Thoracic Imaging and Respiratory Surgery
Imaging of the Thorax
The indications for thoracic radiography in llamas and alpacas are many. Thoracic radiography provides a valuable assessment of any patient with suspected cardiac or pulmonary disease.1 Abnormalities revealed by physical examination, such as cardiac murmurs or harsh or dull lung sounds, warrant further investigation with thoracic radiography. When evaluating a dyspneic animal, normal thoracic radiographs indicate upper airway disease is a more likely cause of clinical signs. Primary respiratory disease such as pneumonia can be differentiated from congestive heart failure on the basis of radiography results.1 Animals with increased white blood cell (WBC) counts or fibrinogen may have thoracic radiography performed during the search for a septic focus. Thoracic radiography is an important screening tool for metastatic disease in patients with neoplasia and also helps define prognosis in these cases.2 As camelids tend to mask illness and clinical signs present late in the disease process, diagnosis of multisystemic disease may be difficult.3 Thoracic radiography may be a useful early screening test for occult thoracic disease resulting in nonspecific clinical signs such as weight loss or decreased feed intake.
Technical Aspects
Thoracic radiographs are acquired during full inspiration. Underinflated lungs may appear abnormally and diffusely opaque (particularly caudodorsally) and result in an improper diagnosis of pulmonary pathology. True pulmonary pathology (such as pneumonia or abscesses) may be difficult or impossible to identify on radiographs if the surrounding lung is not well inflated to provide good contrast. The position of the dorsal lumbodiaphragmatic recess is a useful marker in the assessment of pulmonary inflation. In well-inspired radiographs, this recess should be positioned between the level of the tenth and twelfth thoracic vertebrae. Pulmonary underinflation results in displacement of this recess cranial to this level.4
Given the large size of the adult camelid thorax, relatively high radiographic technique (i.e., kilovolt peak [kVp] and milliampere second [mAs]) is required for proper exposure. Fixed (i.e., nonportable) x-ray tubes are capable of high kVp and mAs settings and are recommended for routine thoracic radiography of adult llamas and alpacas. Thoracic radiography of adults may be attempted with portable x-ray units, particularly in young adult alpacas, but these units require longer exposure times, which may result in significant motion artifact. The caudal thorax predominantly contains air-filled pulmonary parenchyma, and the cranial thorax contains dense forelimb musculature, mediastinum, and heart. Thus, radiographic exposure factors are usually fewer for the caudal thorax than for the cranial thorax. Typical radiographic exposure ranges are listed in Table 59-1, but caution should be used with these techniques, as particular settings will depend on the x-ray tube type and the imaging system employed. Use of a cassette stand facilitates alignment of the radiographic cassette and x-ray beam and also prevents unnecessary exposure of personnel.
TABLE 59-1
Typical Exposure Variables for Adult Camelids and Crias

*With 12:1 grid, 800 system speed.
Significant scatter radiation is generated during exposure of an adult camelid thorax. Scatter radiation is an unavoidable effect of imaging thick body parts at high kVp techniques and is undesirable as it degrades image quality. A grid attenuates scatter radiation, improving image contrast and quality, but necessitates the use of a higher mAs technique. If a grid is not available, an air gap (a space between the patient and the cassette) of 5 to 25 centimeters (cm) also attenuates scatter radiation and may improve image contrast but results in patient magnification.5
Neonatal camelids are smaller and more compliant, making radiography of crias technically easier than in adults. Crias are usually imaged while recumbent, rather than standing as in adults, allowing excellent positioning and easy x-ray tube and cassette alignment. If available, small animal table-based x-ray systems may be employed to image these patients. Sedation is rarely necessary, and both lateral and dorsoventral or ventrodorsal projections may be routinely attained. Example x-ray exposure variables are listed in Table 59-1. Both left and right lateral projections should be routinely acquired. When recumbent, the dependent (i.e., “down”) lung is less aerated than the nondependent (i.e., “up”) lung. For this reason, mild pathology such as early bronchopneumonia in the dependent lung may not be seen radiographically. Dorsoventral projections are often less sensitive to the detection of small or subtle pathology, particularly if it overlaps the heart, making the acquisition of both left and right lateral projections paramount.
