Chapter 5 Respiratory disorders
Introduction
Histophilus somni is of major importance as a cause of suppurative pneumonia (9.29), but, having effects on several organ systems, it is presented as infectious thromboembolic meningoencephalitis in Chapter 9.
Chapter 5 is divided into infectious (viral, bacterial, and other agents) and noninfectious (allergic, iatrogenic, circulatory, and physiological) etiology. Where appropriate, cross-reference is made to other sections for lesions affecting other systems, e.g., both calf diphtheria and laryngeal abscessation (2.42–2.46) are shown in the neonatal chapter, even though they sometimes occur in older animals.
Infectious disorders
Infectious bovine rhinotracheitis (IBR) (“rednose”)
Clinical features
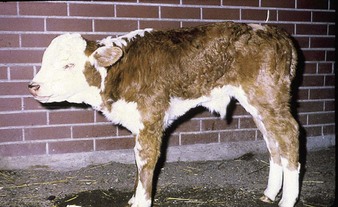
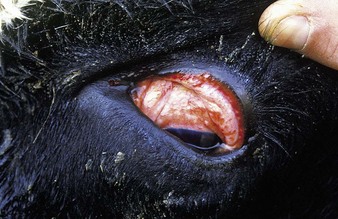
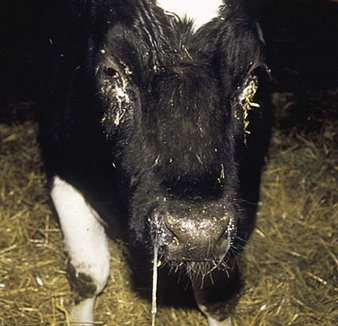
the common respiratory form of IBR has major clinical signs involving the nostrils (hence the alternative name of “rednose”) and the eyes. Feedlot cattle are at particular risk. Within a group of young cattle, several individuals may be affected simultaneously with epiphora and depression. Severely affected animals, such as the crossbred neonate in 5.1, are dull, somnolent, anorexic with a tucked-up belly, and have a mucopurulent nasal discharge, nasal mucosal congestion and lymphadenopathy, and sometimes a harsh cough. The palpebral conjunctivae may be intensely injected or congested in the acute stage (5.2). Characteristic, small, raised, red plaques are visible near the lateral canthus. Secondary infection may lead to a purulent oculonasal discharge (5.3) as well as a typical purulent IBR conjunctivitis, without blepharospasm.
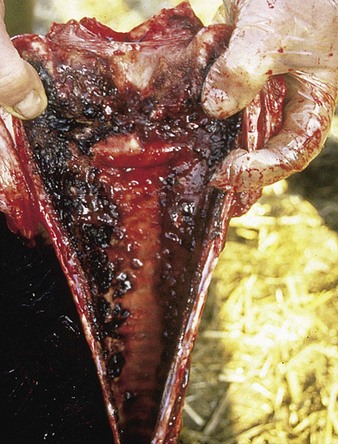
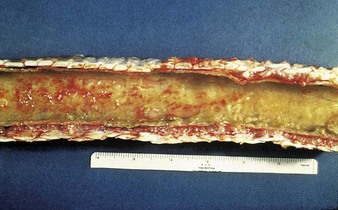
Autopsy examination of this animal (5.4) reveals a severe necrotizing and hemorrhagic laryngotracheitis. Another severe case is shown in 5.5. In severe cases the nasal septum (5.6) sloughs its necrotic mucosa. Epistaxis may follow the rupture of mucosal vessels.
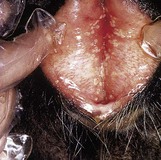
Balanoposthitis can occur with bovine herpesvirus 1 infection (p. 182). In 5.7, the separated vulval lips reveal the multiple, discrete pustules of infectious pustular vulvovaginitis (IPVV). The similarity of the male and female lesions is obvious (compare 10.29).
Pasteurellosis (“shipping fever”, “transit fever”)
Clinical features
severe respiratory distress (5.8), with the head and neck extended, open-mouth breathing, and froth on the lips, is obvious in this calf, which died an hour after the photograph was taken. Severe respiratory signs were evident, with dullness and anorexia, pyrexia, and a moist cough. The cranioventral lung fields reveal wheezing sounds on auscultation. An expiratory grunt is possible. Another tucked-up beef steer (5.9) shows severe dyspnea as open-mouth breathing.
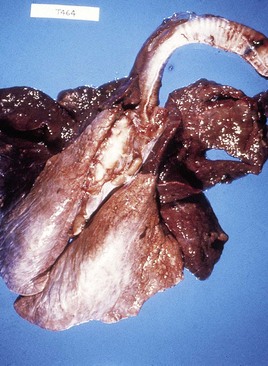
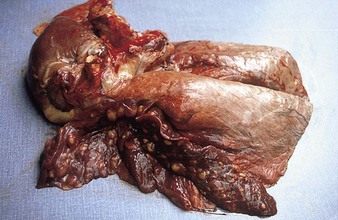
At autopsy examination of another calf (5.10), in addition to froth in the major bronchi, the apical and cardiac lobes are typically dark red, slightly swollen, firm, and contain microabscesses. The diaphragmatic lobes are normal. Such lungs may have fibrin deposits on the pleural surface. Lung changes tend to be symmetrical. In 5.11 the pneumonic areas of the apical and cardiac lobes show scattered, pale yellow abscesses. (See also H. somni, 9.29.)
< div class='tao-gold-member'>
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree