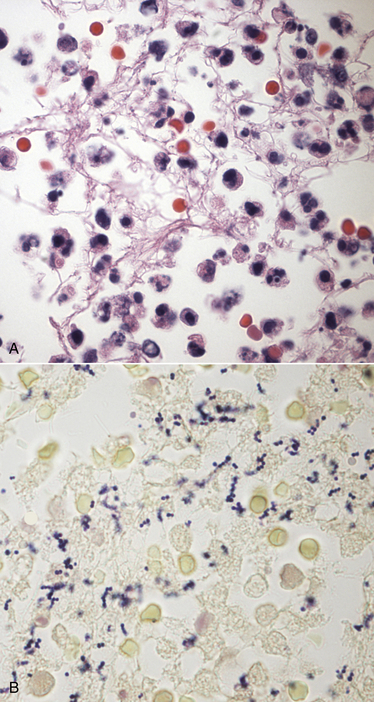
Chapter 35 Staphylococcus spp. are gram positive, nonmotile, non–spore forming, and usually catalase-positive cocci, which occur singly and in pairs or grapelike clusters (Figure 35-1). Most species are facultative anaerobes. Staphylococci colonize the skin and mucous membranes of an enormous variety of animal species, and disease generally follows a breakdown in normal host defenses. Staphylococci can also contaminate surfaces in the environment, where they may survive for several months. FIGURE 35-1 Histopathology of the integument from the left inguinal region and flank of an 8-year old male neutered boxer dog that had severe, regionally extensive acute neutrophilic cellulitis and panniculitis accompanied by septic shock. A methicillin susceptible Staphylococcus pseudintermedius was cultured from an aspirate of cellulitis fluid. The dog had also been treated with prednisone and azathioprine for 4 years to maintain remission for immune-mediated hemolytic anemia and thrombocytopenia. A, H&E stain. Large numbers of degenerate neutrophils are present. B, Staining with Brown and Benn stain reveals numerous of gram-positive coccoid bacteria, individually, in pairs, and in clusters.1000× oil magnification. Staphylococci are classified as coagulase negative or coagulase positive based on their ability to produce coagulase, an enzyme that cleaves fibrinogen into fibrin and results in coagulation of plasma. There are more than 40 species in the genus Staphylococcus,2 many of which are adapted to live on certain host species. The major coagulase-positive staphylococci that infect dogs and cats are Staphylococcus pseudintermedius (previously identified as Staphylococcus intermedius) and Staphylococcus aureus. Staphylococcus schleiferi has also been isolated from dogs (and rarely cats) with pyoderma and otitis externa and has been divided into S. schleiferi subsp. schleiferi, which is coagulase-negative, and S. schleiferi subsp. coagulans, which is coagulase-positive. However, these may instead represent a single species, S. schleiferi, with variable coagulase production.3 Coagulase-negative staphylococci (CoNS) tend to be of lower virulence than coagulase-positive staphylococci but occasionally cause disease in immunocompromised hosts. Examples of coagulase-negative staphylococci are Staphylococcus felis and Staphylococcus epidermidis. S. felis has been recognized as a urinary tract pathogen in cats.4 CoNS are often not identified to the species level by microbiology laboratories. In recent years, the prevalence of antimicrobial drug resistance has increased in Staphylococcus isolates obtained from canine and feline infections. A significant proportion of resistant isolates possess the mecA gene, which encodes an altered penicillin binding protein (PBP), known as PBP2a. The mecA gene is located on a large genetic element, the staphylococcal cassette chromosome. This PBP has low affinity for all β-lactam drugs (penicillins, cephalosporins, and carbapenems). As a result, staphylococci that possess this gene are also resistant to the penicillinase-resistant penicillins (oxacillin and methicillin), and so are termed methicillin-resistant Staphylococcus (MRS) species. Many MRS also possess resistance genes to other antimicrobial drug classes. There is no evidence that MRS are more virulent than methicillin-susceptible staphylococci, but infections with MRS are more difficult to treat with antimicrobial drugs. The prevalence of MRS isolated from dogs has ranged from 0.6% to as high as 67%, depending on the population sampled (healthy versus disease, hospitalized versus outpatients), geographic location, study dates, and sites of specimen collection. Of 89 staphylococcal isolates from dogs with superficial pyoderma seen in 2010 and 2011 by the University of California, Davis, Veterinary Dermatology service, 38.2% were MRS. This compared with 27.3% of 33 isolates from a primary care clinic in the same region.5 Methicillin-resistant S. pseudintermedius (MRSP) isolates are generally resistant to more antimicrobial drug classes than methicillin-resistant S. aureus (MRSA) isolates from dogs and cats. Severe multidrug resistance is also prevalent among CoNS. Before 2005, S. pseudintermedius were identified as S. intermedius. The name S. pseudintermedius was defined when isolates from a dog, cat, horse, and parrot were analyzed using molecular methods.6 Subsequently, a molecular analysis of organisms from dogs and cats that had been previously identified as S. intermedius revealed that they were actually S. pseudintermedius. S. pseudintermedius belongs to the “S. intermedius group” (SIG), which comprises the three closely related species S. intermedius, S. pseudintermedius, and S. delphini. It is difficult to differentiate among these species with phenotypic methods alone; genetic typing methods are required. Because genetic typing methods are not available for routine identification in veterinary diagnostic laboratories, isolates from dogs with phenotypic characteristics typical of S. intermedius are now generally identified as S. pseudintermedius.7 However, some laboratories still describe strains from dogs that have biochemical characteristics that do not quite match that typical for the type strain of S. pseudintermedius as “SIG organisms.” The whole genome of S. pseudintermedius has been sequenced, which is a major contribution to our understanding of the pathogenesis of infections caused by this organism.8 Like S. aureus, S. pseudintermedius possesses numerous mobile genetic elements and potential virulence factors, such as toxin genes that encode for a superantigen, leukotoxin, hemolysins, and exfoliative toxins, as well as an array of proteases. Methicillin resistance has emerged as a serious and widespread problem in S. pseudintermedius isolates from both healthy and sick dogs and cats. Molecular analysis has shown that among dogs, two distinct, major clones of MRSP have spread across Europe and North America, respectively.9 Many isolates of S. pseudintermedius not only are methicillin resistant, but also acquire resistance genes to other classes of antimicrobials, including tetracyclines, trimethoprim-sulfonamides, fluoroquinolones, chloramphenicol, and macrolides. Isolates that are resistant to three or more different classes of antimicrobial drugs are defined as multiple drug resistant (MDR) isolates. Currently, many isolates remain susceptible to chloramphenicol and aminoglycosides, but the prevalence of resistance to chloramphenicol has increased among MRSP isolates. MDR S. pseudintermedius isolates are no more virulent or difficult to disinfect than susceptible isolates, but they are more difficult to eliminate using antimicrobial drugs. Risk factors for MRSP infection identified in dogs include previous hospitalization, living in an urban environment, older age, and, most consistently, previous antimicrobial drug treatment. Household contacts of dogs and cats with MRSP infection are also frequently colonized with MRSP.10 When compared with S. pseudintermedius, S. aureus tends not to colonize dogs. For example, staphylococci were isolated from 37 of 50 healthy dogs from Pennsylvania, and 6 (12%) of the dogs were colonized with S. aureus; 68% were colonized with S. pseudintermedius.11 MRSA strains that colonize dogs may be identical to those that infect in-contact humans, but sometimes strains found on dogs differ from those found on in-contact humans.12 The chance that a dog that is in contact with an infected human will be colonized with MRSA decreases with the time after diagnosis of human MRSA infection, which suggests transient carriage of MRSA by dogs. MRSA was not transferred between apparently healthy dogs in a kennel of dogs, and after treatment of an MRSA wound infection in one dog, all dogs tested negative for MRSA within 2 weeks.13 Although staphylococci are isolated less commonly from cats, colonization with S. aureus is more prevalent and certain S. aureus strains may be commensals of cats; staphylococci were isolated from 17 of 50 healthy cats, 59% of which were colonized with S. aureus and 65% with S. pseudintermedius.14 In dogs and cats, S. aureus infection has been associated with clinical manifestations that include pyoderma, otitis externa, UTIs, surgical site infections, osteomyelitis, and bacteremia. Although MRSA infections are uncommonly diagnosed in dogs and cats, they have been increasingly recognized over the past 2 decades. When canine infections with MRSA do occur, they are often of human hospital origin, but human community-acquired strains have also been isolated. In one study, strong risk factors for MRSA infection (as opposed to methicillin-susceptible S. aureus [MSSA] infection) were the number of antimicrobial courses administered, the number of days admitted to veterinary clinics, and a history of surgical implant placement.15 There has been concern that the use of certain antibiotics, such as cephalosporins and fluoroquinolones, may select for MRSA infections in dogs. Coagulase-positive and coagulase-negative S. schleiferi have primarily been isolated from dogs with superficial pyoderma and otitis externa, as well as from the ear canals of healthy dogs. Rarely, UTIs and respiratory infections with S. schleiferi have been described in dogs.3 In dogs with pyoderma and otitis externa, S. schleiferi is not as prevalent as S. pseudintermedius but in most studies has been isolated as often, or more often, than S. aureus. Coagulase-negative S. schleiferi is a commensal of the human axilla but can cause disease in immunosuppressed humans. Coagulase-positive S. schleiferi may be specialized to colonize dogs. S. schleiferi isolates from dogs with skin disease are often methicillin-resistant as a result of mecA gene carriage. In a study of 225 S. schleiferi isolates from dogs from the eastern United States, of which 52% were coagulase negative and 42% were coagulase positive, methicillin resistance was identified in 57% of the isolates.3 Methicillin resistance was more prevalent among coagulase-negative isolates. Isolates from dogs with pyoderma may be more likely to be methicillin resistant than those from dogs with otitis externa. Signs and Their Pathophysiology S. aureus and S. pseudintermedius possess several adhesins and produce an array of toxins. The clinical effect of these toxins is best characterized for S. aureus infections in humans. A number of enterotoxins have been identified in S. aureus and S. pseudintermedius; these can cause food poisoning in humans but have not been recognized as a cause of disease in dogs and cats. Staphylococcal enterotoxins are heat stable, so they are not inactivated by cooking, even when the bacteria themselves are destroyed. Staphylococcal toxic shock syndrome results from the expression of superantigens, which include toxic shock syndrome toxin 1, as well as a large number of enterotoxins and exotoxins that can also act as superantigens. Cellulitis, necrotizing fasciitis, and toxic shock syndrome has been rarely reported in dogs infected with S. pseudintermedius.16,17 Most toxic shock syndrome and necrotizing fasciitis cases in dogs and cats are instead caused by Streptococcus canis (see Chapter 34). Scalded skin syndrome results from infection of humans with S. aureus strains that produce an exfoliative toxin, which hydrolyses the intercellular glycoprotein desmoglein-1. Strains of S. pseudintermedius that possess exfoliative toxin genes have been isolated more commonly from dogs with pyoderma than from healthy dogs, so exfoliative toxin may also play a role in canine disease. Panton-Valentine toxin (PVL) is a leukocidin that is found in CA-MRSA strains but only a small percentage of HA-MRSA and MSSA strains, and strains that encode PVL have been associated with skin and soft tissue infections and hemorrhagic pneumonia in children and young adults. Although a similar toxin has been identified in S. pseudintermedius, the role of PVL in the pathogenesis of staphylococcal infections in dogs and cats is unknown. Some strains of S. pseudintermedius and S. aureus produce biofilm, which may contribute to the ability of these organisms to persist in the environment, cause nosocomial infections, and chronic recurrent infections that respond poorly to antimicrobial treatment.18
Staphylococcus Infections
Etiology and Epidemiology
Staphylococcus pseudintermedius
Staphylococcus aureus
Staphylococcus schleiferi
Clinical Features
Veterian Key
Fastest Veterinary Medicine Insight Engine



