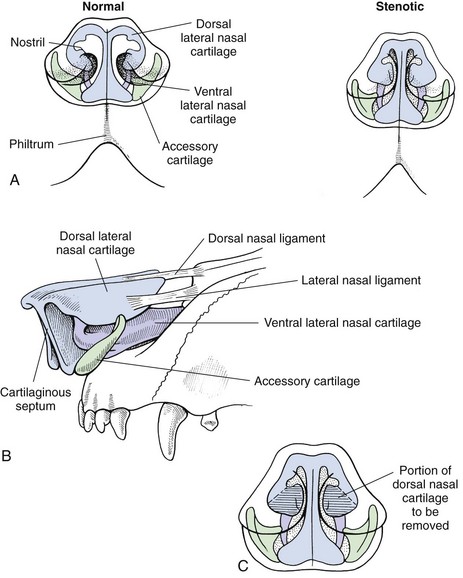
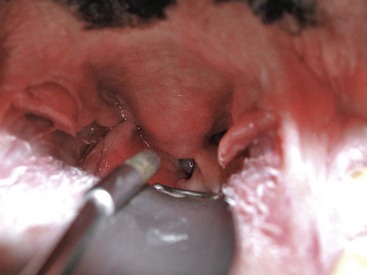
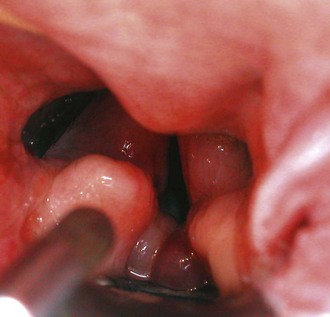
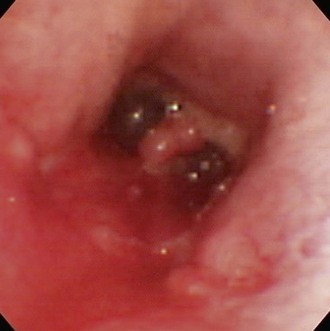
Chapter 29 General Principles and Techniques Upper airway procedures are performed to remove, repair, or bypass areas of obstruction, injury, or disease (Box 29-1). Affected animals may have mild to severe respiratory distress. Mild or moderately dyspneic patients initially should be examined from a distance to prevent exacerbating the condition. Open-mouth breathing, abducted forelimbs, labored breathing, and restlessness indicate moderate to severe respiratory distress that may require emergency therapy. Minimal restraint should be used with severely dyspneic patients, and they should be allowed to maintain the position in which they feel most comfortable. Supplemental oxygen may be given by means of nasal insufflation, tracheostomy tube or catheter, endotracheal intubation, flow-by, face mask (including an Elizabethan collar that has had plastic wrap put over it to create an oxygen-rich environment [see Fig. 4-2]), or oxygen cage (see p. 32). Corticosteroids, sedation, and/or cooling may help relieve some of the distress. Mild sedation may be beneficial for anxious patients (especially those with upper airway obstruction) with moderate to severe respiratory distress. Combinations of intravenous drugs are commonly given; hydromorphone or butorphanol and either acepromazine or diazepam are frequently used in dogs (Box 29-2). In cats, acepromazine or diazepam is recommended (Box 29-3). To cool dyspneic animals, a fan may be directed at the patient; ice packs may be applied to the head, axilla, inguinal area, and extremities; and/or cooled fluids may be administered intravenously. Animals with nasal neoplasia, fungal infection, or foreign bodies may be anemic as a result of profuse epistaxis. Affected animals should be carefully evaluated for clotting abnormalities by assessment of platelet numbers, bleeding from venipuncture sites, or the presence of ecchymosis, petechiation, melena, hematuria, or retinal hemorrhage. If available, coagulative ability may be assessed by determining the activated clotting time, prothrombin time, partial thromboplastin time, and/or mucosal bleeding time. Blood transfusions should be given before surgery if the packed cell volume (PCV) is 20% or lower (see Box 4-1 on p. 30). Bleeding during rhinotomy may be severe, requiring intraoperative blood transfusion or carotid artery ligation, or both. Patients with upper respiratory obstruction or disruption are extreme anesthetic risks. The periods of greatest danger are during induction of anesthesia and during recovery (see p. 921 under “Postoperative Care and Assessment”). For laryngeal examination, care should be taken to avoid drugs that inhibit laryngeal function. If the animal has already been sedated (see previous discussion), an anticholinergic drug (atropine or glycopyrrolate; Table 29-1) should be given if the patient has increased oral secretions or bradycardia. Preoxygenating the patient for 3 to 5 minutes before induction will provide a few extra minutes for examination of the larynx. If the procedure will be more extensive than a laryngeal examination, an opioid (e.g., hydromorphone, butorphanol, buprenorphine) may also be administered at induction; however, it may be best to wait until after intubation to administer these. Propofol is recommended for induction because it has a rapid onset of action and is short-acting. If necessary, it may be given in small incremental doses that maintain laryngeal function (Table 29-2). Even if laryngeal function is temporarily depressed with a rapid bolus of propofol, return to function usually occurs within several minutes. Meanwhile the patient can be intubated and given oxygen, and another look in the oropharynx can be done in a few minutes. In patients that have less respiratory compromise, a combination of diazepam and ketamine may be used for induction because these drugs maintain laryngeal function. Doxapram (Dopram, 1 to 2.2 mg/kg IV) administration during laryngeal examination increases intrinsic laryngeal motion and helps to differentiate normal dogs from dogs with functional laryngeal disease (Tobias et al, 2004). Oxygen should be supplemented during the examination, and oxygen saturation should be monitored with pulse oximetry and by observation of mucous membrane color. As soon as the laryngeal examination is complete, the patient should be intubated and anesthesia maintained with an inhalant anesthetic and oxygen for additional diagnostics or surgery, or to facilitate a smooth recovery. Anesthetic Considerations for Upper Airway Examination *For a short and minimally painful procedure, midazolam is a better choice than diazepam because of the much longer half-lives of diazepam and its active metabolite. Anesthetic Considerations for Patients With Brachycephalic Syndrome *Monitor for hyperthermia in cats. †Buprenorphine is a better analgesic than morphine in cats. ‡For a short and minimally painful procedure, midazolam is a better choice than diazepam because of the much longer half-lives of diazepam and its active metabolite. §Black box warning added by the FDA in October 2010 identified cases of renal failure and death in cats with repeated uses of meloxicam. Meloxicam is approved for single use only in cats in the United States. General anesthesia is preferred for most upper respiratory procedures because it ensures a patent airway, allows controlled ventilation, facilitates asepsis, and is less stressful for patients. Local anesthesia may allow placement of a tracheostomy tube when the patient is comatose or cannot tolerate general anesthesia. Respiratory patients should be managed with extreme care until intubation has been accomplished and ventilation can be assisted. Premedication with any drug that causes hypoventilation is contraindicated in moderately to markedly dyspneic patients. Additionally, every attempt should be made to minimize stress to the patient before induction. Oxygen support, even during placement of an intravenous catheter, may be necessary. Pulse oximetry with a sensor that can be secured to the tail can be especially helpful with perioperative monitoring of the patient in respiratory distress. Before the time of induction, the anesthesia provider should be prepared with airway devices, an anesthesia machine, monitors, and induction and emergency drugs. Preoxygenating the patient for 3 to 5 minutes usually provides adequate oxygen reserve for a safer intubation period. The animal should be allowed to remain in sternal recumbency, and oxygen should be provided by face mask, flow-by, or nasal insufflation if the animal will tolerate it (see Chapter 4). Induction should be rapid (e.g., propofol, thiobarbiturate, etomidate), and intubation should be performed immediately. Mask induction is not recommended in dyspneic patients. Anesthesia should be maintained with an inhalant anesthetic and oxygen. Laryngeal or tracheal procedures may require temporary retraction of the endotracheal tube from the surgical site, placement of an endotracheal tube distal to the surgical site through a tracheotomy, or use of injectable anesthetic drugs. When breathing spontaneously during surgery, the animal may be sighed (given an extra-large breath) every 5 minutes to re-expand distal airways. Oxygen saturation or blood gases (or both) should be monitored from induction until recovery, and until abnormalities have been corrected. Selected anesthetic protocols are provided in Table 29-2. Because the respiratory tract has a normal bacterial flora, prophylactic antibiotics (e.g., cefazolin; Box 29-4) frequently are given before surgery. However, animals with normal immune function undergoing short procedures (e.g., nares resection, laryngeal saccule resection, vocal cordectomy) do not need them. Streptococci, Escherichia coli, Pseudomonas spp., Klebsiella spp., and Bordetella bronchiseptica are the bacteria most commonly isolated from normal dogs. A majority of tracheal cultures are sterile, whereas most pharyngeal cultures are not. The Gram-negative organisms that cause most canine respiratory tract infections are often resistant to commonly used antibiotics. Antimicrobial drug selection is best based on cytologic and culture results of tracheobronchial, pulmonary parenchymal, and/or pleural secretions. Bland aerosol therapy (e.g., sterile 0.9% saline) helps loosen secretions and facilitates their clearance in dogs with tracheostomies; addition of antibiotics to the aerosol generally is unnecessary. However, intratracheal or aerosolized antibiotics may be effective in some dogs with chronic respiratory infection. Lipid-soluble antibiotics that contain a benzene ring reach the highest levels in the normal trachea and bronchus; however, increased permeability associated with inflammation allows numerous antibiotics to achieve high levels during infection. Antibiotics commonly recommended for treatment of upper respiratory disease include ampicillin, fluoroquinolones, cephalosporins, doxycycline, azithromycin, and potentiated sulfonamides (see Box 29-4). The nasal cavity extends from the nostrils to the nasopharyngeal meatus and is separated into two halves by the nasal septum (Fig. 29-1). The septum is mostly cartilaginous but also has bony and membranous portions. The nasal conchae develop from the lateral and dorsal walls of the nasal cavity. The air passages between the conchae are known as the meatus. The paranasal sinuses include a maxillary recess, a frontal sinus, and a sphenoidal sinus. The frontal sinus occupies the supraorbital process of the frontal bone (see Fig. 29-1). The two sides are separated by a median septum, and in dogs each side is divided into rostral, medial, and lateral compartments. The thyroid cartilage forms the ventral and lateral walls of the larynx (Fig. 29-2). It surrounds the lateral aspect of the cricoid cartilage and articulates with the dorsolateral aspect of the cricoid cartilage (caudal) and thyrohyoid bones (cranial). Ventrally the cricothyroid ligament joins the caudal border of the thyroid cartilage to the cricoid cartilage. The cricoid cartilage is a complete ring that is five times wider dorsally than it is ventrally (see Fig. 29-2). It forms the dorsal wall of the larynx and cranially lies within the wings of the thyroid cartilage. The cricoid cartilage articulates at its cranial dorsolateral margin with the arytenoid cartilage, which is paired (see Fig. 29-2). At the entrance to the glottis (laryngeal inlet), the arytenoid cartilages have two cuneiform processes ventrally and two corniculate processes dorsally. The vocal fold attaches to the vocal process of the arytenoid at its ventral aspect. The muscular processes are dorsolateral at the caudal aspect of the arytenoid. The glottis (laryngeal inlet) consists of the vocal folds, the vocal processes of the arytenoid cartilages, and the rima glottidis (see Fig. 29-2). The vocal folds extend dorsally from the vocal processes of the arytenoids to the thyroid cartilage ventrally. Rostral and lateral to the vocal folds are the laryngeal ventricles, or saccules. The laryngeal saccules are mucosal diverticula bounded laterally by the thyroid cartilage and medially by the arytenoid cartilage. The vestibular fold (false vocal cord) forms the rostral border of the laryngeal saccule and attaches to the cuneiform process. Surgical techniques for the management of animals with upper respiratory disease include rhinotomy (see later), tracheotomy (see p. 915), tracheostomy (see p. 916), tracheal resection and anastomosis (see p. 918), ventriculocordectomy (see p. 919), tracheoplasty (see p. 941), arytenoid cartilage lateralization (see p. 935), partial laryngectomy (see p. 936), and surgery for stenotic nares (see p. 928), elongated soft palate (see p. 928), and everted laryngeal saccules (see p. 929). With the animal in ventral recumbency, make a dorsal midline skin incision from the caudal aspect of the nasal planum to the medial canthus of the orbit. Either or both sides of the nasal cavity can be entered through a single midline skin incision. To explore the frontal sinus, extend the incision caudal to a line that connects the zygomatic processes of the frontal bone. Incise the subcutaneous tissue and periosteum on the midline. Elevate the periosteum and reflect it laterally on either or both sides of the nasal cavity. Use a bone saw to create and then elevate a flap of bone over the proposed site of entry into the nasal cavity (Fig. 29-3, A). Save the bone flap (if healthy) and replace it after the nasal cavity has been explored. As an alternative, drill a hole to one side of the nasal septum with a Steinmann pin. Use rongeurs to enlarge the hole, and discard the bone fragments. If necessary, extend the bone removal bilaterally. Gently lavage the nasal passages and remove abnormal tissue. Submit tissues for histologic examination and culture. Use cautery, iced saline, and/or digital pressure to control the hemorrhage. If continued hemorrhage is a problem, pack the nasal cavity with cotton gauze (see p. 921). If a bone flap was made, suture it in place with 3-0 or 4-0 wire placed through predrilled holes in the bone flap and adjacent bone (Fig. 29-3, B). Do not use wire to replace the bone flap if radiation therapy is planned. Close the periosteum and subcutaneous tissues with absorbable suture material in a simple continuous pattern. Close the skin routinely. If rongeurs were used, close the periosteum and subcutaneous tissues, leaving a stoma at the caudal aspect of the incision. Close the skin similarly, leaving a stoma (Fig. 29-3, C). With the animal in dorsal recumbency (Fig. 29-4), make a midline incision in the hard palate. Elevate the mucoperiosteum of the hard palate laterally to the alveolar ridge. Be careful to spare the palatine nerves and vessels as they emerge from the major palatine foramen. Incise the mucoperiosteum and soft palate attachments to the caudal edge of the palatine bone, and extend the incision as far caudally as necessary into the soft palate (full thickness). Retract the edges of the incision with stay sutures. Remove the palatine bone with a power-driven burr or rongeurs and discard it (Fig. 29-5, A). Explore the nasal cavity and remove abnormal tissues. Submit tissues for histologic examination and culture. Close the nasal mucosa of the soft palate with absorbable material in a simple continuous or simple interrupted pattern. Then close the submucosa-periosteum of the hard palate with absorbable suture in an interrupted pattern (Fig. 29-5, B). Finally, close the oral mucosa of the hard and soft palates with monofilament nonabsorbable sutures in a simple continuous pattern. Make an incision with a scalpel or Mayo scissors. When using scissors, insert one blade into the nostril and position it so the incision will be made ventral to the nasal planum and the dorsal lateral nasal cartilage (Fig. 29-6). Angle the incision in a dorsocaudal direction toward the nasomaxillary notch. Incise through all layers and retract the tissue dorsally to expose the vestibule. Explore and resect or biopsy abnormal tissue. Appose the nasal mucosa with 3-0 or 4-0 monofilament absorbable suture. Place two to four sutures in the musculocartilage layer (3-0 or 4-0 monofilament absorbable), and then reappose the skin (3-0 or 4-0 monofilament nonabsorbable). This approach has been used to remove foreign bodies and may be useful for removal or biopsy of rostral nasal masses. Palpate the ridge along the rostrolateral aspect of the right (or left) nasal and incisive bone (Fig. 29-7). Incise the alveolar mucosa along this ridge from the nasal bone to the rostral end of the interincisive suture. Using a periosteal elevator, reflect the mucosa from these bones. Then retract the dorsal lateral nasal cartilage and the ventral lateral nasal cartilage medially. Penetrate the nasal mucosa and explore the rostral nasal cavity. Biopsy abnormal tissue and lavage the area. Close the gingival and buccal mucosa with 3-0 or 4-0 monofilament absorbable suture. Tracheotomy is performed to gain access to the tracheal lumen to remove obstructions, collect specimens, or facilitate airflow. The tracheal incision may be closed or allowed to heal by secondary intention. Position the patient as described on p. 920. Approach the cervical trachea through a ventral cervical midline incision. Extend the incision from the larynx to the sternum as needed to allow adequate exposure. Separate the sternohyoid muscles along their midline, and retract them laterally (Fig. 29-8, A). Dissect the peritracheal connective tissue from the ventral surface of the trachea at the proposed tracheotomy site. Take care to prevent traumatizing the recurrent laryngeal nerves, carotid artery, vagosympathetic trunk, jugular vein, thyroid vessels, or esophagus. Immobilize the trachea between the thumb and the forefinger. Make a horizontal or vertical incision through the wall of the trachea (see Fig. 29-8, A). Place cartilage-encircling sutures (2-0 polypropylene [Prolene] or polybutester [Novafil]) around adjacent cartilages to separate the edges and allow lumen inspection or tube insertion. Suction blood, secretions, and debris from the tracheal lumen. After completion of the procedure, appose the tracheal edges with simple interrupted 3-0 or 4-0 polypropylene sutures. To close the tracheal incision, place sutures through the annular ligaments encircling adjacent cartilages or through the annular ligaments only. Lavage the surgical site with saline. Appose the sternohyoid muscles in a simple continuous pattern with 3-0 or 4-0 absorbable suture (e.g., polydioxanone [PDS], polyglyconate [Maxon], poleglicarpone 25 [Monocryl], glycomer 631 [Biosyn], polyglactin 910 [Vicryl]). Appose the subcutaneous tissues and skin routinely. Make a ventral midline incision from the cricoid cartilage extending 2 to 3 cm caudally. Separate the sternohyoid muscles and make a horizontal (transverse) tracheotomy through the annular ligament between the third and fourth or fourth and fifth tracheal cartilages (see Fig. 29-8, A). Do not extend the incision around more than half the circumference of the trachea. As an alternative, make a vertical tracheotomy across the ventral midline of cartilages 3 through 5. Place cartilage-encircling sutures (2-0 polypropylene [Prolene] or polybutester [Novafil]) around adjacent cartilages to separate the edges and allow for tube insertion. Suction blood and mucus from the lumen, widen the incision if necessary, and insert the tracheostomy tube. Facilitate tube placement by opening a hemostat in the incision, or depress the cartilages cranial to the horizontal incision (Fig. 29-8, B). Alternatively, place tension on the caudal suture to open the incision (Fig. 29-8, C). Resect a small ellipse of cartilage if tube insertion is difficult. Appose the sternohyoid muscles, subcutaneous tissue, and skin cranial and caudal to the tube. Secure the tube by suturing it to the skin or tying it to gauze that is tied around the neck. Expose the proximal cervical trachea with a ventral cervical midline incision. Create a tunnel dorsal to the trachea in the area of the third to sixth tracheal cartilages. Take care to prevent traumatizing the recurrent laryngeal nerves, carotid artery, vagosympathetic trunk, jugular vein, thyroid vessels, or esophagus. Using this tunnel, appose the sternohyoid muscles dorsal to the trachea with horizontal mattress sutures to create a muscle sling to reduce tension on the mucosa-to-skin sutures (Fig. 29-9, A). Beginning with the second or third tracheal cartilages, outline a rectangular segment of tracheal wall three to four cartilage widths