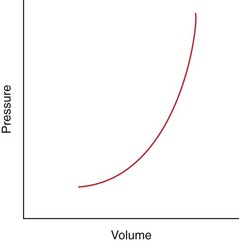
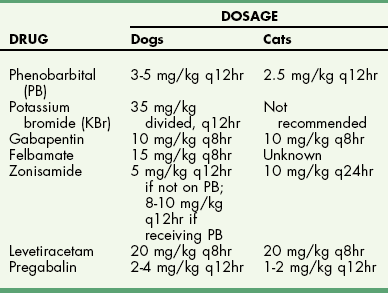
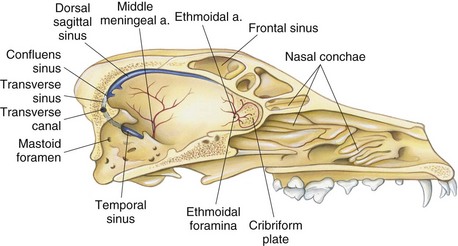
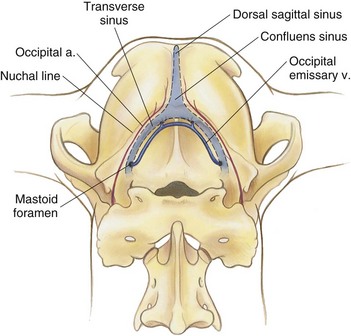
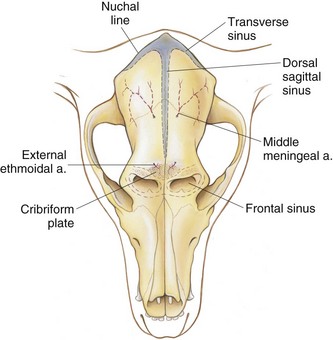
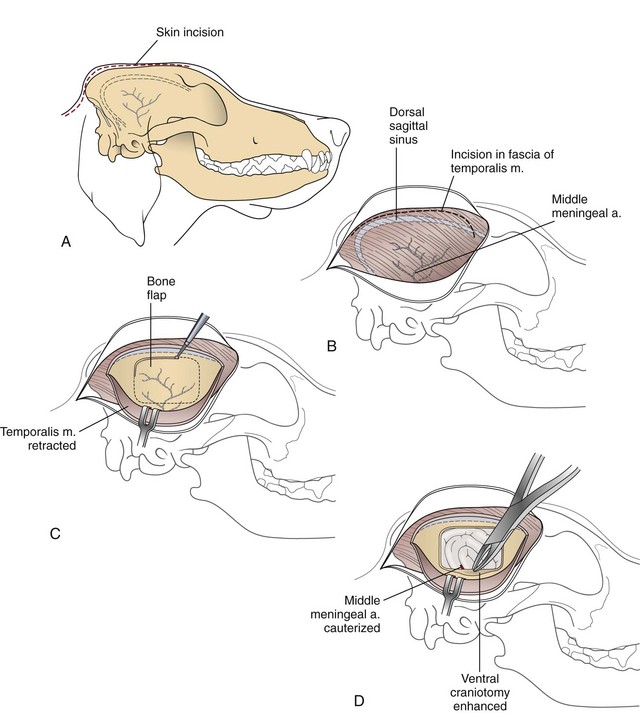
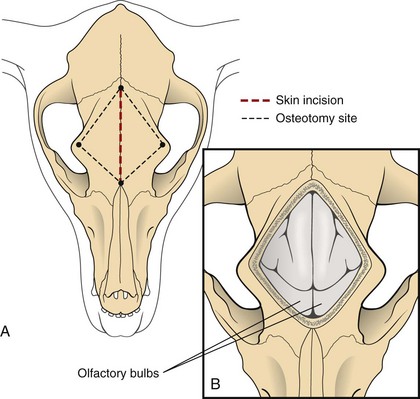
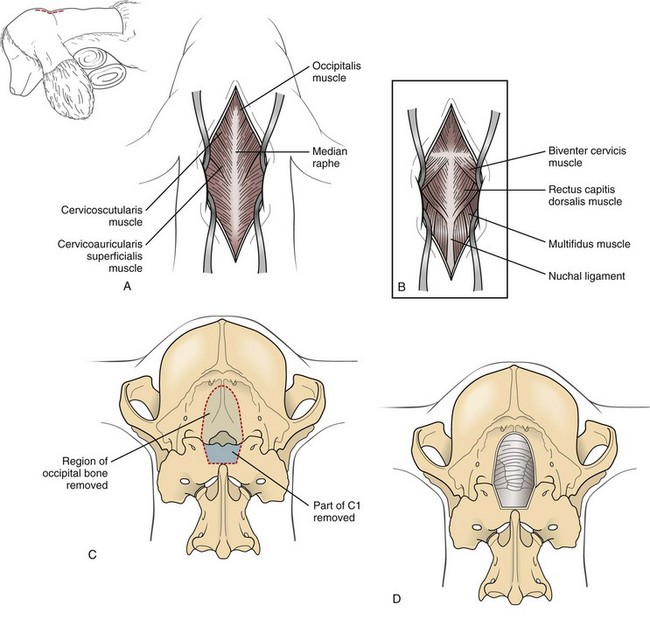
Chapter 39 The three main types of craniotomy are sometimes combined to provide increased exposure to parts of the brain for various conditions; they may be modified when necessary. For example, bilateral rostrotentorial craniotomies may be required at times; this may also involve removing the top of the skull. Increased ventral exposure may be needed in some cases during a rostrotentorial craniotomy; this may require resection of part of the zygomatic arch and/or the coronoid process of the mandible. When working in the caudal fossa region, it may be necessary to extend a suboccipital craniotomy laterally by sacrificing one of the transverse sinuses. Sometimes more room is needed caudally in this approach, which is achieved by partial or complete removal of the dorsal arch of the atlas (C1). Ventral approaches to the brain are seldom performed in dogs and cats and therefore will not be discussed in this chapter. The relationship between ICP and CPP and MAP is as follows: CPP = MAP − ICP. Compliance decreases as intracranial volume increases, as is shown in Figure 39-1. Once an intracranial volume is reached that is close to the limit of the compliance ability of the intracranial compartment, even small increases in intracranial volume (e.g., brain edema, brain vasodilation from anesthesia) will lead to large increases in ICP. The specifics of preoperative management for the brain surgery patient depend to a large degree on the underlying disease process. With increased availability of magnetic resonance imaging (MRI) in dogs and cats, the repertoire of surgical brain disorders in these species has expanded (see also Chapter 15). Patients with disorders typified by slow-growing mass lesions (e.g., brain tumors, intracranial arachnoid cysts) will likely benefit from low doses of oral prednisone (e.g., 0.5 mg/kg every 12 hours) in the time period before surgery is scheduled and for some time afterward. This dose of prednisone alleviates edema surrounding the lesion and typically improves the patient’s neurologic status dramatically within 24 to 48 hours. Glucocorticoids should not be given as an emergency treatment for acute brain injury (particularly the “high-dose methylprednisolone” protocol) because they have repeatedly failed to demonstrate any statistically significant benefit in terms of central nervous system trauma, and they have the potential for serious adverse effects. Adequate fluid support must be provided before anesthesia is given. Concern has arisen that overzealous intravenous fluid administration may exacerbate brain edema, especially in trauma patients; however, fluid restriction is strictly contraindicated. The response of the vasculature to hypotension is vasodilation. In a closed vault such as the cranium, the result is an increase in space taken up by the vasculature. The patient with brain pathology may have additional elevations in ICP caused by this vasodilatory response to hypotension. Conversely, marked hypertension causes an increase in cerebral blood volume and promotes cerebral edema. Therefore, the goal is to keep blood pressure within the normal range, avoiding hypotension and marked hypertension. Patients that present with an increase in ICP often have systemic hypertension—an indication that efforts to reduce ICP are necessary. Normal saline is the fluid of choice for brain surgery. Because of the blood-brain barrier, osmotic pressure, not oncotic pressure, is the driving force for movement of water out of the vasculature and into the interstitial and intracellular fluid compartments of the brain. In other words, sodium is of vital importance. To minimize brain edema, serum tonicity should be maintained. This is best done by using normal saline and rechecking serum sodium levels when diuretics