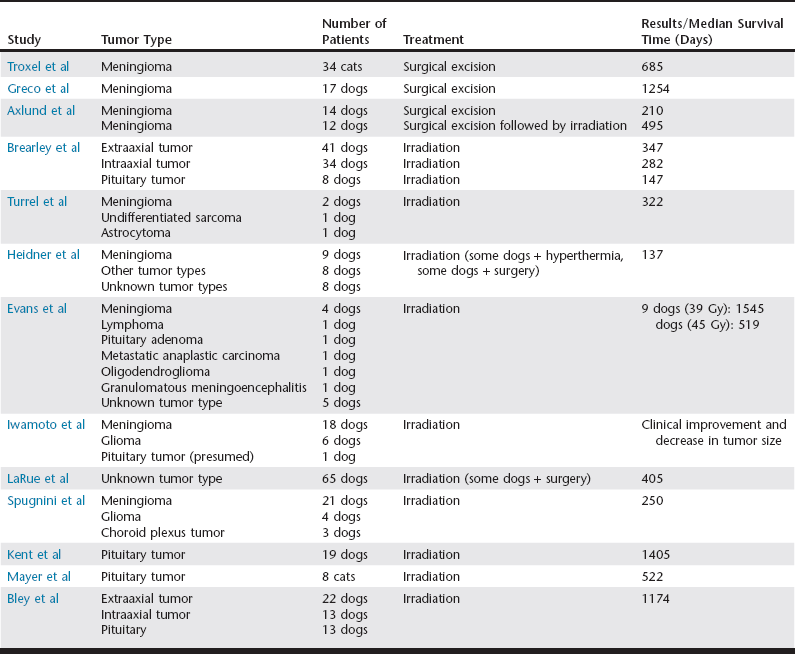
Chapter 227 All intracranial tumors carry a poor prognosis; however, often it is difficult to discuss prognosis because treatment sometimes is initiated without a definitive diagnosis. Definitive treatment relies on an accurate histopathologic tumor diagnosis, although correlations between tumor appearance on magnetic resonance imaging (MRI) and histopathologic diagnoses have been reported (Kraft et al, 1997; Snyder et al, 2006). The patient’s prognosis is related to tumor biologic behavior, as well as severity and progression of clinical signs. Without treatment intracranial tumors offer a grave prognosis. Surgical removal of an intracranial tumor achieves many treatment goals. There is decompression and reduction of intracranial pressure. Moreover, a histopathologic diagnosis can be obtained, which allows for accurate prognostication and additional treatment planning in the case of subtotal resection. Surgical approaches include rostrotentorial, caudotentorial, transfrontal, and suboccipital craniotomies or craniectomies, and combinations of these approaches may be used. The tumor size, degree of invasiveness, and location determine whether or not surgical removal is a viable option; these also guide the neurosurgical approach. Intraaxial tumors, which are located within the brain parenchyma, are more difficult to remove than extraaxial tumors. In addition, approaches to the caudal fossa are difficult and seldom attempted because of the possibility of inducing iatrogenic trauma to the brainstem, which can result in severe clinical signs. However, one recent case report describes a novel basio-occipital surgical approach to relieve compression of the caudal brainstem and cranial cervical spinal cord caused by a meningioma in a canine patient (Barreau et al, 2010). In most cases the goal of surgery is removal of the entire mass. Tumor removal can be achieved using a combination of sharp and blunt dissection, or an ultrasonic aspirator can be used. Adequate dissection can be difficult because many tumors are not well delineated from normal tissue, and adjacent normal brain tissue may be compromised because of peritumoral edema or hemorrhage. Endoscopy has been used to assist in removal of residual intracranial tumor after a debulking procedure (Klopp et al, 2009). This is particularly useful when removing a tumor from the olfactory and frontal regions of the brain as well as the cerebellum. In these procedures, an endoscope increases the visibility of regions that otherwise receive inadequate exposure in routine surgical approaches. The dura mater must be incised or removed to obtain adequate visualization of the underlying brain structures. The defect can be closed using a graft (synthetic or fascial), or it may be left open. In dogs and cats cerebrospinal fluid (CSF) typically does not cause complications when leaked into the surrounding tissues (Niebauer et al, 1991). To protect the underlying brain, the skull defect generally is replaced when using a transfrontal or radical rostrotentorial approach; replacement is not necessary when other approaches are used. In addition, these patients often have elevated intracranial pressure; thus the anesthetist should take measures to avoid or reduce intracranial hypertension, including maintaining normotension (systolic blood pressure of 110 to 160 mm Hg or mean arterial pressure of 80 to 110 mm Hg), eucarbia (35 to 45 mm Hg), and analgesia. Inhalant anesthetics increase cerebral blood flow (see Chapter 13), which can lead to or potentiate intracranial hypertension; injectable agents such as propofol and fentanyl can be used to decrease the requirement of inhalant anesthetics. Diuretics and glucocorticoids may be given to decrease brain edema (Table 227-1). Intracranial surgery can be associated with intraoperative hemorrhage; thus one should be prepared for a blood transfusion. After surgery the patient should be allowed to recover gradually from anesthesia. It is critically important to avoid excitement on recovery, and additional sedation may be required. Analgesia should be continued and titrated to the patient’s needs. TABLE 227-1 Medications Used in the Treatment of Intracranial Tumors Intraoperative complications include hemorrhage, hypotension, intracranial hypertension, and air embolism. Postoperative infection may be of concern after transfrontal craniotomy because the approach involves incision through the contaminated frontal sinus. However, postoperative infection is not a typical complication after intracranial surgery, possibly because of the routine use of perioperative antibiotics (see Table 227-1). Intracranial hypertension is a concern following surgery, and patient positioning can aid in maintaining intracranial pressure within the normal range. Intracranial pressure can be measured directly, or it can be monitored indirectly by observing for changes associated with intracranial hypertension, including Cushing’s response (systemic hypertension with reflex bradycardia) and changes in pupil size and symmetry. The patient’s head should be elevated (approximately 30 degrees), jugular occlusion should be avoided (e.g., no jugular venipuncture or neck leads), and pain and excitement should be prevented. If necessary, diuretics with or without glucocorticoids can be continued in the postoperative period. If the patient is recumbent, urinary catheterization may be necessary. Care should be taken to keep the patient clean and dry, and appropriate bedding with frequent rotation or placement in a sling to prevent the formation of decubital ulcers is required. To avoid aspiration pneumonia the patient should be given nothing by mouth for 24 hours postoperatively. Nutritional and intravenous fluid support is indicated in a patient who cannot maintain adequate nutrition orally. Surgical removal of feline meningiomas may be curative since the entire mass often can be removed in total. Accordingly, surgical excision is the treatment of choice for feline meningiomas. Troxel and associates (2003) reported that cats treated by surgical removal of meningiomas had a significantly longer survival time than cats treated by any other modality (Table 227-2). Niebauer and colleagues (1991) reported that 50% of cats were alive 2 years after surgery. TABLE 227-2 Comparison of Median Survival Times in Dogs and Cats with Intracranial Tumors Treated with Surgical Excision and/or Irradiation
Treatment of Intracranial Tumors
Definitive Therapies
Surgical Excision
Surgical Approaches and Considerations
Anesthetic Considerations
Drug
Dosage
Use/Indication
Carmustine
50 mg/ml q6wk IV (over 15-20 min)
Nitrosourea chemotherapeutic agent; may be used to treat meningioma and glioma
Cefazolin
22 mg/kg q90min IV
Perioperative antibiotic
Cytarabine (cytosine arabinoside)
20-100 mg/m2 q1wk intrathecally; 50 mg/m2 SC for 2 days. Repeat every 3 weeks
Chemotherapeutic agent used to treat CNS lymphoma
Diazepam and midazolam
0.5 mg/kg as needed IV; or 2 mg/kg as needed per rectum
Anticonvulsant used in emergency management of status epilepticus or cluster seizures
Dimenhydrinate
Dog: 25-50 mg q8-24h PO
Cat: 12.5 mg q8-24h PO
Antihistamine used as antiemetic in vestibular disease
Hydroxyurea
Dog: 50 mg/kg once daily, 3 days per week POCat: 25 mg/kg once daily, 3 days per week PO
Chemotherapeutic agent; may be used to treat meningioma
Levetiracetam
20 mg/kg q8h PO
20-60 mg/kg IV for status epilepticus
Anticonvulsant
Lomustine
Dog: 60-90 mg/m2 q3-6wk PO
Cat: 50-60 mg/m2 q6wk PO
Nitrosourea chemotherapeutic agent; may be used to treat meningioma and glioma
Mannitol
0.5-1 g/kg q4h or as needed IV
Osmotic diuretic used to decrease brain edema and lower intracranial pressure
Meclizine
25 mg/dog and 12.5 mg/cat q24h PO
Antihistamine used as antiemetic in vestibular disease
Methylprednisolone sodium succinate
30 mg/kg once IV; or 100 mg/kg given over 24 hr IV
Glucocorticoid used to decrease peritumoral brain edema and lower intracranial pressure
7.5% NaCl
5-20 ml/kg as needed IV
Osmotic diuretic used to decrease brain edema and lower intracranial pressure
Phenobarbital
Loading dosage: 5 mg/kg as needed IV (up to 20 mg/kg total)
Maintenance dosage: 2-8 mg/kg q12h PO (Adjust dose by monitoring blood concentrations)
Anticonvulsant
Potassium bromide
Loading dosage: 200 mg/kg per day every 3-5 days
Maintenance dosage: 30-40 mg/kg q24h PO
Anticonvulsant
Prednisone
0.5-1 mg/kg q12h PO
Glucocorticoid used for supportive treatment of intracranial tumors
Zonisamide
10 mg/kg q12h PO
Anticonvulsant
Intraoperative and Postoperative Considerations
Meningioma
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Treatment of Intracranial Tumors
Only gold members can continue reading. Log In or Register to continue