long and one-third the circumference of the trachea in width. Incise the cartilage and annular ligaments to the depth of the tracheal mucosa (see Fig. 29-9, A). Elevate a cartilage edge with thumb forceps, and dissect the cartilage segment from the mucosa. Place one or two prosthetic tracheal rings cranial and caudal to the stoma if the tracheal cartilages show any weakness or tendency to collapse (see p. 941). Excise a similar segment of skin adjacent to the stoma (excise larger segments of skin if the animal has loose skin folds or abundant subcutaneous fat). Suture the skin directly to the peritracheal fascia laterally and the annular ligaments proximal and distal to the stoma with a series of interrupted intradermal sutures (3-0 or 4-0 polydioxanone or polypropylene). Make an I– or H-shaped incision in the mucosa. Fold the mucosa over the cartilage edges and suture it to the edges of the skin with approximating sutures to complete the tracheostoma (Fig. 29-9, B). Use simple interrupted sutures at the corners and a simple continuous pattern to further appose skin and mucosa (using 4-0 polypropylene suture) (see Fig. 29-9, B). Alternatively, make a full-thickness H-shaped incision in the ventral trachea to create two tracheal flaps that are reflected cranially and caudally and sutured directly to the skin. Expose the involved trachea through a ventral cervical midline incision, lateral thoracotomy (see p. 965), or median sternotomy (see p. 967). Mobilize only enough trachea to allow anastomosis without tension. Preserve as much of the segmental blood and nerve supply to the trachea as possible. Place stay sutures around cartilages cranial and caudal to the resection sites before transecting the trachea. Resect the diseased trachea by splitting a healthy cartilage circumferentially at each end, or by incising annular ligaments adjacent to the intact cartilages (Fig. 29-10, A). Use a No. 11 blade to split the tracheal cartilages at their midpoint. Transect the dorsal tracheal membrane with Metzenbaum scissors. Preplace and then tie three or four simple interrupted sutures (3-0 or 4-0 polypropylene [Prolene], polybutester [Novafil], polydioxanone [PDS], or polyglyconate [Maxon]) in the dorsal tracheal membrane (Fig. 29-10, B). Retract the endotracheal tube into the proximal trachea during resection and placement of sutures in the dorsal tracheal membrane. Remove blood clots and secretions from the lumen, and advance the tube distal to the anastomosis after placing dorsal tracheal membrane sutures. Complete the anastomosis by apposing the split cartilage halves or adjacent intact cartilages with simple interrupted sutures beginning at the ventral midpoint of the trachea. Space additional sutures 2 to 3 mm apart. Place three or four retention sutures to help relieve tension on the anastomosis. Place and tie these sutures so that they encircle an intact cartilage cranial and caudal to the anastomosis, crossing external to the anastomotic site (Fig. 29-10, C). Lavage the area and appose the sternohyoid muscles in a simple continuous pattern. Close the subcutaneous tissues and skin routinely. Alternatively, consider a simple continuous pattern for anastomosis because it has similar biomechanical strength to a simple interrupted pattern augmented with encircling sutures in the neighboring cartilage rings (Demetriou et al, 2006). Position the patient in ventral recumbency with the neck extended. Suspend the maxilla and pull the mandible ventrally to maximally open the mouth. Extend the tongue from the mouth to get maximum exposure of the glottis. Retract the cheeks laterally to improve visualization. Avoid placing padding or hands in the region of the larynx because this may distort the nasopharynx. Remove the central margin of the vocal cord for debarking with a laryngeal or uterine cup biopsy forceps (Fig. 29-11, A). To widen the glottis, use long-handled Metzenbaum scissors and remove as much of the vocal fold extending into the laryngeal lumen as possible (Fig. 29-11, B). With either technique, maintain 1 to 2 mm of mucosa at the dorsal and ventral aspects of the vocal cord. Control hemorrhage with pressure. Remove blood clots and secretions with suction or sponges. Allow the incision to heal by secondary intention. Position the patient in dorsal recumbency with the neck extended over a rolled towel (Fig. 29-12). Expose the larynx using a ventral midline cervical approach, beginning rostral to the basihyoid bone and extending caudally to the proximal trachea. Separate and retract the paired sternohyoid muscles. Identify the midline of the thyroid cartilage. Ligate and divide the laryngeal impar vein if necessary. Incise the cricothyroid ligament with a No. 15 or No. 11 blade. Extend the incision along the midline of the thyroid cartilage as needed to expose the vocal folds. Excise the entire vocal fold from the arytenoid cartilage dorsally and the thyroid cartilage ventrally (see Fig. 