such as mannitol are used. In acute brain injury patients, hypertonic saline and/or colloid solutions (e.g., hetastarch) are usually advised in combination with normal saline to support normal blood pressure while minimizing the likelihood of further brain edema (see Table 4-5 on p. 34). Depending on the specific disease process, it may be necessary to provide supplemental oxygen to the brain surgery patient before and after surgery. This is often required for victims of severe brain injury. Patients who are conscious and are not obviously deteriorating neurologically should be administered supplemental oxygen via face mask, nasal oxygen catheter, or transtracheal oxygen catheter (see p. 31). Face masks tend to stress dogs and cats and should be used only temporarily, until another form of oxygen (O2) delivery can be instituted (e.g., nasal O2). Use of an O2 cage is generally an ineffective method of administering supplemental O2 to severely brain-injured patients because most of these patients require frequent or constant monitoring. Oxygen cages do not allow for concomitant close patient observation (requires opening the cage door) and maintenance of a high-oxygen environment. With nasal and transtracheal O2 catheters, an inspired oxygen concentration of 40% is provided with flow rates of 100 ml/kg/min and 50 ml/kg/min, respectively. In cases of traumatic brain injury, care must be taken to avoid placing nasal O2 catheters farther than the level of the medial canthus (to avoid entering the cranial vault through a fracture site). Inadvertent jugular vein compression, which can cause increased ICP, should be avoided while transtracheal O2 catheters are placed. High flow rates with nasal O2 catheters may induce sneezing, which has the potential to raise ICP. Patients who are losing or have lost consciousness should be intubated and ventilated. In the patient with oscillating levels of consciousness or airway obstruction secondary to trauma, a tracheostomy tube may be indicated for assisted ventilation. Antiseizure drugs should be administered to dogs and cats with brain disorders characterized by recurrent seizure activity. A number of alternatives to phenobarbital and potassium bromide are available for managing seizures in dogs and cats (Table 39-1). Most of these drugs are generic and inexpensive. Zonisamide is particularly advantageous to use in dogs with brain tumors. Phenobarbital (PB) has the tendency to cause profound sedation in dogs with brain tumors, even at low doses; this effect is sufficiently severe that PB is not recommended in these dogs. Zonisamide does not typically cause sedation and is both effective and inexpensive. Other potentially useful antiseizure drugs for dogs include gabapentin, levetiracetam, felbamate, and pregabalin. Pregabalin, the “next generation” of gabapentin, has a longer half-life of elimination than its predecessor and is considered to be more effective. Both pregabalin and gabapentin can cause sedation, although this effect is typically mild. Levetiracetam (LEV) is an extremely useful drug in dogs and cats with brain tumors, particularly when the intravenous form of the drug is used for short-term management. Levetiracetam typically has no side effects, even at high doses. A single bolus injection (20 mg/kg over several minutes) of LEV results in therapeutic plasma levels of the drug for 8 to 12 hours. Levetiracetam has no known drug-drug interactions and does not undergo hepatic metabolism. Oral levetiracetam is a useful oral drug in both dogs and cats, although a “honeymoon effect” is often seen in dogs with this drug, in which it becomes apparently less effective over time. Felbamate is an effective and safe antiseizure drug for dogs that does not cause sedation; however, it is expensive and therefore is used infrequently. For cats, PB, levetiracetam, zonisamide, gabapentin, and pregabalin are useful antiseizure drugs. Phenobarbital is very effective in cats and usually does not cause side effects commonly encountered in dogs. Oral diazepam and bromide are contraindicated in cats. The induction period needs to be highly controlled in these patients. Barbiturates and propofol are the induction agents of choice (Table 39-2). A smooth induction and intubation should be performed, with every effort made to avoid blood pressure extremes and prolonged apnea. Hypertension and tachycardia with intubation may have to be treated by administering esmolol or by deepening the anesthetic with additional boluses of propofol or thiopental. Preferred and Nonpreferred Anesthetic Drugs for Dogs and Cats Undergoing Brain Surgery *Monitor for hyperthermia in cats. Hypotension should be quickly treated with vasopressors such as ephedrine and phenylephrine, but not with fluid boluses (see Table 39-2). The airway needs to be secured very carefully because access to the head will be restricted during surgery. Positioning of the animal with its head elevated is beneficial for patients with increased ICP. Anesthetic agents influence cerebral blood flow (CBF), cerebral metabolism, CO2 reactivity, and cerebral blood pressure autoregulation. Barbiturates, propofol, isoflurane, and sevoflurane all have been shown to decrease cerebral metabolism. Although these anesthetic agents decrease cerebral blood flow, it is the concomitant decrease in cerebral metabolic rate that provides neuroprotection. Barbiturates have been extensively studied in neuroanesthesia and have been used primarily because of their hypnotic effect, decrease in cerebral metabolic rate, and anticonvulsant activity. Even though they cause dose-dependent decreases in cerebral blood flow, barbiturates cause a corresponding decrease in cerebral metabolic rate and cerebral oxygen requirements uniformly throughout the brain. Combined with their apparent ability to increase CSF absorption, barbiturates generally cause a decrease in ICP. Propofol acts very similarly, with decreases to CBF, cerebral metabolic rate, and ICP. It also has significant anticonvulsant properties. However, systemic vasodilation and subsequent hypotension with propofol administration should be anticipated and treated. Although etomidate decreases ICP, it is avoided in neurosurgery patients because it causes increased seizure activity and thus an increased cerebral metabolic rate. Inhalant anesthetics cause a dose-dependent decrease in cerebral metabolic rate, as well as an increase in cerebral perfusion. This is especially desirable during periods of hypotension. Isoflurane is the inhalant anesthetic agent of choice because of its ability to increase CSF absorption and to minimally increase ICP (see Table 39-2). Halothane and desflurane should be avoided because of their greater increases in ICP. In general, anesthetic agents considered preferable for use in brain surgery patients include benzodiazepines (e.g., diazepam, midazolam), barbiturates (e.g., pentobarbital), propofol, opioids, isoflurane, and sevoflurane (see Table 39-2). Opioids have little to no effect on ICP, and pentobarbital, propofol, and benzodiazepines decrease ICP with some neuroprotective properties. The potential adverse effects of opioids on ICP are minimal to nonexistent if appropriate doses are used. The concern is for depression and inadequate respirations causing elevations in PaCO2. Therefore it is recommended that opioids not be used in the preoperative period, but rather that they be used during surgery, when ventilation is controlled, and during the postoperative period. Poorly controlled pain is much more likely to increase ICP than is judicious use of opioids postoperatively. Additionally, continuous rate infusions (CRIs) of opiates (e.g., fentanyl, morphine) begun during surgery and continued into the postoperative period are less likely to lead to respiratory depression with a subsequent ICP increase due to hypercapnia (see p. 138). The α2-agonists dexmedetomidine, medetomidine, and xylazine have some neuroprotective qualities. Both dexmedetomidine and medetomidine cause a decrease in ICP in patients already anesthetized with an inhalant anesthetic. Unfortunately, their variable effects on blood pressure make them less appropriate as induction drugs. However, if