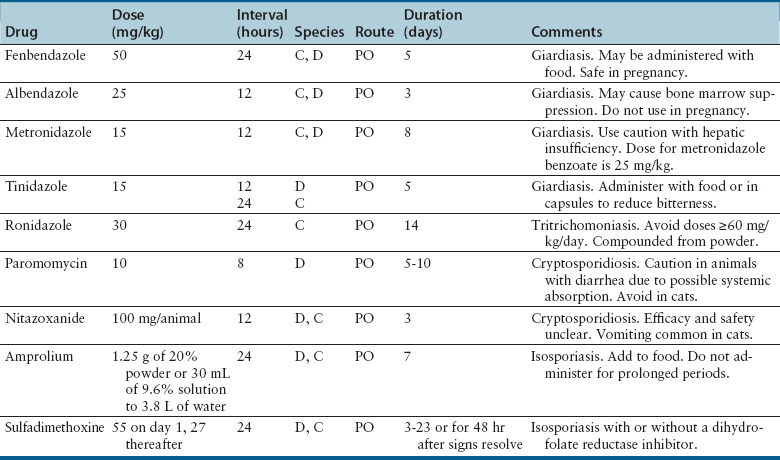
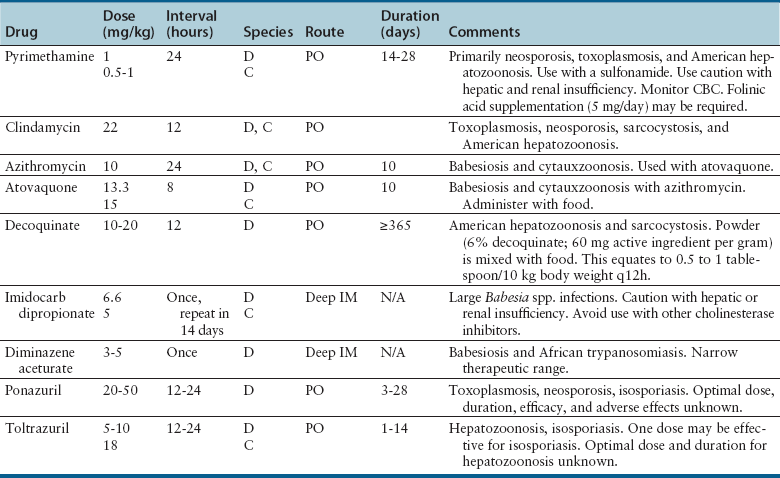
Chapter 10 • Antiprotozoal drugs often have a restricted spectrum of activity, although some are also active against bacteria and fungi. Many interfere with enzyme pathways specific to certain protozoal species. • Treatment with antiprotozoal drugs may not consistently clear an infection. • Resistance to antiprotozoal drugs is a growing problem among protozoal pathogens. • Some antiprotozoal drugs have minimal to no adverse effects, whereas for others, adverse effects severely limit use. • The availability of antiprotozoal drugs is restricted in some countries. The use of antiprotozoal drugs in dogs and cats is frequently extrapolated from their use in human patients or food animal species. Almost all antiprotozoal drugs are not specifically approved for treatment of protozoal infections in dogs and cats. Some antibacterial and antifungal drugs also have antiprotozoal activity (see Chapters 8 and 9 for the use and adverse effects of these drugs). In vitro activity of antiprotozoal drugs, and monitoring of resistance, is more difficult for antiprotozoal drugs because standardized susceptibility testing is not routinely performed for these pathogens. In addition, many antiprotozoal drugs are designed to be active in the lumen of the intestine for treatment of intestinal protozoal infections and the concentration of active drug in the intestinal lumen after oral administration is difficult to measure. Therefore, the concentration of drug to which these pathogens are exposed is often not known. The activity and dosage regimens of antiprotozoal drugs are often based on the results of clinical trials, rather than concentration-exposure relationships between antiprotozoal drugs and the organism of interest. Amprolium is a thiamine analogue that is used to prevent and treat intestinal coccidiosis (Table 10-1). It is available as a feed additive for livestock and is sometimes administered in food or drinking water to puppies and kittens. Adverse effects of anorexia or diarrhea are rare and primarily occur at high doses and with prolonged use. Central nervous system (CNS) signs can result from thiamine deficiency, which is reversible on addition of thiamine to the diet. However, thiamine supplementation may interfere with the drug’s efficacy. Fenbendazole is widely used to treat giardiasis in dogs and cats. It is safer than metronidazole, can be administered to young animals, and has higher efficacy, although treatment failure can still occur. A second course of treatment or administration of fenbendazole in combination with metronidazole can be effective in refractory cases. Administration with food may improve absorption, but the fat content of the food does not influence absorption.1 Adverse effects of fenbendazole are very rare but can include decreased appetite, vomiting, diarrhea, and rarely reversible pancytopenia.2 At high doses used to treat Mesocestoides spp. peritonitis (100 mg/kg q12h), neurologic signs have been observed.3 Febantel is metabolized to a benzimidazole compound and has been used in combination with praziquantel and pyrantel (Drontal Plus) to treat Giardia spp. infection in dogs, although efficacy at label dosages has been variable and some dogs can re-shed low numbers of cysts when treatment is discontinued.4 Albendazole has an affinity for rapidly dividing cells, and although it is used extensively for treatment of parasitic infections in human patients, it been associated with anorexia and reversible bone marrow suppression in dogs and cats especially when high doses are administered for more than 5 days.5 As a result, fenbendazole is used more commonly in small animals. Protozoa reduce nitroimidazoles to nitro anion free radicals, which cause damage to parasite DNA. Some nitroimidazoles are mutagens and carcinogens, but carcinogenesis has not been demonstrated in dogs and cats with long-term use. Metronidazole, ronidazole, and tinidazole have primarily been used to treat enteric protozoal infections. Benznidazole is specifically used to treat infections with Trypanosoma cruzi. Metronidazole is used to treat giardiasis in dogs and cats, although efficacy may be as low as 50%. It also has activity against amoebic infections. The clinical use and adverse effects of metronidazole are described in Chapter 8. Doses of metronidazole used for treatment of giardiasis have the potential to be associated with neurotoxicity, so fenbendazole is preferred because of greater safety and efficacy. Metronidazole can be combined with fenbendazole for refractory giardiasis. Tinidazole is a 5-nitroimidazole that has amoebicidal, giardicidal, trichomonicidal, and anaerobic bactericidal activity. It is sometimes used as a single-dose treatment for giardiasis in human patients. The efficacy of tinidazole for treatment for giardiasis in dogs and cats has not been evaluated, and the half-life in dogs (4.4 hours) and cats (8.4 hours) is shorter than that in human patients (>12 hours).6,7 Tinidazole is very well absorbed in dogs and cats, with a bioavailability of 100%. Adverse effects are similar to those of metronidazole. Like metronidazole, tinidazole has a bitter taste. Ronidazole is the drug of choice for treatment of Tritrichomonas foetus infections, which are less responsive to metronidazole and tinidazole.8–10 Resistance to ronidazole has been identified in some isolates of T. foetus and is associated with treatment failure in infected cats.11 Resistance is thought to result from increased oxygen-scavenging capacity by the parasite, whereby oxygen competes effectively with ronidazole and other nitroimidazoles for ferredoxin-bound electrons. Ronidazole is absorbed rapidly and completely after oral administration to cats. Some compounded formulations may have decreased efficacy as a result of low ronidazole content or differences in drug release at the site of action (the large bowel). A modified-release formulation that is delivered to the colon may have improved efficacy.12 Decreased appetite, vomiting, and neurologic signs can occur in dogs and cats, especially at doses above 30 mg/kg q12h in cats and at doses as low as 10 mg/kg/d in dogs.13 Once daily dosing is probably sufficient because of the long half-life of the drug in cats.12 Doses of 20 mg/kg or less may not effectively clear infection with T. foetus. Neurologic signs result from γ-aminobutyric acid (GABA) antagonism in the CNS and include ataxia, decreased mentation, agitation, tremors, and hyperesthesia, which occur up to 9 days after the start of treatment and resolve when the drug is discontinued.13 Nitazoxanide is a nitrothiazolyl-salicylamide derivative that has activity against Giardia spp., Cryptosporidium spp., Sarcocystis neurona, some anaerobic bacteria, Helicobacter spp., and Campylobacter jejuni. It inhibits the pyruvate-ferredoxin/flavodoxin oxidoreductase enzyme-dependent electron transfer reaction that is essential for anaerobic metabolism in these organisms. Resistance has been documented in Giardia spp.14 Reports of nitazoxanide use in dogs and cats have been rare, and its efficacy in dogs and cats is largely unknown. An equine formulation (Navigator) that was used to treat equine protozoal meningoencephalitis caused by Sarcocystis neurona has been removed from the market. Doses have been extrapolated from those used for human patients. Nitazoxanide treatment of cats co-infected with Cryptosporidium spp. and T. foetus led to cessation of shedding during treatment, but infection was not eliminated.15 Vomiting occurred frequently, especially at higher doses (75 mg/kg PO q12h). In humans, nitazoxanide is rapidly absorbed from the gastrointestinal tract and metabolized to the active metabolite tizoxanide, which is highly protein bound. After hepatic glucuronidation, it is excreted in urine and bile. Trimethoprim, pyrimethamine, ormetoprim, and sulfadiazine inhibit parasite replication through folate antagonism. Synergistic combinations of sulfadiazine with trimethoprim or pyrimethamine are primarily used to treat toxoplasmosis, neosporosis, and intestinal coccidiosis (Isospora spp. infections) in dogs and cats. A combination of pyrimethamine, trimethoprim-sulfadiazine, and clindamycin has also been used to treat Hepatozoon americanum infections.16 The mechanisms of action, use, and adverse effects of trimethoprim and sulfonamides are discussed in Chapter 8. Pyrimethamine is well tolerated. Gastrointestinal signs such as vomiting, diarrhea, and decreased appetite occur in some treated animals. Bone