29-12). Close the defect by apposing the mucosa in a simple continuous appositional pattern using 4-0 to 5-0 monofilament absorbable suture (see Fig. 29-12). Appose the cricothyroid ligament and thyroid cartilage with simple interrupted sutures. Appose the sternohyoid muscles in a simple continuous pattern with 3-0 or 4-0 monofilament absorbable suture. Close the subcutaneous tissues and skin routinely. An assortment of long-handled instruments is beneficial. Skin hooks, laryngeal or uterine cup biopsy forceps, and tracheal prostheses (see p. 941) are needed for some procedures. Rhinotomy requires an oscillating saw, chisel and mallet, or rongeurs, as well as periosteal elevators and curettes. Nonreactive monofilament suture (e.g., polypropylene [PDS], polybutester [Novafil], polydioxanone [PDS], polyglyconate [Maxon]) is recommended for surgery of the upper respiratory tract. Patients must be closely monitored during anesthetic recovery for hemorrhage, coughing, gagging, or aspiration. They should be kept intubated as long as possible and reintubated or have a tracheostomy tube placed if respiratory distress occurs after extubation. Supplemental oxygen should be provided if necessary during recovery, and excitement and pain should be minimized by postoperative analgesics (see Table 12-3 on p. 141 and Box 12-2 on p. 138). Inserting a nasal oxygen catheter at the conclusion of surgery facilitates oxygen delivery (see Box 4-4 on p. 33). Alternatively, the animal may be placed in an oxygen cage. Positioning the patient in sternal recumbency may facilitate respiration. Postoperative corticosteroids (dexamethasone [Azium], 0.5 to 2 mg/kg IV, IM, or SC; start with the lower dose and use the higher dose only if the low dose has failed to reduce swelling, and if swelling is sufficiently severe that the animal cannot breathe). Single large doses or repeated administration of dexamethasone typically causes gastrointestinal erosions, ulcers, and/or hemorrhage and may reduce mucosal swelling and edema. Prophylactic antibiotics can be discontinued immediately after surgery. Water may be offered 6 to 12 hours after surgery; soft food made into meatballs may be offered 12 to 24 hours postoperatively if gagging, regurgitation, or vomiting does not occur. Meatballs should be fed one at a time for 5 to 7 days after nasopharyngeal or laryngeal procedures to slow ingestion. Exercise should be restricted for 4 weeks. A harness rather than a collar should be used for 2 to 4 weeks to prevent incisional, tracheal, or laryngeal trauma. After rhinotomy, animals should be recovered in a slightly head-down position, they should be kept quiet, and some sneezing should be expected. Breathing sounds are typically harsh and resonant after this procedure. Initially, nasal discharge is expected to be bloody, but it should gradually become serous and diminish in volume after the nasal cavity has re-epithelialized (generally within a week). If bleeding continues after surgery, the nasal cavity can be packed with sterile gauze. If gauze strips are used (see p. 953), the end of the gauze can be exited from the nostril or a dorsal stoma and sutured to the side of the face. The packing is removed 1 or 2 days after surgery. A blood transfusion may be needed if hemorrhage is significant (see Box 4-1 on p. 30). Movement of the skin flap covering the bony defect is expected following dorsal rhinotomy, and subcutaneous emphysema may occur but should be self-limiting. Subcutaneous air accumulates if the bone flap is not replaced adequately, or if an adequate stoma is not left in the subcutaneous tissues and skin for air to exit. Subcutaneous air accumulation generally is not a problem if an adequate stoma is left. The stoma will contract and heal within 5 to 10 days. Do not allow the animal to chew on hard objects for at least 3 to 4 weeks following ventral rhinotomy until the palate incision is healed. Feed soft food for a minimum of 7 to 14 days following surgery. Animals occasionally may be depressed and unwilling to eat for several days; in such cases, consider placement of an enteral feeding tube (e.g., esophagostomy; see p. 102). Acute respiratory obstruction caused by mucosal swelling, edema, irritation, and increased mucus production and/or laryngeal or tracheal collapse may occur after upper respiratory surgery and must be relieved promptly (Box 29-5). Infection can be a problem because the nose, nasopharynx, larynx, and trachea have a resident bacterial flora. Using strict aseptic technique and lavaging contaminated tissues usually prevent infection. Injury to the recurrent laryngeal nerve may cause laryngeal spasms, paresis, or paralysis, leading to aspiration pneumonia. Mucostasis may occur after nerve damage. Gentle tissue handling, appropriate dissection, and careful tissue retraction prevent nerve damage. Intensive monitoring of a patient with a temporary tracheostomy tube is required to avoid life-threatening