared nuclear protein p63 (marker of prostatic basal cells), androgen receptor expression, and PCNA to normal prostatic tissue from intact dogs. PIN foci had higher p63 expression, higher PCNA index, and heterogenous androgen receptor expression, suggesting similarities to human low grade PIN.163 The role of hormones in prostate development and tumor progression is also unclear in the dog. Castration does not provide a protective effect and, in fact, may contribute to tumor development and/or progression, although prostatic carcinoma may behave differently in the intact male compared to the neutered male.135,136 Normal prostate development and regulation is androgen dependent in both humans and dogs; however, neoplastic human prostate remains androgen dependent unlike in the dog. Androgen receptor expression within the nuclei can be identified in 90% to 95% of normal noncastrated prostatic secretory epithelial cells and in the majority of acinar basal cells.144,145,164,165 In neutered dogs and dogs with prostatic carcinoma, nuclear androgen expression decreases and is usually lost.144,145 The role of estrogen and progesterone has yet to be fully defined, although nuclear estrogen receptor expression appears to be decreased in prostatic carcinoma tissue compared to normal and hyperplastic prostate tissue.141,165 Chromosomal abnormalities of the neoplastic prostate have only recently been studied, and preliminary data suggest abnormalities such as aneuploidy, centromeric fusions, polysomy of chromosome 13, and hyperdiploidy may occur in the dog.166–168 Because of its aggressive behavior at the time of diagnosis, some investigators have considered mechanisms that may contribute to prostate carcinoma progression and metastasis. The role of cyclooxygenase (COX) has received considerable attention in the human and veterinary literature and inhibition of COX-2 may play a role in the management of prostate tumors. Expression of COX-2 was noted in 75% of prostate carcinomas in one study, whereas none of the normal prostate tissue had expression.169 Two other studies support the notion that COX-2 and its downstream prostaglandin E2 production play a role in prostate carcinomas.170,171 Indeed, a clinical study identified COX-2 protein expression in 88% of 16 prostate carcinomas examined and further showed a survival benefit in dogs treated with either piroxicam or carprofen.172 As stated earlier, prostatic carcinoma has a predilection for bone, which may be mediated in part by transforming growth factor-β (TGF-β), parathyroid hormone–related protein (PTHrP), and endothelin.173–175 PTHrP mediates pathologic bone resorption in many different tumors, including prostate carcinomas, which may encourage release of TGF-β into the microenvironment. In a positive feedback loop, canine prostatic carcinoma cells can increase gene transcription for PTHrP in response to exogenous TGF-β.174 Although PTHrP and TGF-β may be important in establishing skeletal metastases, it is interesting to note that prostate metastases are more commonly osteoblastic in nature. In a rat model, osteoblast activation was increased following incubation with normal canine prostate protein homogenates through an endothelin-dependent pathway, suggesting a possible contribution to bone metastasis formation.175
Tumors of the Male Reproductive System
Canine Testicular Tumors
Risk Factors
Pathology and Pathogenesis
Natural Behavior
History and Clinical Signs
Diagnostic Techniques and Staging
Treatment and Prognosis
Comparative Aspects
Feline Testicular Tumors
Canine Prostate Tumors
Risk Factors
Natural Behavior
Pathology/Pathogenesis
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Veterian Key
Fastest Veterinary Medicine Insight Engine