Mediastinum (Including Heart and Great Vessels)
Normal anatomy of the heart in adult llamas and alpaca crias has been described.4,6 In both crias and adults, the long axis of the heart should be generally parallel with the ribs and perpendicular to the spine (Figures 59-1 and 59-2).4,6 The heart has a tapered shape, with a broad base and a narrow apex, on lateral projections. The heart does not vary appreciably between left and right lateral recumbent projections in crias; however, the heart has either a rounded or a more angular shape on dorsoventral and ventrodorsal projections.
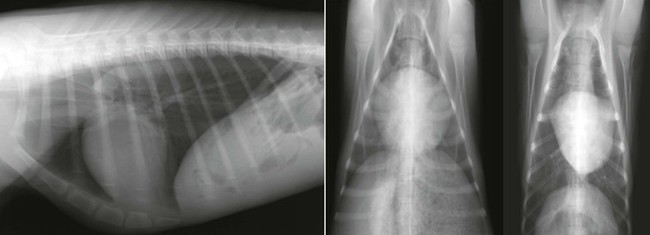
Figure 59-2 Right lateral, dorsoventral, and ventrodorsal projections of different normal alpaca crias. Note the rounded appearance to the heart on the dorsoventral projection and the more angular shape of the heart on the ventrodorsal projection.
Very few studies regarding radiographic changes to the heart with different cardiovascular diseases in camelids have been published; however, some general principles apply. The left atrium is not normally appreciated in camelids as a distinct structure, but if enlarged will appear as a protrusion of the heart just caudal to the carina.1 Left ventricular enlargement will result in increased cardiac height on lateral projections, with dorsal deviation of the carina. Right-sided cardiomegaly frequently results in the appearance of a widened heart on lateral projections, with increased contact of the sternum. Regardless of cause, detection of cardiomegaly necessitates the performance of echocardiography to better characterize cardiac abnormalities.
Prioritization of differential diagnoses for radiographic enlargement of the heart depends on which cardiac chambers are enlarged, signalment, and clinical findings. Congenital anomalies are the primary differentials for juveniles with cardiomegaly. Right-sided enlargement is expected with tricuspid valve atresia and tricuspid valve dysplasia but has also been described in other more complex anomalies.1,7,8 Left-sided cardiomegaly is expected with most types of ventricular septal defects (VSDs) and patent ductus arteriosus (PDA). Acquired cardiac diseases are the primary differential diagnoses for cardiomegaly in adult camelids and include nutritional muscular dystrophies, cardiotoxin ingestion (such as Nerium oleander or ionophore exposure), and infectious endocarditis and myocarditis.1,9–11 Other than nutritional deficiencies, primary diseases of the myocardium are rare. Reports of poor systolic function (a phenotype compatible with dilated cardiomyopathy in other species) have been described but are likely secondary to previous myocarditis.1
Experienced observers typically rely on subjective impressions of cardiac size when evaluating the heart, but quantitative measurement may be helpful in questionable cases or for the inexperienced evaluator. The heart in both crias and adults spans two to three intercostal spaces on lateral projections. Although this rule is easily applied, it may not be particularly sensitive for detection of mild cardiac enlargement.1 A common method of assessing heart size is the vertebral heart scale (Figure 59-3). For this method, the long and short axes of the heart are measured. These two lengths are then compared with the spine (starting with the fourth thoracic vertebral body), and the number of vertebrae spanned by these lengths are recorded to the nearest 0.1 vertebrae. The number of vertebrae spanned by these two measurements is the vertebral heart scale.12 In alpaca crias, the mean vertebral heart scale is 9.4, being between 8.1 and 10.7 in most animals. Because of the size of camelids, the entire thoracic spine cannot be included on one thoracic radiograph in adult llamas and alpacas, so a modification of the vertebral heart scale is necessary. The cardiac long and short axes are summed and divided by the length of the third to fifth vertebral bodies. The mean modified heart score in adult llamas is 2.8, being between 2.5 and 3.1 in most animals. It should be noted that the vertebral heart scale has been questioned as a diagnostic tool in other species.13 The vertebral heart scale may differ between different measurers and the normal range may exclude normal animals and include some with abnormalities of the heart. This rule is probably best applied with caution and used as an additional piece of evidence when evaluating the heart.
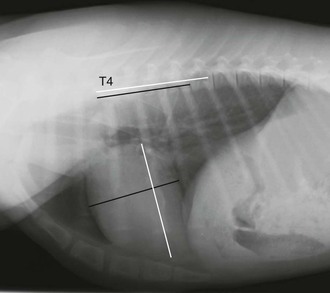
Figure 59-3 Lateral projection of a normal alpaca cria with lines indicating the normal positioning of lines drawn for the vertebral heart scale. A label has been placed at the level of the fourth thoracic vertebral body. The vertebral heart scale in this cria is approximately 8.7.