complications, particularly in smaller animals. Complications associated with tube tracheostomy include gagging, vomiting, coughing, tube obstruction, tube dislodgment, emphysema, tracheal stenosis, tracheal malacia, and tracheocutaneous or tracheoesophageal fistula. Some animals occlude the tracheostomy tube when the neck is flexed, and when they sleep with bedding. Major complications (occlusion, dislodgment) were reported in 44% of cats with temporary tracheostomies (Guenther-Yenke and Rosanski, 2007). Cuffed tracheostomy tubes and endotracheal tubes may cause pressure necrosis of the tracheal mucosa or cartilages, which may result in tracheal strictures. Animals with permanent tracheostomy have high complication and mortality rates primarily caused by occlusion of the stoma from mucus, blood, or stricture. In particular, cats are at high risk for acute occlusion and sudden death (Stepnik et al, 2009). The long-term main complication of permanent tracheostomy is stomal occlusion from accumulated mucus, skin folds, or stenosis. Mucus accumulation, coughing, and gagging may also occur because of tracheal irritation. Demetriou, JL, Hughes, R, Sissener, TR. Pullout strength for three suture patterns used for canine tracheal anastomosis. Vet Surg. 2006;35:278. Guenther-Yenke, CL, Rosanski, EA. Tracheostomy in cats: 23 cases (1998-2006). J Fel Med Surg. 2007;9:451. Stepnik, MW, Mehl, ML, Hardie, EM, et al. Outcome of permanent tracheostomy for treatment of upper airway obstruction in cats: 21 cases (1990-2007). J Am Vet Med Assoc. 2009;234:638. Tobias, KM, Jackson, AM, Harvey, RC. Effects of doxapram HCl on laryngeal function of normal dogs and dogs with naturally occurring laryngeal paralysis. Vet Anaesth Analg. 2004;31:258. Specific Diseases Brachycephalic syndrome refers to upper airway obstruction attributable to a combination of anatomic abnormalities seen in brachycephalic dogs (Boxes 29-6 and 29-7). Concurrent tracheal hypoplasia or advanced laryngeal collapse and other abnormalities often contribute to respiratory distress. Brachycephalic animals typically have a compressed face with poorly developed nares and a distorted nasopharynx. The head shape is the result of an inherited developmental defect in the bones of the base of the skull. These bones grow to a normal width but have a reduced length. The soft tissues of the head are not proportionately reduced and often appear redundant. Stenotic nares are congenital malformations of the nasal cartilages that result in medial collapse and partial occlusion of the external nares (Figs. 29-13 and 29-14). Airflow into the nasal cavity is restricted and greater inspiratory effort is necessary, causing mild to severe dyspnea. Resistance to airflow through the nasal cavity in normal dogs is 76% to 80% of total resistance, depending on the volume of airflow. As more and more negative pressure is exerted to breathe, intratracheal and intrapharyngeal pressures can become high enough to cause surrounding tissues to collapse. Elongated soft palate is the most common component of brachycephalic syndrome and is often diagnosed in brachycephalic dogs (Fig. 29-15). The elongated soft palate is pulled caudally during inspiration, obstructing the dorsal aspect of the glottis. It is sometimes drawn between the corniculate processes of the arytenoid cartilages; this increases inspiratory effort and causes more turbulent airflow. The laryngeal mucosa becomes inflamed and edematous, further narrowing the airway. The tip of the soft palate is blown into the nasopharynx during expiration. Affected dogs may have trouble swallowing because normal occlusion of the airway during deglutition compromises ventilation. Dysfunctional swallowing may produce aspiration pneumonia. Some affected animals with concurrent gastrointestinal problems (e.g., esophagitis, hiatal hernia) have improved following surgical treatment of the brachycephalic syndrome. Laryngeal saccule eversion (Fig. 29-16) is diagnosed less often than elongated soft palate or stenotic nares but has been in reported in 58% to 66% of dogs with brachycephalic syndrome (Riecks et al, 2007; Fasanella et al, 2010). It is uncommon to have everted laryngeal saccules as the only abnormality. Eversion of the laryngeal saccules is considered the first stage of laryngeal collapse. Increased airflow resistance and increased negative pressure generated to move air past obstructed areas (stenotic nares, dorsal glottis) pull the saccules from their crypts, causing them to swell. Once everted, the saccules are continuously irritated by turbulent airflow and become increasingly edematous. The saccules obstruct the ventral aspect of the glottis and further inhibit airflow. It may be difficult for an inexperienced examiner to differentiate everted laryngeal saccules from the vocal folds because of their close proximity. Everted saccules partially or completely obscure the vocal folds and may be unilateral or bilateral. In addition to components already described, aryepiglottic collapse, corniculate collapse, tonsil eversion, redundant pharyngeal folds, pharyngeal collapse, macroglossia, and nasopharyngeal turbinates (Fig. 29-17) may contribute to the severity of respiratory distress. The presence of nasopharyngeal turbinates has been identified in 21% of dogs with brachycephalic syndrome (Ginn et al, 2008). Hypoplastic trachea is congenital tracheal stenosis that affects the entire length of the trachea. In addition, rigid tracheal cartilages are apposed or overlap, and the dorsal tracheal membrane is narrow or obscured. Tracheal hypoplasia can be associated with continuous respiratory distress, coughing, and recurrent tracheitis but may be tolerated in the absence of concurrent respiratory or cardiovascular disease. Tracheal hypoplasia can be diagnosed radiographically by determining the tracheal-to-thoracic inlet diameter ratios (Box 29-8). No definitive treatment for hypoplastic trachea is known, only symptomatic therapy and correction of other anatomic defects to alleviate clinical signs. The extent to which hypoplastic trachea contributes to the severity of clinical signs in dogs with brachycephalic airway syndrome is unknown. General anesthesia is necessary to evaluate the pharynx and larynx (see p. 907). This is initially provided with a rigid laryngoscope. An elongated soft palate overlies the epiglottis by at least a few millimeters but often by more than 1 cm (see Fig. 29-15). The soft palate often is thickened and has a roughened, inflamed tip. Dorsally displacing the soft palate with a tongue depressor improves visualization of the arytenoid cartilages. The arytenoids frequently are inflamed and edematous. Tonsils may be inflamed and everted from their crypts. Everted laryngeal saccules appear as spherical protrusions of tissue that obscure the vocal folds (see Fig. 29-16). Functional assessment of the larynx rules out concurrent laryngeal paralysis (p. 932), and laryngeal anatomy should be evaluated for other signs of collapse (p. 930). A flexible endoscope allows complete evaluation of the nasopharynx (to assess for nasopharyngeal turbinates or masses), trachea (to assess for hypoplastic trachea or tracheal collapse [p. 938]), and bronchial tree. A large percentage of bronchial abnormalities were recently documented in a consecutive series of brachycephalic dogs; however, the presence of bronchial collapse or stenosis does not appear to affect long-term outcome (De Lorenzi et al, 2009). Similarly, a high prevalence of gastrointestinal tract lesions was identified in a series of dogs with brachycephalic syndrome. Endoscopy of the upper gastrointestinal tract should be considered in brachycephalic dogs with signs of gagging, retching, regurgitation, or vomiting. A weight reduction program should be instituted for obese animals. Exercise restriction and elimination of precipitating causes may be beneficial when clinical signs are mild. Sedation (see Box 29-2), corticosteroids, supplemental oxygen (see Table 4-4 on p. 32), and cooling may be necessary for moderate to severe respiratory distress. An anti-inflammatory dose of a corticosteroid (dexamethasone [Azium], 0.5 to 2 mg/kg IV, IM, or SC; start with the lower dose and use the higher dose only if the low dose has failed to reduce swelling, and if swelling is sufficiently severe that the animal cannot breathe) may reduce nasopharyngeal and upper airway edema. Large doses or repeated administration of dexamethasone typically causes gastrointestinal irritation, erosion, or ulceration.
Surgery of the Upper Respiratory System
Preoperative Management
Anesthesia
![]() TABLE 29-1
TABLE 29-1


![]() TABLE 29-2
TABLE 29-2



Antibiotics
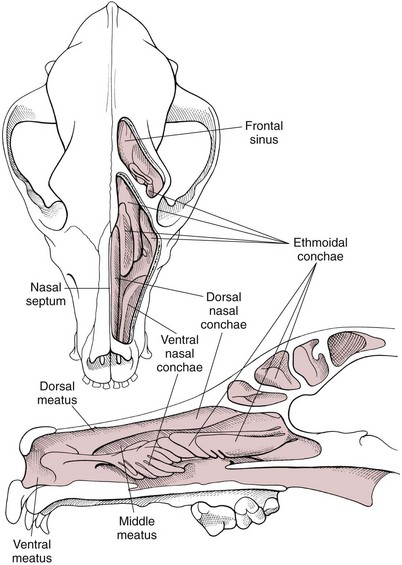
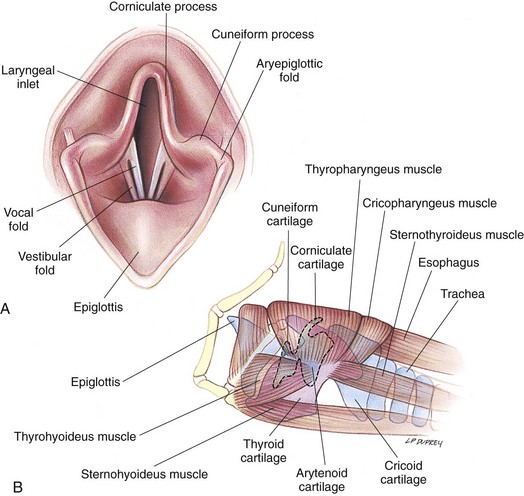
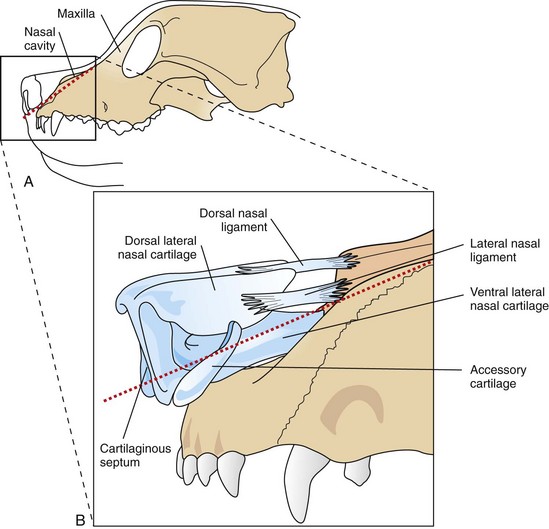
Surgical Anatomy
Surgical Technique
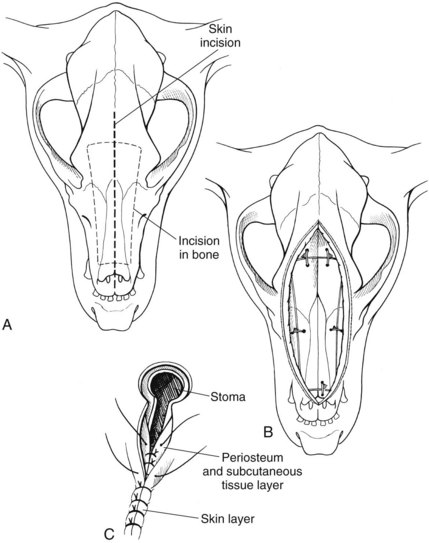
Rhinotomy
Dorsal approach to the nasal cavity and paranasal sinuses
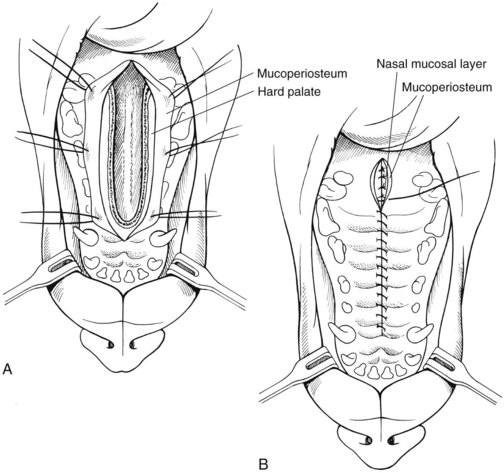
Ventral approach to the nasal cavity
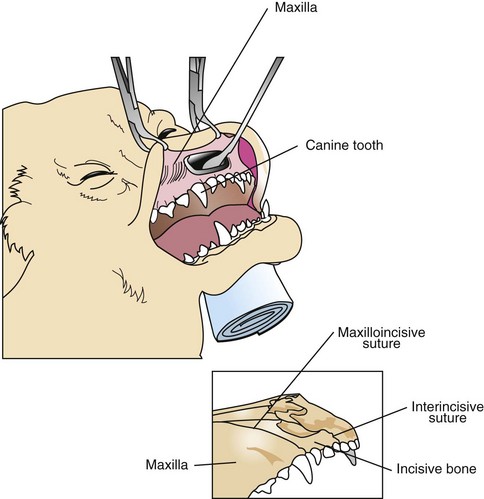
Lateral approach to the rostral nasal cavity
Intraoral approach to the rostral nasal cavity
Tracheotomy
Tracheostomy
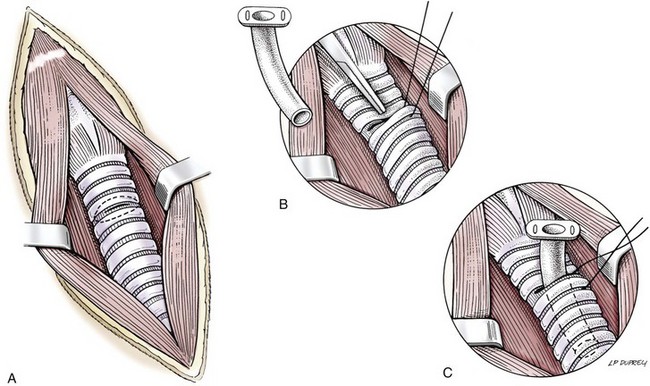
Temporary tracheostomy
Permanent tracheostomy
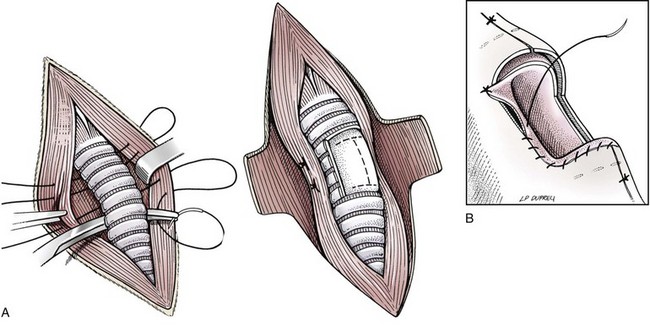
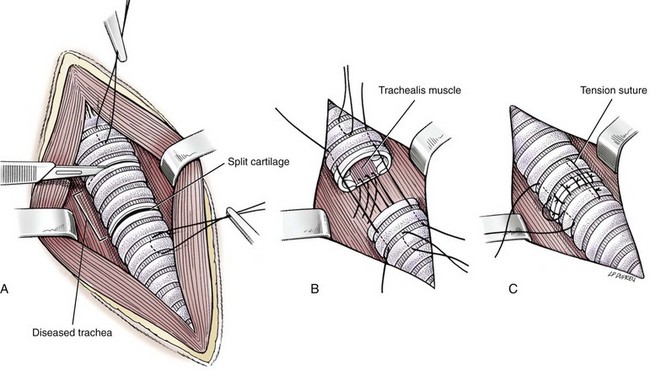
Tracheal Resection and Anastomosis
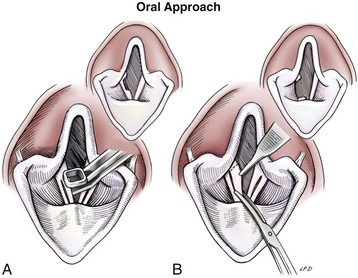
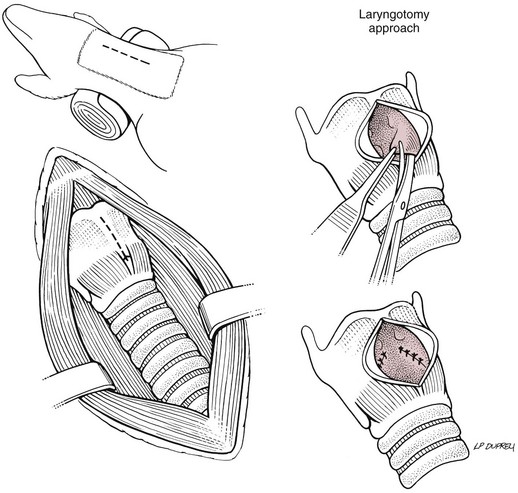
Ventriculocordectomy
Oral approach
Laryngotomy approach
Suture Materials and Special Instruments
Postoperative Care and Assessment
Complications
References
Brachycephalic Syndrome
General Considerations and Clinically Relevant Pathophysiology
Diagnosis
Diagnostic Imaging
Pharyngoscopy/laryngoscopy
Flexible endoscopy
Medical Management
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Surgery of the Upper Respiratory System