RABBIT
Biological average values for the domestic rabbit
Table 1.2 gives the biological parameters for domestic rabbits.
Table 1.2 Biological parameters for the domestic rabbit.
| Biological parameter | Domestic rabbit |
| Weight (kg) | 1.5 (Netherland dwarf) to 10 (New Zealand whites and Belgian hares) |
| Rectal body temperature (°C) | 38.5–40 |
| Respiratory rate at rest (breaths per minute) | 30–60 |
| Heart rate at rest (beats per minute) | 130 (New Zealand whites) to 325 (Netherland dwarf) |
| Gestation length (days) | 29–35 (average 31) |
| Litter size | 4–10 |
| Age at sexual maturity (months) | |
| Male | 5–8 |
| Female | 4–7 |
| Lifespan (years) | 6–10 |
Musculoskeletal system
The skeletal system of rabbits is light. As a percentage of body weight, the rabbit’s skeleton is 7–8%, whereas the domestic cat’s skeleton is 12–13%. This makes rabbits prone to fractures, especially of the spine and the hindlimbs.
Skull
The mandible is narrower than the maxilla, and the temporomandibular joint has a wide surface area, allowing lateral movement of the mandible in relation to the maxilla.
Axial skeleton
The cervical vertebrae are box-like and small and give mobility. The thoracic vertebrae possess attachments to the 12 paired ribs, which are flattened in comparison to cat’s ribs. The pelvis is narrow and positioned vertically. The iliac wings meet the ischium and pubis at the acetabulum, where an accessory bone unique to rabbits, called the os acetabuli, lies. The pubis forms the floor of the pelvis and borders the obturator foramen which is oval in rabbits.
Appendicular skeleton
The scapula is slender and distally has a hooked suprahamate process projecting caudally from the hamate process. The scapula articulates with the humerus which in turn articulates with the radius and ulna. In rabbits, the ulna fuses to the radius in older animals and the two bones are deeply bowed. The radius and ulna articulate with the carpal bones which in turn articulate with the metacarpals and the five digits.
The femur is flatter than a cat ventrodorsally, and the tibia and fibula are fused in the rabbit. The tibia articulates distally with the tarsal bones where there is a prominent calcaneus bone. The tarsals articulate with the metatarsals which articulate with the four hindlimb digits.
The hindlimbs are well muscled and powerful.
Respiratory anatomy
Upper respiratory tract
Rabbits, like horses, are nasal breathers, with the nasopharynx permanently locked around the epiglottis; hence, upper respiratory disease or evidence of mouth breathing is problematic. The nasolacrimal ducts open onto the rostral floor of the nasal passage. The epiglottis is not visible easily from the oral cavity, making direct intubation difficult. It is narrow and elongated and leads into the larynx which has limited vocal fold development. The larynx leads into the trachea which has incomplete C-shaped cartilage rings for support.
Lower respiratory tract
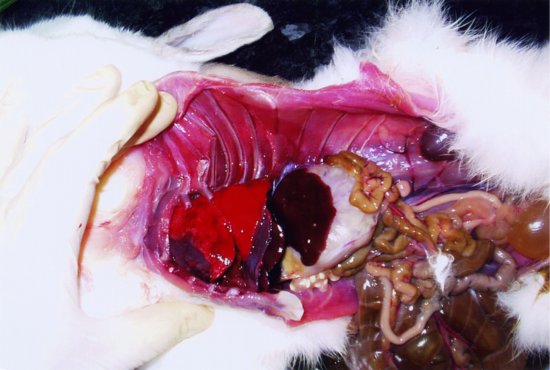
The trachea bifurcates into two primary bronchi. There are two lungs, which are relatively small in proportion to the overall rabbit’s body size. This means that even minor lung disease may cause serious problems. Each lung has three lobes, with the cranial ones being the smallest (see Figure 1.1).
Figure 1.1 Lateral post-mortem view of a rabbit with the chest and abdominal walls removed. The structures from left to right (cranial to caudal) are the dark red heart, bright red lungs (three lobes), darker brown diaphragm and liver, pale cream stomach, yellow brown small intestines (the dark brown large intestines and caecum are reflected ventrally) and yellow-coloured urinary bladder. The left kidney may be seen dorsally as a dark brown structure in the mid-abdomen tucked under a fold of skin. (Fraser and Girling, 2009)
Respiratory physiology
The impetus for inspiration derives from the muscular contraction and flattening of the diaphragm. The lung parenchyma possesses a cellular population that is well supplied with anaphylactic mediating chemicals. These are strong enough to cause fluid extravasation and blood pooling as well as spasms within the walls of the main pulmonary arterial supply, leading to rapid right-sided heart failure.
Digestive system
Oral cavity
The dental formula is
I 2/1 C 0/0 Pm 3/2 M 3/3.
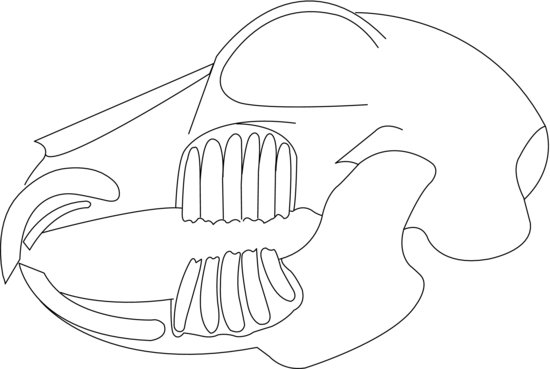
The teeth are elodont (‘open rooted’), allowing continual growth throughout the rabbit’s life. The molar enamel is folded providing an uneven occlusal surface with the ipsilateral jaw which allows interlocking. Wear is kept even by the lateral movement of the mandible, allowing independent left and right arcades to engage in mastication. The incisors differentiate Lagomorpha from Rodentia as rabbits and hares have two smaller incisors, or ‘peg teeth’, behind the upper two, whereas rodents have only two upper incisors. The larger incisors only have enamel on the labial surface, whereas the smaller maxillary peg teeth have enamel on the labial and lingual sides. This creates a wedge-shaped bite-plane where the lower incisors close immediately behind the upper large incisors and fit into a groove made by the peg teeth. The permanent incisors are present at birth, although the peg teeth are replaced by permanent peg teeth at around the second week of life. The deciduous premolars present at birth are replaced and joined by permanent molars by the fourth week of life. There are no canines; instead, there is a gap, or diastema, between the incisors and premolars (Figure 1.2).
Figure 1.2 Lateral diagram of a normal rabbit skull showing the relation of tooth roots to the orbit and jawbones. Note peg-teeth incisors behind main incisors in maxilla.
Stomach
The stomach is a large, simple structure, with a strong cardiac sphincter (see Figure 1.1). This makes vomiting in the rabbit virtually impossible. There is a main body, or fundus, and a pyloric section with a well-formed pyloric sphincter. The lining of the stomach wall contains acid-secreting and separate pepsinogen-secreting cells. The pH of the rabbit’s stomach contents is surprisingly lower than a cat’s or dog’s at 1.5–1.8. In addition, a healthy rabbit’s stomach never truly empties.
Small intestine
The total length of the small intestine in the average rabbit may be some 2–3 feet! It is difficult to determine the divisions between duodenum, jejunum and ileum as they all have a similar diameter.
Caecum and large intestinal anatomy
At the junction of the ileum and caecum lies the sacculus rotundus. This is a swelling of the gut, infiltrated with lymphoid tissue and a common site for foreign body impactions. The caecum is large, sacculated and spiral-shaped, finishing in a blind-ended, thickened, finger-like projection known as the vermiform appendix, which also contains lymphoid tissue. The bulk of the caecum is thin walled and possesses a semi-fluid digestive content.
The start of the large intestine is the ampulla coli which sits near to the sacculus rotundus and caecum. It is a smooth-walled portion of the gut with some lymphoid infiltration of its walls, unlike the rest of the large intestine. It is also distinguished by having bands of fibrous tissue (known as taeniae) which create sacculations (also known as haustra). At the end of the proximal colon, the taeniae and haustra cease, and the gut is then known as the fusus coli, its walls becoming thickened and smooth because of the presence of large members of nerve ganglia which act as pacemakers for contraction waves in the large bowel. The distal descending colon continues through the pelvis to empty through the rectum and anus. There are a couple of anal glands just inside the anus, one on either side, emptying their secretions onto the faecal pellets.
Large intestinal physiology
Two types of faecal pellets are produced by the rabbit. One is a true faecal pellet, comprising waste material in a dry, light brown spherical form. The other is a much darker, mucus-covered pellet known as a caecotroph. The caecotroph is eaten directly from the anus, as soon as it is produced, which in the wild is during the middle of the day when the rabbit is underground. In captivity, they are often produced overnight, but may be produced at any time. The caecotroph contains plant material from which all of the nutrients have yet to be extracted.
The large bowel can produce two types of pellets due to the contraction waves in the large intestine and caecum. The proximal colon can separate out food as the haustra or sacculations of the colon hold on to the smaller particles. The larger particles become pushed towards the colon lumen. The haustra then push the small particles towards the caecum by contracting, and the segmental contractions of the colon itself propel the larger particles towards the rectum producing a waste pellet. When caecotrophs are produced, the haustra dramatically reduce their contractions, and instead the segmental activity drives material from the caecum through the distal colon where they are covered in mucus and then eaten directly from the anus. The caecum is thus the powerhouse filled with microbes which turn the ingesta into volatile fatty acids (VFAs) which can either nourish the caecal epithelium (butyrates) or be absorbed and converted to glucose by the liver (acetates). A high-fibre diet is important to maintain the balance of VFAs which should be made of predominantly acetates followed by butyrates and then propionates. Decreases in fibre levels increase butyrates and propionates at the expense of acetates, which results in reduction in the normal peristalsis of the gut and leads to hypomotility disorders and ileus or gut stasis.
Liver
The rabbit liver has four lobes. There is a gall bladder, which has an opening separate from the pancreatic duct into the proximal duodenum. The main bile pigment is biliverdin, rather than bilirubin seen in cats and dogs.
Pancreas
The pancreas is a diffuse organ, suspended in the loop of the duodenum. There is one single pancreatic duct, separate from the bile duct, emptying into the proximal duodenum.
Urinary anatomy
Kidney
The kidneys are bean shaped. The right kidney is more cranial than the left, and they are often separated from the ventral lumbar spine by large fat deposits. A single ureter arises from each kidney and traverses across the abdominal cavity to empty into the urinary bladder.
Bladder
The bladder lining is composed of transitional cell epithelium. The urethra in the male rabbit exits through the pelvis and out through the penis. In females, the urethra opens onto the floor of the vagina.
Renal physiology
Rabbit’s urine is alkaline with a pH varying between 6.5 and 8, but it will become acidic if the rabbit has been anorectic for 24 hours or more. The urine contains varying amounts of calcium carbonate. This is because it has no ability to alter how much calcium is absorbed from the gut, and so any excess calcium must be excreted by the kidneys into the urine. This can be seen as a tan-coloured silt. Porphyrin pigments may also be seen in rabbit’s urine. These are plant pigments and make the urine appear anywhere from a dark yellow to a deep wine-red in colour. This may mimic haematuria; therefore, to diagnose blood in the urine, it is necessary to examine it microscopically.
Cardiovascular system
Heart
The rabbit heart is small in relation to body size. The right atrioventricular valve has only two cusps instead of three. The pulmonary artery also has a large amount of smooth muscle in its wall which can contract vigorously during anaphylactic shock, causing immediate right-sided cardiac overload and failure.
Blood vessels for sampling
Vascular access in rabbits includes
Lateral ear vein
This runs along the lateral margin of either ear. It may be accessed using a 25 or 27 gauge needle or catheter and used for slow intravenous injections and blood sampling.
Cephalic vein
This runs in a similar position to that seen in cats and dogs. It may be split into two in some individuals, but may be used for intravenous fluids and sampling (Figure 1.3).
Saphenous vein
This runs across the lateral aspect of the hock, as in cats and dogs, and may also be used for venipuncture.
Jugular vein
The jugular veins are prominent in the rabbit but they form the major part of the drainage of blood from the orbit of the eye. If a haematoma or thrombus forms and blocks the lumen of a jugular vein, severe orbital oedema may occur, with possible damaging effects.
Lymphatic system
Spleen
The spleen is a flattened structure, oblong in nature and attached to the greater curvature of the stomach, and is thus found predominantly on the left side.
Thymus
The thymus is a large structure in the cranial thoracic compartment even in the adult rabbit. It provides the body with the T-cell lymphocytes.
Lymph nodes
The root of the mesentery supporting the digestive tract is well supplied with lymph nodes, as is the hilar area of the lungs where the two main bronchi diverge to supply each lung. In addition, there are superficial lymph nodes in the popliteal, prescapular and submandibular areas.
Reproductive anatomy
Male
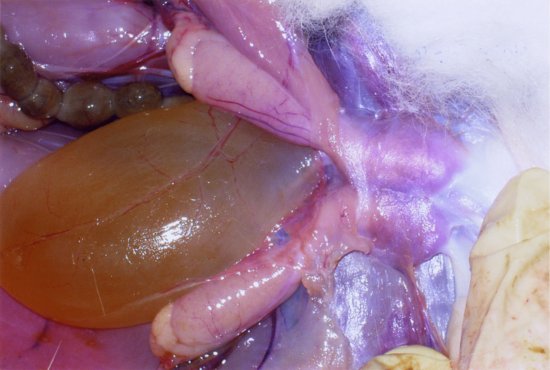
The paired testes can move from an inguinal position within the thin-skinned scrotal sacs, to an intra-abdominal position through the open inguinal canal (see Figure 1.4). The scrotal sacs are sparsely haired and lie on either side of the anogenital area.
Figure 1.4 Close-up of the caudal abdomen of a male rabbit showing the retracted testes and the inguinal canals and the full urinary bladder. (Fraser and Girling, 2009)
The accessory sex glands in the buck attached to the urethra in the caudal abdomen are: dorsal and smaller ventral prostate; bilobed vesicular gland; bilobed coagulating gland and a bilobed bulbourethral gland. The prepuce has numerous small preputial glands in the dermis, and there are a couple of inguinal glands situated on either side of the penis which secrete a brown-coloured sebum clearly seen adjacent to the anus.
Female
The ovaries are supported by the ovarian ligament and lie caudal to each respective kidney. The ovarian artery often splits into two parts after leaving the aorta, and it, along with the rest of the reproductive tract, is frequently encased in large amounts of fat.
The uterus is duplex – there is no common uterine body. Instead there are two separate uteri with separate cervices emptying into the vagina. The vagina is large and thin walled, with the urethra opening onto its floor cranial to the pelvis. The vulva therefore is a common opening for the reproductive and urinary systems unlike many rodents. It lies just cranial to the anus and is flanked on either side by the inguinal glands, as with the buck.
The doe has on average four pairs of mammary glands extending from the inguinal region to the axillary areas.
Reproductive physiology
Male
The buck rabbit has the same sexual hormones as in cats and dogs, but they are on a seasonal time clock triggered by the lengthening daylight of spring. This is mediated through the pineal gland in the brain which has neural links from the eyes and controls the hormone melatonin. It in turn controls the pituitary release of follicle-stimulating and luteinising hormones which then act upon the testes.
Female
Does are induced ovulators. Waves of follicles swell and regress during the course of the season, starting to increase in activity in early spring. If not mated, these follicles will often dominate the cycle for 12–16 days at a time. There is no real anoestrus phase in does; instead, a slight waning in activity for 1–2 days occurs before a return to heat. During peak sexual activity, the vulva is often deeply congested and almost purple in colour and considerably enlarged.
Once mated, the male’s semen may form a copulatory plug, which is a gelatinous accumulation of sperm which drops out of the doe’s vagina 4–6 hours post-mating. Gestation lasts from 29 to 35 days, with the foetus forming a haemochorial placenta (where the outer chorion layer of the foetal placental membrane burrows into the lining of the uterus so that it directly attaches to the blood in the intrauterine vessels) at about day 13. This is a common time for abortions to occur. A pregnant doe will remove fur from her ventrum to line the nest in the latter few days prior to parturition.
Dystocia is uncommon. The doe only nurses the kittens once a day for 20 minutes or so, often in the early morning. It is therefore not uncommon for owners to think that the doe is neglecting her young as she will often spend the rest of the time eating and away from the litter.
Pseudopregnancy often occurs after an unsuccessful mating or mounting activity by another buck or doe. A corpus luteum forms and this lasts for 15–17 days during which time the doe may produce milk and build a nest. At this time, the doe is susceptible to mastitis.
Neonatology
The young kits or kittens are altricial in nature, that is they are totally dependent on the mother for nutrition and survival for the first few weeks of life. They are born blind, deaf and furless. Fur growth appears around day 5–6, the eyes open at day 8–10 and the ears at 11–12. Weaning occurs around 6 weeks of age, with the young taking solid food from 2 to 3 weeks.
Sexing
The young may be sexed from 4 to 5 weeks of age. Gentle pressure is placed on either side of the reproductive or anal area to protrude the vulva or penis. The vulva of the young doe is rounded and has a central slit in midline and projects cranially. The penis of the young buck is more conical and pointed, with no central slit and tends to project caudally when protruded. Once the buck is older, the testes descend into the scrotum.
Skin
Lop breeds, particularly does, have extra skin folds called ‘dewlaps’ around the ventral neck region. In addition, such extra folds of skin may be found around the anogenital area, leading to increased risk of urine and faecal soiling.
Rabbits do not have keratinised footpads. Instead they have thick fur covering the areas of the toes and metatarsals which are pressed flat to the ground.
In addition to the para-anal scent glands mentioned above, there are a series of discrete submandibular chin glands. These are used to mark territory and also, in the case of does, to mark their young to distinguish them from others.
The rabbit has no skin sweat glands except a few along the margins of the lips. This means that they are very prone to heat stress at temperatures greater than 28°C.
The presence of many vibrissae or sensitive hairs around the lips and chin are important since rabbits cannot see anything immediately below their mouths, and so rely on touch to manipulate food towards the mouth.
Eyes
Rabbits have prominent eyes, which allow a near 360° field of vision. There is a prominent third eyelid, which moves from the medial canthus of the eye and possesses a large amount of reactive lymphoid tissue within its structure and a Harderian tear gland at its base. This is often enlarged in the buck during the breeding season and possesses two lobes in both the sexes.
Haematology
The most notable feature is the staining of the rabbit neutrophil, which resembles the cat or dog eosinophil, and so is often known as the pseudoeosinophil. Many rabbits have more lymphocytes than pseudoeosinophils, resembling other mammals such as cattle, rather than cats and dogs, in which the neutrophil is the commonest white blood cell.
RAT AND MOUSE
Biological average values for the rat and mouse
The normal biological values for the rat and mouse are given in Table 1.3.
Table 1.3 Biological parameters for the rat and mouse.
| Biological parameter | Average range rat | Average range mouse |
| Weight (g) | 400–1000 | 25–50 |
| Rectal body temperature (°C) | 37.6–38.6 | 37–38 |
| Respiration rate at rest (breaths per minute) | 60–140 | 100–280 |
| Heart rate at rest (beats per minute) | 250–450 | 500–600 |
| Gestation length (days) | 20–22 | 19–21 |
| Litter size | 6–16 | 8–12 |
| Age sexual maturity (weeks) | ||
| Male | 8 | 6 |
| Female | 10 | 7 |
| Oestrus interval (days) | 4–5 | 4–5 |
| Lifespan (years) | 3–4 | 2–3 |
Musculoskeletal system
Skull
The skull of both species is elongated. The eyes are laterally situated, and there is a long snout and a shallow cranium. The maxilla is narrower than the mandible. The temporomandibular joint is elongated craniocaudally, allowing the mandible to move rostrally and caudally in relation to the maxilla. This allows the incisors to be engaged for gnawing, while the molars are disengaged. Alternatively, the molars may be engaged for mastication prior to swallowing, while the incisors are disengaged. The two procedures cannot occur at the same time. The rostral symphysis, joining each half of the mandible, is also articulated allowing movement of each hemimandible independently of the other.
Axial skeleton
The pelvis of the female mouse is joined at the pubis and ischial areas midline by fibrous tissue. This allows separation of the pelvis during parturition in the mouse. There are no fibrous areas to the pelvis of the female rat and consequently no pelvic separation occurs.
Appendicular skeleton
The scapula articulates at its coracoid process with the clavicles as well as the humerus. There are four metacarpal bones in the rat, with four digits. Occasionally the vestigial remnant of digit 1 is present.
The hindlimbs have a strong laterally bowed fibula in both species. The tibia articulates with five metatarsal bones at the hock joint. Consequently there are five digits in the hindlimbs of these species. Rats and mice are plantigrade in their stance, that is they walk with the whole of the metatarsal bone area flat to the ground.
Male rats may still have open growth plates in many of their long bones well into the second year of their lives, whereas mice close their growth plates in the first 3–4 months of life.
Respiratory system
The nares of both species are prominent and surrounded by an area of hairless skin containing some sweat glands.
There is a vomeronasal organ in the floor of the nasal passages, accessed via two small stoma in the roof of the mouth just caudal to the maxillary incisors. This organ is responsible for detecting pheromones secreted by other individuals.
The right lung of the rat is divided into three distinct lobes, whereas the left lung is undivided. The chest cavity itself is smaller in proportion to the abdominal cavity than is the case in cats and dogs, meaning that rats and mice have little respiratory reserve.
Digestive system
Oral cavity
The lips of mice, and particularly rats, are deeply divided exposing the upper incisors, with large areas of loose folds of skin forming the cheeks.
Both species have pigmented yellow – orange enamel coating the labial aspect of the incisors. The maxillary incisors are one-third to one-quarter of the length of the mandibular incisors. There is a chisel shape to their occlusal surfaces due to the absence of enamel on the lingual aspect of the incisors making them wear quicker on this side. The mandibular incisors are also mobile and loosely rooted in the lower jaw. Their dental formula is
I 1/1 C 0/0 Pm 0/0 M 3/3.
There is no evidence of any deciduous or ‘milk’ teeth being present in either species. The molars grow extremely slowly.
Both species have a diastema. In the case of rats, this gap is particularly noticeable and large enough to allow them to draw their cheeks into the gap to effectively close off the back of the mouth. This enables them to gnaw, without consuming the material they are nibbling.
The tongue is relatively mobile and its surface is covered with small, backward-pointing papillae.
Stomach
The stomach of the mouse and rat is elongated and narrow. In the rat, in particular, the stomach is divided into two regions. The most cranial is known as the proventricular region and is covered by a thin, whitened lining of aglandular mucosa. The oesophagus enters the stomach halfway along the length of its lesser curvature. The caudal area of the stomach is covered by a redder, thicker, glandular mucosa known as the pyloric region.
Small intestine
The small intestine comprises the largest portion of the gastrointestinal tract. The bile duct enters the first part of the duodenum direct from the liver in the rat, which has no gall bladder. The mouse does have a gall bladder which empties into the first part of the duodenum.
Large intestine
The ileum enters the large intestine at the junction of the caecum and the large intestine on the left side of the abdomen. The caecum is a medium-sized organ in the mouse and rat, reflecting their omnivorous nature, and forms a blind-ended pouch which is flexed back on itself.
Liver
The liver is divided in both species into four lobes. There is a gall bladder present in the mouse but not in the rat. The liver sits cranial to the stomach. Biliverdin is the prominent bile pigment in rats and mice.
Pancreas
The pancreas lies along the proximal aspect of the duodenal loop. In both species, it empties through a series of ducts into the bile duct. Its function appears to be the same as in cats and dogs, producing both insulin and glucagon for glucose homeostasis and the digestive enzymes – amylase, lipase and trypsinogen.
Urinary system
Kidney
The kidneys are bean shaped. The right kidney sits in a depression in the right lobe of the liver, and the left kidney is slightly more caudal. Each empties through its ureter which enters the bladder at the trigone area.
Bladder
The bladder is lined with transitional epithelium and empties through the urethra. In the female mouse and rat, the urethra empties through a separate urinary papilla rather than onto the floor of the vagina as it does with higher mammals. The female mouse and rat therefore have three orifices caudoventrally: the anus most caudally, the reproductive tract entrance next cranially and the urinary papilla the most cranial of the three.
Cardiovascular system
Heart
The heart of the mouse and rat has four chambers, as in other mammals. As with rabbits, the chest compartment is relatively small in comparison to the abdomen, and the heart therefore appears relatively large in relation to the rest of the chest. The heart occupies the fourth to sixth rib spaces.
Blood vessels for sampling
Useful vessels from which to sample blood are the lateral tail veins. These are best accessed after first warming the tail, or lightly sedating the mouse or rat to allow dilation of the vessels. A 25–27 gauge needle or butterfly catheter is required. Some mild pressure at the tail base allows further dilation.
In the rat, the femoral vein may also be used for sampling. This is found on the medial aspect of the thigh, close to its junction with the inguinal area, just caudal to the femur. This vessel is best used only under anaesthetic due to the difficulty of accessing it in a conscious rat.
For small capillary samples, a microcapillary tube may be gently pushed into the medial canthus of the eye socket in the anaesthetised rat or mouse. This collects blood from the orbital sinus.
Lymphatic system
Spleen
The spleen of male mice is often twice the size of that in females. In both species, it is a strap-like organ sitting along the greater curvature of the stomach.
Thymus
The thymus is an obvious organ in the cranial chest, and may be split into several smaller islands of tissue. It is frequently present in the adult rat or mouse.
Lymph nodes
The lymph nodes follow similar patterns to those seen in the rabbit. The mesenteric lymph nodes can become very prominent in certain bacterial infections.
Reproductive anatomy
Male
The male rat and mouse reproductive systems are nearly identical in design.
The testes are large and can move between the abdomen and the scrotal sacs, although somewhat inhibited by a large fat body attached to the tail of each testicle extending through the open inguinal canal. Each testis descends into the scrotum around the fifth week of age in the rat and the third to fourth week in the mouse.
The vasa deferentia are joined by the opening of the small ampullary glands which open into a swelling of the vas deferens known as the ampulla just before they join the urethra. Other accessory sex glands, the vesicular glands, the coagulating glands (which are joined together) and the two parts of the prostate (the ventral and dorsal lobes), open into the urethra itself. As the urethra exits the pelvic canal, a paired bulbourethral gland empties into its lumen.
These accessory glands produce nutrients and supporting fluids for the spermatozoa. In addition, the coagulating glands are responsible for allowing a plug of sperm to form in the female’s vagina immediately after mating. The penis has an os penis in both species. There is a preputial gland in the small prepuce, which is used for territorial marking. Male mice and rats have no nipples.
Female
The rat uterus has two separate uterine horns which come together at two separate cervices. From the outside these appear to merge to form a common uterine body, and so it is referred to as bicornuate in nature. The vagina itself has no lumen in the immature rat. Instead, at puberty, the solid mass of tissue forms its own lumen, breaking through to the surface at the time of the first ovulation.
The mouse uterus is almost exactly the same except that the two separate uterine horns do fuse just before the cervix, making it truly bicornuate and so there is just the one cervical opening into the vagina. The vagina is also non-patent in the immature state.
Mammary tissue is extensive in both female rats and mice. In mice there are normally five pairs of mammary glands, three in the axillary region, with mammary tissue extending dorsally nearly to midline! The other two pairs of glands are in the inguinal region, with mammary tissue extending around the anus and tail base. In rats there are more commonly six pairs of mammary glands. Three are located in the axillary region, again with some tissue moving onto the lateral chest wall. The other three glands are inguinally located.