Evaluation of the caudal vena cava is important in animals that are suspected to right heart dysfunction.1 The caudal vena cava is visible in all llamas and alpacas as a structure extending cranioventrally to the heart from the diaphragm. An ascending course of the caudal vena cava may indicate enlargement of the heart or displacement of the heart by a mass or atelectasis.1 An objective criterion for the caudal vena cava diameter has been established in camelids. In adult llamas, the height of the caudal vena cava should be no greater than approximately the height of the fourth thoracic vertebral body (typical range 0.6–1.0). A caudal vena cava diameter greater than this range indicates congestion, which is often associated with right heart failure.1 The margins of the descending aorta are indistinct on lateral projections. Cranial mediastinal great vessels (such as the cranial vena cava or the brachiocephalic trunk) are not seen unless gas is present within the mediastinum.
The trachea is contained within the mediastinum and is easily seen on radiographs. The trachea in camelids typically diverges from the spine at an angle of approximately 15 degrees. Dorsal displacement of the caudal thoracic trachea may occur with cardiomegaly, and dorsal displacement of the cranial thoracic trachea or caudal cervical trachea may be seen with cranial mediastinal masses.1 The camelid trachea has unique anatomic features. A “tracheal bronchus” is present in camelids, representing the primary lobar bronchus to the right cranial lung lobe, similar to that present in the bovine. In many camelids, a focal dorsoventral increase in tracheal diameter is seen at this level, which in some animals may be quite prominent. This should not be misdiagnosed as tracheal pathology.
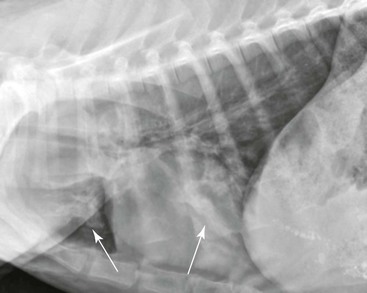
The esophagus is not normally seen radiographically in neonatal or adult camelids. In some normal animals, a small amount of gas may be seen in a portion of the esophagus secondary to normal intermittent regurgitation associated with rumination or transient aerophagia, but this should not be repeatable on additional projections of the same area. Megaesophagus is diagnosed if the esophagus persistently appears moderately to severely distended with gas, saliva, or feed material (Figure 59-4). The esophageal dilation should be categorized as generalized if the entire esophagus is involved or segmental if only focal dilation is present. Generalized megaesophagus may be secondary to toxin exposure, esophagitis, choke, stricture, neoplasia, or lodged foreign objects of the distal esophagus.14 Generalized megaesophagus has also been reported as an iatrogenic side effect of shock collar overuse.15 Segmental megaesophagus may be seen with more proximal strictures, neoplasia, or secondary to cervical trauma such as carotid hematoma or esophageal puncture during venipuncture attempts. Vascular ring anomalies should be the first differential diagnosis considered in a young animal with segmental megaesophagus cranial to the heart base, whose clinical signs start soon after weaning.16
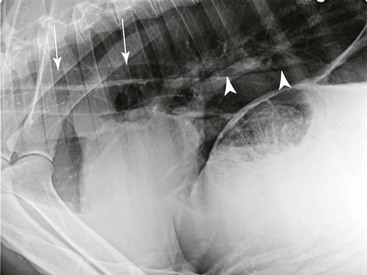
Figure 59-4 Lateral projection of an adult llama with mild megaesophagus. The thoracic esophagus is distended with gas (the dorsal esophageal margin is indicated by arrows). Fluid is pooled within the caudal thoracic esophagus and has a horizontal dorsal border (indicated by arrowheads).
Pulmonary Parenchyma and Pulmonary Vasculature
When evaluating lungs, the clinician should first determine if the parenchyma is normal. Normal pulmonary parenchyma should be relatively dark compared with the pulmonary vasculature. Large bronchial walls are normally seen near the carina, but peripheral, small bronchi are usually not seen. Many technical or patient factors may hinder this determination. Underexposure or poor pulmonary inflation results in relatively opaque pulmonary parenchyma. Superimposed fat in an obese animal may increase overall lung opacity. Overlying fiber, particularly if wet, may add significantly to lung opacity, sometimes with a bizarre appearance (Figure 59-5).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree