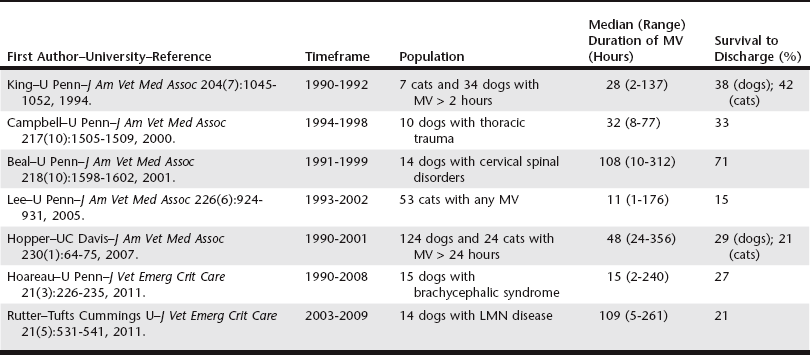
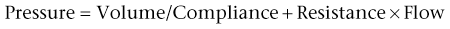Chapter 11 There are only three large retrospective studies of MV in veterinary medicine (Table 11-1). Another set of four studies researched specific populations of canine patients (see Table 11-1). The prognosis for survival to discharge varies greatly depending on the primary disease process. Survival to discharge is reported to be up to 86% for animals with toxicoses, who are usually younger patients with reversible causes and no pulmonary parenchymal disease; 40% for those with pulmonary contusions; and 11% for post–cardiopulmonary resuscitation (CPR) patients (Hopper, 2007). The overall prognosis ranges from 21% to 71% in dogs and from 15% to 42% in cats (see Table 11-1). Selection of drugs for anesthetic induction and sedation depends on the clinician’s preference and the patient’s status (see Chapter 13). Some unconscious patients may not require drugs, but most will not tolerate MV without sedation. Various options are available. Most clinicians use a combination of benzodiazepine and an opioid and then add propofol, barbiturates, ketamine, or dexmedetomidine as needed. For the benzodiazepines, midazolam is preferred over diazepam because diazepam binds to plastic syringes and infusion lines, can precipitate with other drugs, and is less available. Fentanyl is usually the opioid of choice, at “anesthetic doses” of 6 to 20 µg/kg/hr. Propofol is generally a safe anesthetic but its use generates some concerns, notably involving volume, cost, hyperlipidemia, and bacterial contamination because of the soy-based carrier. Long-term propofol use in cats is discouraged because it can cause Heinz body anemia. Pentobarbital was most widely used (65% to 80%) in veterinary medicine in the 1990s but has become obsolete. Although induction and plane of anesthesia are smooth on barbiturates, tremors and seizures can potentially occur at the time of weaning. Ketamine has some cardiovascular sparing effects and requires small volumes when administered as a constant-rate infusion. Dexmedetomidine has cardiovascular side effects including bradycardia, hypotension, and vasoconstriction but can be used at “microdoses” to enhance analgesia and sedation and to decrease the opioid and propofol requirement in some patients. • Mode A/C (or CMV) to provide maximum ventilatory support until the patient is stabilized. Some clinicians initially use “hybrid” modes such as SIMV. • Peak airway pressure of 15 to 25 cm H2O. • Tidal volume of 10 ± 2 ml/kg, although in severe hypoxemic failure (“protective lung strategy”), tidal volumes are aimed for smaller than normal volumes (4 to 6 ml/kg). • Respiratory rate of 20 breaths per minute. • Inspiratory time of 1 sec, giving an inspiratory : expiratory ratio of 1 : 2. • PEEP 2 to 5 cm H2O, although higher levels of PEEP can recruit and prevent derecruitment of alveolar units (“open lung strategy”). Providing early nutrition is essential for the ventilated patient. Ventilated patients can be fed enterally or parenterally. The combination of central parenteral nutrition and enteral nutrition can provide the patient’s full caloric requirement and promote enterocyte health (see Chapter 7). Enteral nutrition may also decrease the risk of bacterial translocation. When enteral feeding is instituted, residual gastric contents should be monitored and a promotility agent added. With a tracheostomy, MV patients often can tolerate the machine with mild sedation, eliminating the need for anesthesia. Tracheostomy also avoids complications associated with the ET tube including ranula and macroglossia. The reported tracheostomy rate in veterinary medicine is 20% to 30%, a number similar to that for human patients, but is increased to 70% for patients in ventilatory failure. A high incidence of complications is reported in cats with tracheostomy, so appropriate warning and proper care are warranted (Box 11-1).
Ventilator Therapy for the Critical Patient
Prognosis
Placing a Patient on the Mechanical Ventilator
Choosing the Correct Settings of the Mechanical Ventilator
Patient Care
Ventilator Therapy for the Critical Patient