Reproductive physiology
The female rat is non-seasonally polyoestrus. The commonest time for heat to occur is during the night. The first cycling activity occurs around 8 weeks of age in the female rat, with the cycle lasting 4–5 days in total. Ovulation is spontaneous and occurs towards the end of the 12-hour-long heat. There is a reduction in reproductive activity in the female rat over 18 months of age.
During mating, the semen deposited in the female rat’s vagina forms a copulatory plug which sits in the cranial vagina, blocking both cervices. This dries and falls out within a few hours of mating, but seems to play an important role in the success of mating. It is often eaten rapidly after being passed.
Gestation length is around 21 days. The placentation of the rat and mouse is discoidal, that is the area of attachment is disc-like with the chorion of the placenta in contact with the blood stream of the dam’s uterus. There may be a bloody mucous discharge from the vagina around 14 days, which is normal. This stops within 2–3 days. Mammary development occurs at around days 12–14 and at that stage the foetuses may be palpated.
Parturition is rarely complicated. There is no separation of the pelvis in the female rat, although the female mouse’s pelvis does separate at the ischial and pubic sutures. Parturition occurs in the afternoon and is followed by a post-partum oestrus.
Pseudopregnancy is seen in both rats and mice. During this time the female may build a nest; there may be some mammary development and no signs of a heat for up to 2 weeks.
There are a couple of important physiological reproductive phenomena in mice and rats. One of these is the Whitten effect. This is when a group of anoestrus females will all come into heat spontaneously some 72 hours after being exposed to the pheromones of a male. This has beneficial effects when it comes to successful rapid breeding. The other is the Bruce effect. This is when a female in the early stages of gestation will reabsorb the embryos and come back into heat when presented with a new male. By preferentially allowing successful mating with a new male, this is thought to have a beneficial effect on genetic diversity.
Neonatology
Rat and mouse pups are altricial. They are born blind, deaf and hairless. The ear canals open around days 4–5 and the eyes at around 2 weeks of age. The first few hairs are also seen in the first week of life. The pups are born without teeth, the incisors becoming visible at 1–2 weeks of age with the molars developing later.
The female rat and mouse are prone to cannibalism if disturbed with their young in the first few weeks after parturition. It is therefore important to leave the female rat and mouse alone during this period, only disturbing them to replenish food and clear the worst of any cage soiling.
Sexing
Sexing may be done from 4 to 6 weeks of age. In males the urinary papilla is slightly larger than the female and further away from the anus. It may be possible in the sexually mature female to see the small reproductive tract entrance as a transverse slit in between the anus and urinary papilla. Also, in male mice and rats, no nipples are visible. In mature males, if the rat or mouse is gently suspended in a vertical position with the head uppermost, the testes will often descend into the scrotal sacs and are then obvious.
Skin
Rats and mice possess no generalised sweat glands and so are prone to heat stress at temperatures above 26–28°C. There are some sweat glands present on the soles of the feet as well as the nares.
There is a layer of brown fat between the shoulder blades dorsally; its function is not clearly known, but it decreases with age and may play a role in thermoregulation.
The tails of rats and mice are relatively hairless. As rats age, there is an increasing number of coarse skin scales present on the tail surface making blood sampling difficult. Rats should not be grasped by the tip of the tail as the skin may slough in this region.
White fur will often yellow in rats as they age, and most rats will show evidence of a yellow hue to the skin on the back with time.
The vibrissae around the lips and nose are important for detecting vibrations and determining where food is due to their inability, as with rabbits, to see food immediately below their mouths.
Eyes
Rats and mice have a prominent set of small eyes located laterally. The albino breeds lack pigment in their irises or retinas, and so their eyes appear pink red.
Haematology
The haematological parameters are similar to those seen in cats or dogs, except that the lymphocyte, as opposed to the neutrophil, is the most common white blood cell.
GERBIL AND HAMSTER
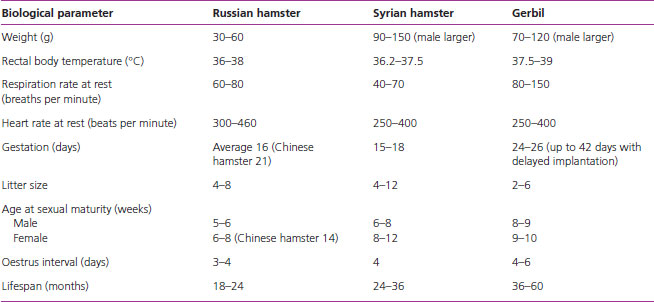
Biological average values for the gerbil and hamster
Table 1.4 gives the average normal values for the basic biological values for gerbils and hamsters.
Table 1.4 Biological parameters for the gerbil and hamster.
Musculoskeletal system
Skull
The skull of the gerbil is not dissimilar to that of the rat or mouse; in the hamster, the skull is shortened, particularly in the Russian and Chinese hamster subspecies.
Axial skeleton
The axial skeleton is much the same as for the rat and mouse, except the hamster has much fewer coccygeal vertebrae (only seven or so).
Appendicular skeleton
Gerbils have a longer femur and tibial length, giving them longer hindlimbs equipped for jumping. Their normal stance is bipedal, standing erect on their hindlimbs. Hamsters are a much shorter-legged creature, stockier in build and walk predominantly on all fours.
The forelimbs have four digits, and the hindlimbs have five in both species.
Respiratory system
As with rats and mice, the chest cavity is small in relation to the abdomen, but the situation is not so pronounced as that seen in rats.
Digestive system
Oral cavity
The incisors in both species are continuously erupting (open rooted), and both species have orange pigmentation of the enamel surfaces. The dental formula is
I 1/1 C 0/0 Pm 0/0 M 3/3.
Hamsters are born with the incisors fully erupted, and use them to grasp the nipples of the female enabling them to suck effectively.