used in low doses in a balanced anesthesia protocol, they may reduce the hypertension followed by hypotension seen in these patients (see Table 39-2). Important vascular structures to avoid during brain surgery are the dorsal sagittal sinus and the transverse sinuses (Fig. 39-2). Laceration of these structures can cause life-threatening hemorrhage. Ligation or obstruction of the dorsal sagittal sinus (particularly the caudal two-thirds of the sinus), the confluens sinuum (the midline juncture of the paired transverse sinuses), or both transverse sinuses will likely lead to fatal brain swelling. During the suboccipital approach, it is common to experience hemorrhage from occipital emissary veins at the ventrolateral limits of the supraoccipital bone during periosteal muscle elevation; this hemorrhage is controlled by pushing bone wax into the mastoid foramen, through which this vessel traverses. For all standard approaches, the author prefers a midline incision. Unless brain exposure is very large (e.g., removal of the top portion of the calvarium) or the craniotomy procedure is likely to lead to an obviously abnormal cosmetic appearance postoperatively (e.g., removal of the zygomatic process of the frontal bone), the craniotomy defect is not repaired. If repair of the skull defect is considered necessary, titanium mesh and polymethylmethacrylate (PMMA) can be used (Fig. 39-3). The PMMA is placed on either side of the mesh; the mesh keeps the PMMA from sagging into the craniotomy site during hardening. Titanium rather than steel is used so that repeat MRI is possible if necessary. Postoperative CSF leakage is a clinical problem encountered in human brain surgery; however, this phenomenon does not occur in dogs and cats following craniotomy. Replacing resected dura/arachnoid with autologous or synthetic dural substitute generally is not indicated in dogs and cats. A layer of Gelfoam can be placed over the exposed brain before the time of closure. Shave the skin and aseptically prepare the area from the level of the medial canthi rostrally to the cranial aspect of the second cervical vertebra caudally; the lateral limits of the prepared region are the zygomatic arches. Shave and aseptically prepare any aspect of the ears that may be in the surgical area. Make a midline incision from approximately the level of the lateral canthi to several centimeters past the external occipital protuberance. Sharply incise the cutaneous coli muscles and transect the cervicoscutilaris muscle near midline, leaving about a centimeter for reattachment during closing. Sharply incise the temporalis fascia with a No. 11 blade, leaving at least several millimeters attached to the skull for reattachment purposes. Reflect the temporalis musculature ventrally by elevating it off the side of the skull with Freer periosteal elevators (cats and small dogs) or Army-Navy osteotomes (large dogs). Create an oval outline of the intended defect with a high-speed air drill. Deepen the edges of this outline until a thin layer of periosteum is left. The ventral aspect of the defect does not have to be drilled full thickness. Once the dorsal and lateral aspects of the craniotomy defect outline are completely drilled, use a periosteal elevator to lever the bone flap off the skull after positioning the elevator in the dorsal aspect of the defect (Fig. 39-4). Once the bone flap is removed, further enlarge the craniotomy defect using rongeurs. After removal of the bone flap, use bipolar cautery to stop hemorrhage from bleeding meningeal vessels. This bone can be removed in a variety of sizes and shapes with the use of a high-speed air drill or an oscillating saw blade. A triangular shape is often chosen for a unilateral approach, a diamond pattern for a bilateral approach (Fig. 39-5). Because of the limited exposure (most olfactory meningiomas occupy the majority of the olfactory bulb region bilaterally) provided by the unilateral approach, the bilateral approach is typically performed. If the bone plate is to be replaced, use the saw blade or a small drill bit when removing the bone. The most common indication for a suboccipital craniotomy in dogs is to relieve compression at the craniocervical junction in cases of Chiari-like malformation (CLM); in this scenario, the craniotomy is combined with a dorsal laminectomy of the atlas (C1) and is referred to as a foramen magnum decompression (FMD). For other purposes (e.g., brain tumor removal), the suboccipital craniotomy is typically performed without including the C1 dorsal laminectomy. For either version of this procedure, place the dog in sternal recumbency with the neck ventroflexed. Shave and aseptically prepare the dorsal aspect of the head and neck from the level of the bregma to the level of the third or fourth cervical vertebra, with a width approximately equal to the width of the atlas. Make a dorsal midline incision extending from approximately 1 cm rostral to the external occipital protuberance cranially to the middle of the second cervical vertebra caudally. Separate the superficial dorsal cervical musculature (Fig. 39-6, A) at the median raphe, exposing the underlying biventer cervicis muscles. Separate the paired biventer cervicis muscles on midline, exposing the rectus capitis dorsalis muscles (Fig. 39-6, B). Remove the caudal aspects of the rectus capitis dorsalis muscles from the cranial half of C2 using sharp dissection and periosteal elevation, and split these muscles on the midline. Sharply incise the cranial aspects of the rectus capitis dorsalis muscles from the nuchal crest, exposing the caudal portion of the occiput and the arch of the atlas. Control hemorrhage with bipolar electrocautery. Use a high-speed air drill with a 3- to 4-mm diameter round drill bit and Lempert rongeurs to remove a portion of the occiput and the dorsal aspect of the first cervical vertebra (Fig. 39-6, C, D). Approximately 75% of the length of the dorsal arch of C1 is routinely removed. In cases in which there appears to be dorsal constriction at C1/C2 (see Chapter 40), remove the entire arch of C1 as well as part of the dorsal lamina of C2. All postoperative craniotomy patients should be recovered in an intensive care unit and monitored for 3 to 5 days postoperatively. The cage should be padded (e.g., bubble-wrap) to avoid injury to the surgical site. Because postoperative brain surgery patients may have subtle pharyngeal dysfunction, nothing is given per os for the first 24 hours. After this time, small “meatballs” of canned food are offered several times a day, in addition to water. For patients on oral prednisone therapy, this therapy is gradually tapered over 5 to 7 days. Antibiotics are continued according to the specifics of the disease process and clinician preference. Anticonvulsant drugs are administered as needed. Analgesic drugs are also administered (see Table 39-2). Especially in cats, serial PCV measurements should be made for the initial 24 hours post surgery and blood transfusion performed if necessary. Oxygenation and ventilation status should be assessed via blood gas analysis, depending on the specific case. Supplemental oxygen is provided, if necessary. For patients that are unable or unwilling to voluntarily ambulate, frequent turning (e.g., every 4 hours) is essential to avoid pulmonary atelectasis and subsequent pneumonia. The patient’s neurologic status should be assessed several times per day, and any deterioration should be addressed medically (e.g., mannitol) and/or surgically (reimaging and reoperation). Suspected pneumonia (typically older, large dogs following brain tumor removal) should be treated aggressively (e.g., nebulization/coupage, broad-spectrum IV antibiotics) and in a timely manner. Following hospital discharge, recheck examinations focus on the rate of neurologic recovery and on wound healing.
Surgery of the Brain
General Principles and Techniques
General Considerations
Preoperative Management
Anesthetic Considerations
![]() TABLE 39-2
TABLE 39-2



Standard Surgical Approaches to the Brain
Rostrotentorial (Lateral) Craniotomy
Transfrontal Craniotomy
Suboccipital Craniotomy (Foramen Magnum Decompression [FMD])
Postoperative Care and Assessment