marrow suppression can occur with prolonged treatment at higher doses as a result of folic acid deficiency. In human patients, concurrent administration of folinic acid is recommended when high doses are used for treatment of toxoplasmosis. Folinic acid, but not folic acid supplementation also reverses marrow suppression in dogs treated with pyrimethamine.17 The CBC should be monitored weekly during treatment, and supplementation should be provided if leukopenia develops and continued treatment is necessary. Stomatitis, ulcerative glossitis, and exfoliative dermatitis have also been described in human patients as a result of folic acid deficiency.18 Other adverse effects of pyrimethamine-sulfadiazine combinations result from the sulfadiazine component (see Chapter 8). Clindamycin, azithromycin, and clarithromycin have antiprotozoal activity. The use of these macrolides and lincosamides in dogs and cats and their adverse effects are discussed in Chapter 8. Clindamycin is the most widely used antiprotozoal for treatment of toxoplasmosis and neosporosis in dogs and cats. Although clindamycin inhibits shedding of Toxoplasma gondii oocysts by cats,19 clinical efficacy of clindamycin for treating toxoplasmosis in dogs and cats has been questioned by experts and in published studies. Trimethoprim-sulfonamides are a suitable alternative, or if clindamycin is used, pyrimethamine may be used in combination. In human patients, pyrimethamine and clindamycin are used as a substitute for pyrimethamine and sulfadiazine for treatment of toxoplasmosis in sulfadiazine-sensitive individuals. Azithromycin is used in combination with atovaquone for treatment of babesiosis and cytauxzoonosis. Paromomycin is the only aminoglycoside antibiotic that has efficacy against protozoa. It is poorly absorbed from the gastrointestinal tract and so has been used to treat enteric protozoal infections, particularly cryptosporidiosis. It is ineffective for treatment of tritrichomoniasis in cats.10 Furthermore, when used to treat intestinal protozoal infections in cats, paromomycin has been absorbed systemically because of intestinal mucosal compromise, with resultant acute renal failure, deafness, and cataract formation.20 As a result, its use has been limited. In human patients, paromomycin has been used topically to treat cutaneous leishmaniasis and parenterally to treat visceral leishmaniasis.21 Doxycycline has primarily been used for malaria prophylaxis in humans. Ciprofloxacin is thought to inhibit DNA gyrase within a chloroplast organelle (the apicoplast) of apicomplexan parasites (see the triazines, later). It is an alternative to sulfadiazine for treatment of isosporiasis in human patients.18 Tetracyclines and ciprofloxacin have not been widely used for prevention or treatment of protozoal infections in dogs and cats with the possible exception of doxycycline as part of combination treatment for babesiosis (see Chapter 75). Atovaquone is a hydroxynaphthoquinone that inhibits electron transport in protozoa by targeting the cytochrome bc1 complex (Table 10-2). It has been used in combination with other antiprotozoal drugs as an alternative treatment for malaria, toxoplasmosis, and pneumocystosis in human patients.18 In veterinary medicine, atovaquone is used in combination with azithromycin for treatment of Babesia gibsoni and Babesia conradae infections and cytauxzoonosis (see Chapters 75 and 76), but is expensive. Resistance has been reported in Plasmodium spp., T. gondii, Pneumocystis jirovecii, and canine B. gibsoni strains as a result of mutations in the cytochrome bc1 complex. TABLE 10-2 Suggested Doses of Drugs Primarily Used to Treat Systemic Protozoal Diseases Excluding Leishmaniosis and Trypanosomiasis in Small Animals
Antiprotozoal Drugs
Introduction
Antiprotozoal Drugs Used Primarily for Gastrointestinal Infections
Benzimidazoles
Nitroimidazoles
Metronidazole
Tinidazole
Ronidazole
Nitozoxanide
Antibacterial Drugs with Broad-Spectrum Antiprotozoal Activity
Pyrimethamine
Macrolides and Lincosamides
Paromomycin
Tetracyclines and Ciprofloxacin
Antiprotozoal Drugs Used for Systemic Protozoal Infections
Atovaquone
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Antiprotozoal Drugs
Only gold members can continue reading. Log In or Register to continue