The molars do grow continually, but at a very slow rate. In addition, there appears to be no evidence of deciduous teeth. Both species possess a diastema. The mandible is generally wider than the maxilla.
The cheek pouches of the hamster are its most distinguishing feature. These are not present at birth, but rather develop during the second week of life from a solid cord of cells which disintegrate, creating the cavities. The entrances to the cheek pouches open into the diastema. Each cheek pouch extends caudal to the respective ear. They are lined with stratified squamous epithelium, and have a reduced local immune system and lymphatic function. This can be a problem if the cheek pouch becomes infected.
Stomach
The stomach of the hamster has two separate areas. The oesophagus enters the proximal portion. This portion is non-glandular and has a bacterial population that allows limited microbial breakdown of food. It is sharply divided by a deep groove from the distal area of the stomach, which is glandular, with a redder lining composed of the acid- and pepsinogen-secreting cells that start the process of enzymatic digestion.
The gerbil has two areas to the stomach but they are less clearly demarcated, and the proximal portion does not support a significant microbial population.
Small intestine
The hamster’s small intestine is extremely long, being three to four times its own body length! The gerbil has a similar layout to the mouse.
Large intestine
In the hamster the caecum is a sacculated and enlarged organ sitting in the ventral left portion of the abdomen at the ileocaecal junction. It has fine divisions within it, which may function to increase its surface area and aid fibre fermentation. The gerbil has a similar layout to the mouse.
Liver
The liver in both species is divided into four lobes. In both hamsters and gerbils, a gall bladder is present. A bile duct empties into the duodenum accompanied by the pancreatic duct.
Pancreas
The pancreas is found adjacent to the descending duodenum. It has a similar structure and function to that seen in the rat and mouse.
Urinary system
Kidney
The kidneys are similar to the rat and mouse kidney. The gerbil is very good at concentrating its urine, being a desert-dwelling species, and to do this it has very long loops of Henle which contain a countercurrent multiplier system. In the hamster, the renal papilla is particularly long and protrudes from each kidney into its ureter.
Bladder
The bladder of gerbils and hamsters is essentially the same as that seen in rats and mice.
Cardiovascular system
Heart
The heart is similar in form to the rat and mouse heart.
Blood vessels for sampling
The hamster has very few accessible external vessels for blood sampling. This is principally due to its much reduced tail length, which provides the main vascular access in the rat and mouse. The gerbil’s tail breaks off easily, making its use for blood sampling very restricted. Vessels used therefore include the jugular veins and the femoral veins, both of which require the hamster or gerbil to be sedated or anaesthetised. Capillary samples may be taken from the orbital sinus as described in the rat and mouse.
Lymphatic system
Spleen
The structure and position of the spleen is much the same for both species as that seen in the rat.
Thymus
The thymus is again a prominent organ in the cranial chest and often persists in the adult. It provides the T-cell lymphocytes.
Lymph nodes
The presence of lymphatic tissue is the same as that seen in the mouse and rat.
Reproductive anatomy
Male
The male hamster has a smaller fat body attached to the testicle than has the rat. The testes are freely moveable between the abdominal cavity and the scrotal sacs. A small os penis is present in the penile structure.
The male gerbil is similar to the male rat and mouse. The main difference is the slightly smaller size of the testes in relation to the overall body size and the presence of a pigmented scrotum.
Female
The hamster uterus is bicornuate. It has two separate cervices opening into a common vagina, although the uterine horns appear to join externally to produce a common uterine body. The vagina is, as with the rat, not patent at birth. It opens after the tenth day of life, rather than at puberty as in the rat.
The gerbil reproductive tract is similar to that of the mouse. The main difference is that, while there is only one cervical opening into the vagina, the division between the left and right uterine lumens persists to within a few millimetres of this single cervical orifice.
The female hamster has six to seven pairs of mammary glands stretching in a continuous band from the axillary region to the inguinal and perianal region.
The female gerbil has four pairs of mammary glands. Two pairs are found in the axillary region and two pairs in the inguinal region.
Reproductive physiology
The female hamster is seasonally polyoestrus with cycling and fertility dropping off during the winter period. The reproductive cycle is short, lasting 4 days. The female hamster develops a creamy white vaginal discharge around the first day following oestrus. This may be mistaken for a pathological discharge as it has an odour. Ovulation is spontaneous and generally occurs overnight. Phantom pregnancy does occur in the hamster, postponing oestrus for 7–13 days. Gestation itself lasts for 15–18 days in the Syrian hamster, an average of 21 days in the Chinese hamster and an average of 16 days in the Russian hamster. Successful mating is followed by the presence of a copulatory plug of coagulated semen 24 hours later. Pregnancy can be confirmed by failure to produce the copious white discharge 5 days after mating, and an increase in weight at around day 10. There is no evidence of pelvic separation at parturition. There is reduced fertility in the female hamster after 1 year of life.
Gerbils form a monogamous pair, that is they pair for life. The female gerbil is seasonally polyoestrus and a spontaneous ovulator. The oestrus cycle lasts for 4–6 days. Oestrus lasts for 24 hours, and may occur within 14–20 hours of parturition. Gestation lasts an average of 26 days, but may take up to 42 days if mating has occurred at the post-partum heat as when the female is still feeding the young, the fertilised ova will not implant, so prolonging the interval from mating to parturition.
Neonatology
The young hamster is altricial. The pale pink colour of the skin is replaced by some darker pigmentation after the first 2–3 days, with the eyes opening at 2 weeks of age. Weaning occurs around 3–4 weeks of age, with the female hamster becoming sexually mature at 6–8 weeks (up to 14 weeks for the Chinese hamster) and the male at 8–9 weeks.
The young gerbil is also altricial. The skin is a pale pink at birth but darkens by the end of the first week with the appearance of the first few hairs. The teeth erupt in the first few days of life. The eyes open at 2 weeks of age and the ears around days 4–5. Weaning occurs at 3–4 weeks of age. The female gerbil becomes sexually mature at 9–10 weeks of age when the vaginal opening becomes patent. The male gerbil becomes sexually mature at 8–9 weeks of age, with the testes descending into the scrotal sac at 5 weeks.
It is inadvisable to disturb the female hamster with her young as cannibalism can occur. However, a common protective action of the female is to place the young into her cheek pouches to move them, and this may make it look as if she is ‘eating’ the young. Gerbils are less prone to abandoning or abusing their young if disturbed.
Sexing
This may be performed from 4 weeks of age. In the immature gerbil and hamster, the differences are determined by anogenital distances as with the rat and mouse. In the sexually mature hamster, the male has a pointed outline to its rear, owing to the descended testes, whereas the female has a more rounded appearance. In both, it is relatively easy to determine the sex once mature if the individual is supported in a vertical position with the head uppermost. In this position, the testes will descend into the scrotal sacs where they are clearly visible!
Skin
Hamster and gerbil skin has no sweat glands. Gerbils, however, can tolerate wider temperature ranges, up to 29–30°C, although if the humidity levels increase above 50% they will rapidly suffer from heat exhaustion.
In hamsters, there is a pair of oval, raised, flank scent glands situated on each flank cranial to the thigh region. In the mature adult, particularly the male, they may become darkly pigmented. The secretions of these glands may matt the sparsely covered fur, increasing their prominence.
In gerbils, there is a large ventral sebaceous scent gland in the region of the umbilical scar. This is devoid of fur, secretes a yellow sebaceous fluid and is more prominent in males. This is a predilection site for the development of adenocarcinoma in adults. The tail of gerbils is fully furred, but has a series of fracture planes allowing a degloving injury if a gerbil is grasped by the tail. The soft tissue structure never regrows, and the denuded vertebrae will die off leaving a stump.
Haematology
The lifespan of the gerbil erythrocyte is short, lasting only 10 days. This is why so many gerbil red cells show degenerative basophilic speckling when stained with Romanowsky stains. Gerbil blood is often lipaemic, and this has been blamed on their high-fat (sunflower seed) diet. In addition, the blood parameters vary depending on the sex. The male gerbil has a higher packed cell volume, white blood cell and lymphocyte count than the female. Hamster haematology is similar to that seen in mice.
GUINEA PIG AND CHINCHILLA
Biological average values for the guinea pig and chinchilla
The average normal values for guinea pigs and chinchillas are given in Table 1.5.
Table 1.5 Biological parameters for the guinea pig and chinchilla.
| Biological parameter | Guinea pig | Chinchilla |
| Weight (g) | 600–1200 | 400–550 |
| Rectal body temperature (°C) | 37.2–39.5 | 37.8–39.2 |
| Respiration rate at rest (breaths per minute) | 60–140 | 50–60 |
| Heart rate at rest (beats per minute) | 100–180 | 120–160 |
| Gestation length (days) | 59–72 (average 63) | 111 |
| Litter size | 1–6 (average 3) | 1–5 (average 2) |
| Age at sexual maturity (months) | ||
| Male | 2–3 | 6–7 |
| Female | 1.5–2 | 8–9 |
| Oestrus interval (days) | 16 | 30–50 |
| Lifespan (years) | 3–8 | 6–10 |
Musculoskeletal system
Guinea pig
Skull
The skull is rodent shaped, with an elongated nose, low forehead and widely spaced eyes. There are moderately large tympanic bullae which house the middle ear and are clearly visible on radiographs.
Axial skeleton
The vertebral structure is the same as that seen in the rat and mouse, except the number of coccygeal vertebrae is much reduced at 4–6 and they are less mobile. There are 13 ribs; the last two are more cartilaginous than mineralised. Guinea pigs also possess clavicles.
The pelvis of the female is joined at the pubis and ischium by a fibrocartilaginous suture line, allowing separation of the pelvis prior to and during parturition. If the female guinea pig has not had a litter by the time she has reached 1 year of age, this suture line mineralises and prevents future separation. Female guinea pigs not mated before 1 year should therefore not be mated for the rest of their life as dystocia problems are common.
Appendicular skeleton
The forelimbs and hindlimbs are relatively long in comparison to the rat and mouse, but the same bone formulas exist. The main difference is that the guinea pig has four digits on each forelimb and only three digits on each hindlimb.
Chinchilla
Skull
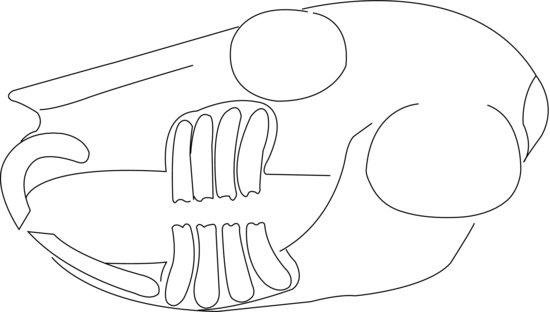
The bones of the skull are more domed than the guinea pig, although still distinctly rodent-like (Figure 1.5). Like the guinea pig, the chinchilla has very large tympanic bullae which are clearly visible as coiled, snail-shell-like features on radiographs.
Figure 1.5 Lateral diagram of the skull of a normal chinchilla showing the relation of tooth roots to the orbit and jawbones. Note how close the roots of the third and fourth cheek teeth are to the inner aspect of the eye; hence, root elongation often causes watering of the eyes.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree



