Chapter 15 Spring Viremia of Carp Virus
Spring viremia of carp (SVC) is a reportable disease caused by a rhabdovirus (Rhabdovirus carpio) frequently referred to as spring viremia of carp virus (SVCV). Until recently (2002), SVCV had not been found in North America; it is considered a foreign animal disease (FAD) in the United States. In summer 2002 the disease was identified in koi (Cyprinus carpio) from a North Carolina fish farm that also owned pond facilities in Virginia.10 Subsequently, cases have been confirmed in other states. Warg et al review the U.S. cases from 2002 to 2004, summarize the diagnosis and pathophysiology of SVCV, and describe genetic relationships among various viral strains.23
Diagnosis is usually made with viral isolation (from spleen or caudal kidney) and serum antibody titers. Diagnosis should be confirmed with virus neutralization.15 The disease is not restricted to koi and actually may affect several carp species and some other cyprinids (Box 15-1).3,7 All suspect cases should be necropsied and the U.S. Department of Agriculture (USDA) contacted for proper routing of diagnostic samples. Confirmed cases must be reported to the USDA.
Box 15-1 Fish Species Susceptible to Spring Viremia of Carp Virus (SVCV)
A complete summary of the disease and diagnostic procedures may be found on the Office International des Epizooties (OIE) website.16 It is important to note that SVCV-infected fish may also have a gram-negative bacterial infection.18
CLINICAL SIGNS
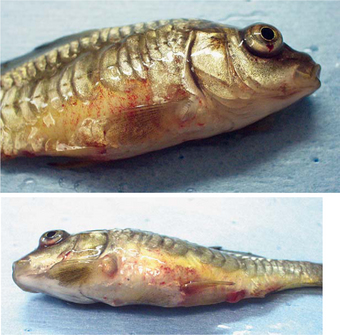
Early in the course of SVCV, affected fish may appear weak and may congregate in areas of slow-flowing water. Infected fish may present with a variety of clinical signs, including but not limited to lethargy, ascites, exophthalmia, pale gills, overall darkening of the body surface, persistent fecal casts, skin and branchial hemorrhages, and distention/protrusion of the vent2 (Figure 15-1).
On necropsy, affected fish may have generalized edema (the fluid may be sanguineous), swim bladder (and other organ) hemorrhages, and intestinal inflammation. The gastrointestinal tract may contain mucus and not ingesta.14
DIAGNOSIS
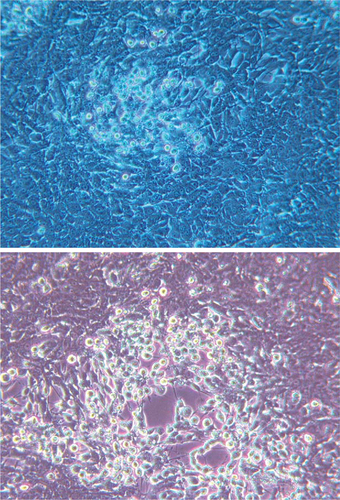
A variety of serology and polymerase chain reaction (PCR)–based assays are available to veterinarians and fish health professionals for documenting the presence of SVCV. The World Organization for Animal Health (OIE) set the international standards for diagnosing SVCV in 2000. The diagnosis of SVC in clinically infected fish may be carried out by rapid immunologic assays such as direct immunofluorescence (IF) or enzyme-linked immunosorbent assays (ELISAs) on infected tissues. However, OIE recommends that the results from these tests be further confirmed by virus isolation in cell culture (Figure 15-2), followed by virus neutralization (VN) test. In situations in which it is not possible to isolate the virus (e.g., decomposed clinical samples), clinical signs of SVC and a positive direct IF test or ELISA are considered sufficient to initiate control measures.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree