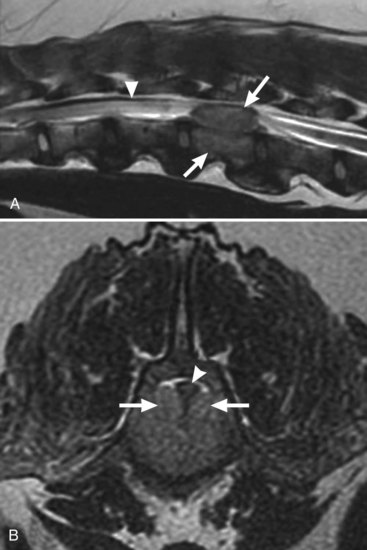
Chapter 39 The current availability of cross-sectional imaging, in particular magnetic resonance imaging (MRI), has enhanced our ability to identify spinal tumors. Increased owner interest in the pursuit of diagnostics and treatment, combined with the developing fields of veterinary neurosurgery and oncology, is leading to more frequent definitive diagnoses and therapeutic interventions. In addition to surgery, radiation therapy has emerged as a useful primary or adjunctive option for improving the quality of life and survival for animals with spinal tumors. With these resources, through clinical studies and pioneering efforts of veterinary neurosurgeons and oncologists, rapid advancement of neuro-oncology is under way. This chapter reviews the diagnosis and treatment of spinal tumors. Following general discussions of diagnosis and treatment, information regarding individual tumor types is presented according to their anatomic relationship with respect to the meninges and spinal cord (i.e., extradural or intradural/extra-axial or intra-axial).* In dogs, most spinal tumors occur in animals older than 5 years.84,120,160,161 Affected dogs range in age from 5 months to 12 years.160,161 With the exception of extrarenal nephroblastoma, a tumor of young dogs, there does not seem to be an association between tumor type and age. In cats with spinal tumors, the average age at presentation is approximately 8 years.86 Cats with spinal lymphoma tend to present at a younger age, at an average of 3.5 to 4 years.74,86,137 Cats with nonlymphoid spinal tumors present at an older age, at an average of 7 to 12 years.79,86 An association between tumor type and breed of dog is lacking. However, large-breed dogs represent more than 50% of reported cases.78,84,160,161 In a report on 72 dogs with spinal tumors, 66.7% were large breeds, 23.6% were small breeds, and 9.7% did not have breed reported.84 Although no sex predilection has been reported, in a few reports male dogs predominate.78,84,160,161 Most affected cats are domestic shorthair cats.74,79,86,137 Cats with spinal lymphoma often are feline leukemia virus (FeLV)-positive, and cats with nonlymphoid spinal tumors tend to be FeLV-negative.74,79,86,137 Classically, dogs with spinal neoplasia have a chronic progressive clinical history. In many affected dogs, clinical signs are present for at least 1 month before presentation.84,160 However, approximately 50% of dogs may present with an acute history.84 Therefore, an acute presentation of clinical signs does not differentiate neoplastic disease from other disorders affecting the spinal cord. The onset and rate of progression of clinical signs depend on the growth rate of the tumor, the degree of compression or invasion of the neural structures, and secondary consequences such as spinal cord ischemia or hemorrhage. In most cases, the time between onset of clinical signs and presentation is longest for intradural/extra-axial and shortest for intra-axial lesions. The average duration of clinical signs before presentation has been reported to be 5.7 weeks for intradural/extra-axial, 3 to 4 weeks for extradural, and 1.7 weeks for intra-axial lesions.84 Differences in time to presentation likely reflect tumor biology (i.e., slow-growing tumors cause chronic deterioration) rather than an inherent difference related to anatomic location with respect to the meninges and spinal cord. In cats with spinal tumors, the average duration of clinical signs is approximately 50 days.86 Cats with lymphoma tend to have a more rapid onset.74,86,137 Clinical signs are related to compression or invasion of the vertebra(e) or neural structures. The most common clinical sign in dogs with spinal tumors is pain. Pain may localize to the site of the lesion or may be referred to sites innervated by affected spinal cord segments or nerve roots. In cats, the most common clinical sign is paresis or paralysis.86 In some animals, pain is the only clinical sign. When clinical signs are considered in relation to site of tumor, animals with lesions affecting the spinal cord commonly exhibit pain, varying degrees of paresis/paralysis, and occasionally fecal/urinary incontinence. Often, signs are asymmetric. In animals with lesions isolated to the peripheral nerves, signs may involve only a single limb and may consist of paresis/paralysis or may appear as lameness. Alternatively, the animal may hold the affected limb off the ground (root signature). Ultimately, the anatomic localization of the lesion determines the nature of the clinical signs. Lesions affecting the C1-C5 spinal cord segments result in general proprioceptive ataxia and upper motor neuron paresis/paralysis of all four limbs. Lesions affecting the C6-T2 spinal cord segments result in lower motor neuron paresis/paralysis involving the thoracic limbs and general proprioceptive ataxia/upper motor neuron paresis/paralysis involving the pelvic limbs. Lesions affecting the T3-L3 spinal cord segments result in general proprioceptive ataxia/upper motor neuron paresis/paralysis involving only the pelvic limbs. Lesions affecting the L4-S1 spinal cord segments, nerve roots, or peripheral nerves originating from these spinal cord segments result in lower motor neuron paresis/paralysis involving only the pelvic limbs. Finally, lesions involving the S1-S3 spinal cord segments, nerve roots, or peripheral nerves originating from these spinal cord segments results in urinary/fecal incontinence and may not affect pelvic limb strength or gait. (See Chapter 26 for details on neurologic examination and localization.) With respect to location along the vertebral column, spinal tumors in dogs tend to occur most commonly in the cervical region.78 With respect to location in relationship to the meninges and spinal cord, extradural tumors account for 50% or more of cases.78,84,120,144 In some reports, intradural/extra-axial tumors account for 27% to 35% of cases, with the remainder of cases being intra-axial.78,120 In another report, intra-axial tumors accounted for 27%, and intradural/extra-axial tumors accounted for 17% of cases.84 The minimum database required for animals with suspected spinal tumors should include complete blood count, chemistry profile, urinalysis, and, for cats, FeLV/feline immunodeficiency virus (FIV) testing. Animals with secondary tumors frequently display clinical signs related to a primary neoplasm elsewhere. Even nonspecific signs of systemic illness such as lethargy, anorexia, or weight loss or abnormalities involving the minimum database should increase suspicion for a secondary spinal tumor.96 In such cases, diagnostic evaluation should be directed initially toward identification and histologic or cytologic diagnosis of the primary tumor. Knowledge of the diagnosis and the presence of metastasis may provide prognostic information that will influence the decision to perform more advanced diagnostics or invasive procedures. In all animals, a three-view radiographic evaluation of the thoracic cavity and ultrasonographic evaluation of the abdominal cavity aimed at excluding the presence of an underlying primary tumor should be performed. Following this initial screening, the diagnostic workup will focus on imaging the vertebral column and spinal cord. MRI is the gold standard modality for evaluation of the spinal cord, vertebral column, and adjacent soft tissue structures.68,77 MRI provides excellent discrimination of soft tissue structures with excellent resolution. Images can be obtained in multiple anatomic planes without loss of image quality. Given these attributes, MRI enables better visualization of the primary lesion, recognition of secondary changes to the spinal cord, and identification of extravertebral extension of a lesion. Extravertebral extension is not as readily identified with other imaging modalities. Finally, MRI evaluation using appropriate imaging sequences before and after administration of intravenous contrast media combined with lesion characteristics such as anatomic location in relationship to the vertebrae, meninges, and spinal cord, number of lesions, intensity, shape, and definition of borders may provide an accurate presumptive diagnosis. It is important to note that MRI provides excellent discrimination of tumors from other disease processes. The main disadvantages of MRI are the need for general anesthesia, the expense, and the technical expertise required for image procurement and interpretation. The relative importance of these drawbacks is outweighed by the diagnostic information gained with the use of MRI. Other imaging techniques may also play a role in the diagnosis of spinal tumors. Radiography is an inexpensive means of evaluating the vertebral column and adjacent soft tissue structures. To obtain images of diagnostic quality, sedation or anesthesia is often required for proper positioning. Additional collimated images centered on a questionable or definitive lesion may improve image quality, thereby aiding in image interpretation. The utility of spinal radiographs is limited to investigation of bony structures; consequently, these are useful for detection of primary or secondary vertebral tumors only if sufficient bone lysis is present.96 Plain radiography is incapable of providing a specific evaluation of the nervous tissue, epidural space, and subarachnoid space. In addition to allowing radiographic identification of bony abnormalities of the vertebral column, myelography can facilitate localization and definition of the extent of spinal lesions compared with plain radiography alone. On the basis of myelographic pattern, lesions can be ascribed to three patterns: extradural, intradural/extra-axial, and intra-axial. CT allows more accurate assessment of bony structures over radiography. However, diagnostic information provided by CT may not be as accurate as that provided by myelography for defining soft tissue lesions in the vertebral canal, especially with intra-axial lesions.38 CT myelography has increased diagnostic sensitivity over CT alone. As with neoplasia elsewhere in the body, surgical excision is the treatment of choice for local disease, with the exception of tumor types that are highly radiation- and chemotherapy-responsive (e.g., lymphoma, multiple myeloma). Cross-sectional imaging should be used to aid in the determination of the feasibility of surgical intervention. Given the lack of redundancy and the delicate nature of nervous tissue, as well as the need for biomechanical stability, resection of spinal tumors is often limited to gross cytoreduction with tumors involving the vertebral column. It is important to note that gross cytoreduction may afford relief of clinical signs for an extended period of time in some animals and may increase the efficacy of adjuvant therapies such as radiation therapy and chemotherapy.78 When possible, postoperative adjunctive therapy should be considered for animals with incompletely excised tumors or those at risk for metastasis. Radiation therapy is a useful alternative or adjunctive local treatment for tumors of the vertebral column or spinal cord. It is indicated to improve local control of tumors that are incompletely excised, or may be used as sole therapy for tumors that are inoperable. It may be used in a definitive manner to control a tumor or in a palliative manner to relieve pain or other clinical signs resulting from osteolysis or spinal cord and or nerve compression caused by a tumor. Although complete excision offers the best option for long-term control of neoplasia, radiation therapy presents an alternative when surgery is contraindicated or is unsuccessful because of tumor size or location. Only limited information is available in the veterinary literature to describe outcome following radiation therapy for spinal tumors. In one small study, dogs treated with definitive radiation therapy for spinal tumors (primarily meningioma) survived a median of 17 months (range, 6.5 to 70 months).135 High doses of radiation therapy are effective for killing tumor cells. Unfortunately, radiation therapy injures surrounding normal tissues, limiting the dose that can be achieved. Adverse effects of radiation are classified as early (acute) or late (chronic).54,91 Early effects occur in proliferating tissues such as epithelium and bone marrow.138 They start during administration of radiation therapy and resolve within weeks of completion of radiation therapy. Early morbidity is uncommon with radiation of the spinal cord or vertebrae. Although rare with standard fractionation, initial worsening of clinical signs is possible and might be attributable to acute infarction or peritumoral edema.55 Late adverse effects involve nonproliferating tissues such as nervous tissue, vascular tissue, and bone. These effects occur several months to years after radiation therapy55,138 and include white matter necrosis; atrophy, hemorrhage, or infarction of nervous tissues; chronic progressive myelitis due to vascular injury; and fibrosis/gliosis.106,118 Radiation-induced second tumors (typically sarcomas) in the treatment field represent an extremely rare late effect observed many years after radiation therapy.3,92 Development of vertebral osteosarcoma secondary to radiation therapy has been reported in the dog.33 Late effects are significant because they do not resolve and can be life-limiting. Consequently, these effects limit the total radiation dose that can be administered. Because late effects are also related to the dose per fraction, radiation therapy to the spinal cord or vertebrae typically involves the administration of multiple small fractions to decrease risk. The greater length or volume of the irradiated section of the spinal cord, concurrent chemotherapy, and prior central nervous system damage may also influence (increase) the risk of late side effects.119 Although potentially devastating, the expected risk of serious late radiation effects is very low (5%) with definitive radiation protocols.56,60 In general, animals treated with palliative protocols are not expected to survive long enough to experience late effects, so increased risk is accepted. Transient demyelination may occur within the first 6 months after radiation therapy, resulting in worsening of clinical signs. A unique clinical presentation of this radiation toxicity, Lhermitte’s syndrome, is well documented in human beings, and anecdotally similar signs have been described in veterinary patients that have undergone spinal radiation therapy.39,54,55 In people, this reversible toxicity can occur 2 to 4 months after treatment and manifests clinically as abnormal twitching, shock-like sensations radiating down the spine and into the extremities (the animal may act uncomfortable or may look at or lick the radiation therapy site or the innervated area), or paresis.39,54 Affected patients are treated with corticosteroids, and signs typically resolve over a few months. It is a challenge to distinguish transient demyelination from progressive disease; additional cross-sectional imaging may be required. Intensity-modulated radiation therapy increases dosing precision through multiple beams of radiation shaped by a dynamic multileaf collimator with “leaves” that move as directed by computer control to conform the treatment field to the tumor, even altering the dose within the treatment field.67,156 This technique allowed avoidance of spinal cord injury with delivery of a high dose to the canine paraspinal region.67 Image-guided radiation therapy verifies tumor position at each treatment and ensures accurate localization of the radiation therapy dose. Helical tomotherapy is a form of image-guided intensity-modulated radiation therapy. Before each treatment, the tomotherapy unit (Aredia, Novartis Pharma, East Hanover, NJ) performs a megavoltage CT and compares this with the planning CT. This information is used to adjust patient position to precisely repeat the position for each treatment. Radiation is delivered by a linear accelerator with a circular gantry. A multileaf collimator–modulated, fan-shaped beam rotates around the patient multiple times during each treatment.53 Stereotactic radiation therapy involves three-dimensional localization of the tumor with imaging following immobilization of the part of the patient to be radiated.76 Multiple beams of radiation are administered from different directions to deliver a high dose to a more localized area (small lesions), typically as one to a few treatments. Samarium-153 EDTMP (samarium-153 lexidronam, Sm-153) is an injectable radioisotope that concentrates in areas of osteoblastic activity. This drug has been used for palliation of pain due to osteosarcoma and metastatic bone tumors in dogs. Response rates were 60% to 80% for 3 months.8,75 Side effects primarily involve myelosuppression. With the exception of lymphoma, plasma cell neoplasia, and osteosarcoma, only limited information is available about the use of chemotherapy in treating spinal tumors. Chemotherapy for intradural central nervous system tumors presents a unique challenge in that many chemotherapy agents do not penetrate the blood–spinal cord barrier. In addition to limiting passive diffusion, the blood–spinal cord barrier serves as a site for normal expression of p-glycoprotein, an adenosine triphosphate (ATP)-dependent efflux pump that actively removes substrate chemotherapy agents that have crossed the blood–spinal cord barrier. Despite the fact that some tumors and associated inflammation may disrupt the blood–spinal cord barrier, limitations remain. In addition, it is important to note that, with the exception of hematopoietic tumors, significant response of gross disease neoplasia to chemotherapy is unlikely and is not expected to be rapid. For this reason, with the exception of lymphoma, leukemia, multiple myeloma, and disseminated histiocytic sarcoma, chemotherapy is unlikely to be used as the primary treatment for spinal tumors. For chemotherapy-responsive tumors, treatment protocols are chosen on the basis of known efficacy. Chemotherapy may also be used adjunctively for tumor types that have a higher likelihood of metastasis. In general, agents are well tolerated with serious side effects reported in <5% of animals.23 Clinicians administering chemotherapy must understand which adverse effects are associated with each drug and how to manage these effects.148 Referral to an oncologist should be considered. It is important to consider safety issues when treating patients with spinal tumors with cytotoxic cancer chemotherapy. These agents are mutagenic, carcinogenic, and teratogenic, so it is important to follow safety precautions during preparation, administration, and disposal. In addition, patients with spinal cord compression may suffer from urinary incontinence, which could result in exposure of staff and owners to excreted chemotherapy drugs and metabolites. Clinicians prescribing chemotherapy must understand the risks and routes of excretion of these agents and are responsible for educating staff and clients.64 Recommended precautions should be followed, including preparing drugs in a biosafety cabinet, use of closed system transfer devices, wearing personal protective gear, administering drugs in a low-traffic area, and properly disposing of waste through a certified company. Women who are pregnant or nursing, couples actively trying to conceive, and children should not handle chemotherapy drugs. For further information, the reader is directed to http://www.cdc.gov/niosh/docs/2004-165/2004-165b.html#. Bisphosphonates may be useful in the treatment of spinal tumors causing bone lysis. These drugs inhibit bone resorption without inhibiting mineralization primarily through induction of osteoclast apoptosis, resulting in decreased osteolysis, stabilized or improved mineralization, and pain relief.42,93 In vitro studies of canine osteosarcoma have shown that bisphosphonates may have anticancer benefits as well.5,43,114 The most commonly used bisphosphonate in veterinary oncology is pamidronate (Aredia, Novartis Pharma, East Hanover, NJ). This drug is administered as an intravenous infusion every 4 weeks at a dosage of 1 to 2 mg/kg. Pamidronate has been reported to cause both tubular and glomerular nephrotoxicity in people,110 and preclinical studies in dogs demonstrated dose- and infusion length–dependent nephropathy.42 Consequently, it is administered IV slowly (over 2 hours), diluted in 0.9% NaCl (250 mL for large dogs, 100 mL for small dogs and cats). Twenty-eight percent of dogs with osteosarcoma treated with pamidronate achieved reductions in pain scores.41 Other bisphosphonates are being investigated, most recently zoledronate (Zometa, Novartis Pharma, East Hanover, NJ).40 Corticosteroids may be used to reduce peritumoral inflammation and spinal cord edema, possibly improving clinical signs. Before administration of corticosteroids, clinicians should rule out lymphoma as a possible cause, as the use of corticosteroids may induce remission, making a definitive diagnosis difficult. Additionally, dogs with multicentric lymphoma treated with corticosteroids before multidrug chemotherapy protocols experience shorter remission times.122 It is important for clinicians to recognize pain associated with spinal tumors and to provide analgesia. For less painful patients, nonsteroidal antiinflammatory drug therapy (if corticosteroids are not used) with or without tramadol may be adequate. With increasing levels of pain, opioids, gabapentin, and amantadine may be used. For severe pain, surgical decompression and radiation therapy may be the only effective options. See Chapters 22 and 23 for more information concerning pain management. Vertebral tumors are the most common spinal neoplasms. As a general rule, lesions involving the vertebrae result in extradural compression of the spinal cord. Large-breed dogs (≥30 kg) are most frequently affected, with the reported median age ranging from 6 to 10 years.* Golden Retrievers, Labrador Retrievers, and German Shepherd Dogs are commonly reported.31,61,96 Male dogs tend to outnumber female dogs.30,96,161 Vertebral neoplasia can be categorized into primary and secondary tumors. In most studies, primary vertebral tumors occur more frequently than secondary.49,78,84,120,160 However, in one study, secondary tumors outnumbered primary tumors.96 Secondary tumors appear to be more common in small-breed dogs.25 Although benign tumors may occur, most vertebral tumors are malignant. The most common primary vertebral tumor in the dog is osteosarcoma.25,80,96 Likewise, osteosarcoma is the most common vertebral tumor in cats.86,87,127 Other tumor types include fibrosarcoma, chondrosarcoma, and hemangiosarcoma.† Primary vertebral neoplasia typically affects a single vertebra. Often, the body of the vertebra is affected, and the pedicle and lamina are less commonly involved.96 Osteosarcoma involving adjacent vertebrae has been documented.95 Most often, the cause of the tumor is unknown; however, radiation-induced vertebral osteosarcoma has been observed.33 Hematopoietic tumors also can involve the vertebrae, but given their frequent multicentric distribution, it is difficult to classify them as primary vertebral tumors. Multiple myeloma, solitary plasma cell neoplasia, and lymphoma are the most commonly encountered hematopoietic tumors affecting the vertebral column (Figure 39-1). Secondary vertebral tumors are the result of metastatic spread from a distant primary tumor or local extension from adjacent soft tissue structures. The most commonly identified metastatic tumors are either vascular in origin (e.g., hemangiosarcoma) or epithelial (thyroid, mammary, prostatic, and urinary [transitional cell] in origin.51,96 Pain is the clinical sign most frequently displayed by affected animals. Tetraparesis/paralysis, hemiparesis, or paraparesis/paralysis based on the anatomic location of the lesion, alone or in conjunction with pain, also may be encountered. Rarely, dogs with primary vertebral neoplasia display signs of systemic illness; however, dogs with secondary vertebral tumors often demonstrate lethargy, anorexia, and weight loss.96 This emphasizes the importance of a minimum database and thorough screening of the thoracic and abdominal cavities before specific diagnostic investigation of the vertebral column is conducted. In contrast to other spinal tumors, radiography of the vertebral column generally discloses abnormalities in animals with vertebral neoplasia.96 Although most lesions are relatively conspicuous, careful attention to proper positioning is essential for correct interpretation of radiographs, given the anatomic complexity of the vertebral column. Radiographic findings include lysis, proliferative changes, and a mixed pattern of lysis and proliferation.96,160 Pathologic fracture is possible. Radiographic changes observed in affected vertebra(e) are similar, regardless of the histologic tumor type.96 However, the presence of a soft tissue mass adjacent to the affected vertebra(e) is more commonly identified in secondary vertebral tumors.96 Additionally, hematopoietic tumors such as multiple myeloma and some metastatic tumors tend to affect more than one vertebra.96 Radiographically, multiple myeloma is characterized by multifocal well-circumscribed lytic lesions affecting multiple vertebrae.58,129 In animals in which radiographic changes are lacking, lesion identification is improved by myelography.160 Vertebral tumors result in an extradural myelographic pattern.103,144,153 Moreover, CT provides improved lesion detection, along with better characterization of bony changes.38 Ultimately, MRI offers enhanced discrimination and characterization of lesions over other imaging modalities. Lesions display variable changes in signal intensity, which allow correct localization of the affected vertebra(e). In comparison to normal vertebrae, lesions are often hypointense on T1-weighted (T1W) images and hyperintense on T2W images, and demonstrate variability in enhancement after intravenous contrast administration.68 Unfortunately, degenerative and nonneoplastic pathologic processes may result in similar changes in signal intensity. Therefore, care must be exercised with image interpretation. Ultimately, regardless of the imaging modality utilized, pathognomonic findings that allow definitive diagnosis of vertebral neoplasia are lacking. Consequently, definitive diagnosis is based on histologic or cytologic evaluation. Depending on the location along the vertebral column, specimens for pathologic evaluation may be obtained via percutaneous fluoroscopically guided or CT-guided biopsy or needle aspiration. Some anatomic locations necessitate surgical biopsy, given the proximity to vital structures. Treatment of solitary primary vertebral tumors is focused on surgical resection. In most cases, only cytoreductive surgery is achievable. In rare instances where lesions involve a small tumor volume affecting an aspect of the vertebra that can be removed, gross resection is possible. Vertebrectomy has been described in the dog.15,163 Despite this, little is known about the utility of the technique in clinical cases, as its application is limited to an isolated case report.77 Given the delicate nature of the spinal cord and therefore inherent significant risks of morbidity (i.e., paralysis) and mortality associated with the procedure, only clinicians with extensive neurosurgical experience should consider vertebrectomy. Moreover, few patients are acceptable candidates for vertebrectomy. Ultimately, vertebrectomy necessitates not only technical expertise for surgical removal of the vertebra but also strict adherence to the same principles and considerations used in the management of spinal fractures to ensure adequate stability of the vertebral column postoperatively. In addition to treating the tumor, surgical resection may result in decompression of the spinal cord and/or nerve roots and clinical improvement. In cats with nonlymphoid vertebral and spinal tumors, maximum neurologic improvement occurred within 1 week of cytoreductive surgery in 25 of 26 cats.126 Plasma cell neoplasia and lymphoma are very responsive to radiation therapy, and radiation therapy is a consideration for other tumor types following cytoreduction, or if the tumor is unresectable. Chemotherapy has been used primarily for hematopoietic tumor and osteosarcoma. Limited information is available describing the efficacy of therapeutic intervention and survival in animals with vertebral tumors. The following sections detail specific vertebral tumor types. Osteochondroma/Osteochondromatosis/Multiple Cartilaginous Exostoses.: Clinically significant osteochondromas are uncommon. This benign lesion involves bones that develop by endochondral ossification, so osteochondroma can affect the vertebrae. Also referred to as solitary or multiple cartilaginous exostoses or osteochondromatosis, lesions may affect single or multiple bones, respectively. Although its classification as a neoplasm is uncertain, its inclusion here is based on its imaging characteristics, which are similar to those of vertebral neoplasms, its clinical behavior, and its potential to undergo malignant transformation (see later). The lesion is believed to arise secondary to migration of chondrocytes from the physeal region into the metaphyseal region of the bone, where continued cartilage formation occurs rather than differentiation into bone.131 The histologic appearance of a cartilaginous cap over the cortex of the bone is diagnostic for osteochondroma. In dogs, no breed or gender predilection exists. The possibility of inherited predisposition has been demonstrated in dogs.20,48 Immature dogs tend to be affected, although the lesion may develop in mature dogs.36 In young dogs, growth of lesions ceases with skeletal maturity. However, solitary or multiple cartilaginous exostoses have the potential to undergo late malignant transformation to chondrosarcoma or osteosarcoma.35,52,107 Osteochondromas occur in cats that have reached skeletal maturity. Solitary osteochondromas typically affect older cats and are more commonly associated with joints. The polyostotic condition, feline osteochondromatosis (also known as multiple cartilaginous exostoses), affects young adult cats.117 Although histologically identical, feline osteochondromatosis differs significantly from the condition in dogs, as no hereditary predisposition has been identified, lesions occur in skeletally mature cats in any location on any bone, and lesions in cats continue to progress beyond skeletal maturity. Osteochondromatosis is associated with feline leukemia virus, with most cats testing positive. In some instances, type C viral particles, suggestive of FeLV or feline sarcoma virus (FeSV), have been identified in bony lesions.116,117 Lesions appear to grow continuously, and new lesions form over time. Malignant transformation to osteosarcoma has been described.37 With vertebral involvement, clinical signs are associated with the anatomic location of the lesion(s) and are related to compression of neural tissue. Presumptive diagnosis is based on finding single or multiple well-circumscribed, calcified lesion(s) affecting the vertebrae and other bones. With MRI, the cartilaginous cap can be identified as a structure that on T1W images is hypointense, and on T2W images is hyperintense compared with spinal cord and isointense compared with cerebrospinal fluid.136 Histopathology is required for definitive diagnosis. Although not evaluated in affected animals, the thickness of the cartilaginous component of the lesion can be used to monitor for malignant transformation. In human beings, cartilage caps greater than 2 to 3 cm thick are consistent with malignant transformation to chondrosarcoma.66 In animals, malignant transformation may be associated with recurrence of a lesion or development of a more rapid growth rate. In dogs and cats, treatment involves surgical resection. Reports of animals treated with surgical resection have been limited to those with single lesions. Depending on lesion location, resection may involve laminectomy alone or combined with removal of an articular process and pedicle using a pneumatic drill. Depending on the extent of resection, careful assessment of vertebral stability may be necessary. In rare instances, resection followed by vertebral stabilization may be needed. In cases in which the lesion is accessible for surgical resection, the prognosis is favorable. Postoperatively, most animals return to normal function. Reported follow-up times range from 9 months to over 2 years, during which time animals have remained normal.11,100,121,123 In one report, acute paralysis developed 4 weeks postoperatively secondary to vertebral instability and resultant subluxation; this highlights the need for careful assessment regarding vertebral stability in animals undergoing extensive laminectomy.131 Over the long term, cats with osteochondromatosis are most commonly euthanized for pain, lameness, or loss of function due to lesions.
Spinal Neoplasia
Signalment
Breed/Gender
Clinical Signs
Tumor Location
Diagnostic Testing
Treatment
Surgical Therapy
Radiation Therapy
Chemotherapy
Symptomatic Care
Specific Tumor Types
Vertebral Neoplasia
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Spinal Neoplasia
Only gold members can continue reading. Log In or Register to continue