Reproductive Anatomy and Physiology in the Male
Anatomy of the Male Reproductive Organs
The male reproductive organs in the llama and alpaca have been well described.1 The scrotum is nonpendulous and situated high in the perineal region at the level of the ischial arch. The testicles are relatively small in comparison with other domestic livestock and directed caudodorsally. The epididymis faces laterally along the dorsal border of the testis, with the head curving around the cranial pole of the testis. Histologic and histochemical studies on llamas have defined six segments of the epididymis. These regional differences represent different secretory functions that may play a role in the process of sperm maturation.2 In the llama, the ductus deferens is 1 millimeter (mm) in diameter at the junction of the epididymis. It widens to 2 mm in the abdominal cavity toward the pelvic urethra (ampulla). The length of the ductus deferens is about 40 centimeters (cm).
The most notable feature in the anatomy of the internal genitalia of camelids is the absence of seminal vesicles. The prostate is usually described as a small H-shaped gland (3 cm × 3 cm × 2 cm) firmly attached to the dorsolateral aspect of the pelvic urethra near the trigone of the bladder. Numerous prostatic ducts direct the secretion of this gland directly into the colliculis seminalis. The two bulbourethral glands are ovoid and located on the dorsolateral aspect of the pelvic urethra, above and just cranial to the ischial arch. They measure approximately 2 cm in diameter in the adult llama and are partly covered by the bulbocavernosus muscle.1
Growth of the Testicle
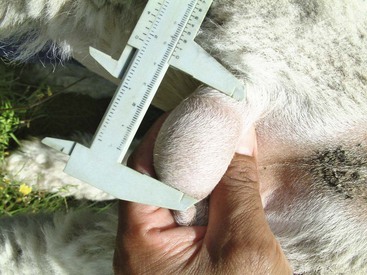
The growth of the testicle in llamas and alpacas appears to be slow. The testicles are in scrotal position at birth, but they are difficult to palpate because of their very small size. By age 6 months, they should be felt within the scrotum. They are soft, and it is almost impossible to distinguish between the testis and the epididymis. By age 12 months, the testicles should be easily palpable within the scrotum, and the tail of the epididymis is readily palpable. Future herd sires may be selected at this age on the basis of testicular size, body conformation, and other characteristics. Thereafter, the testicles should be resilient on palpation and freely movable within the scrotum. Testicular size is often estimated by length and width measurements with a Vernier caliper (Figure 15-1). The average testicular size at different ages in camelids in Peru is presented in Table 15-1 and does not seem to be different from those reported in Australia.3–5
TABLE 15-1
Mean Testicular Size, Testicle Weight and Testosterone Concentrations by Age in 150 Llamas, 320 Alpacas, and 32 Vicunas
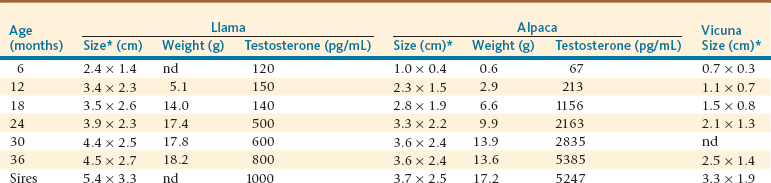
cm, Centimeter; g, gram; nd, not determined; pg/mL, picogram per milliliter.
The histologic architecture of the testis follows the same changes observed in other species during development. Studies on histomorphology of the prepuberal testis in South American camelids (SACs) are scarce. In 7- to 10-month-old guanacos, gonocytes are scarce; spermatogonia are observed in the parietal side of the sexual cords, and the interstitial tissue is loose. By age 18 to 24 months, the seminiferous tubules are fully developed, and all their histologic components are present. Spermatogenesis is evidenced by the presence of meiotic divisions of primary spermatocytes. Sertoli cells are present, and the germinal cells are associated in cell populations. The interstitial tissue is completely occupied by Leydig cells with some capillaries and lymph vessels.6
Spermatogenesis has been described only in the llama.7 Spermatogonia population is present in two categories, A and B. Spermatogonia A have nuclei of different shapes, from spherical to ovoid; by contrast, the nucleus of spermatogonia B is round and smaller than that of spermatogonia A. The cycle of the seminiferous tubule has eight stages. The frequency of each stage in the llama has been described.
The effect of season on spermatogenesis has been described in the male guanaco and vicuña but remain to be studied in the alpaca and llama.6
Puberty
Puberty, defined as presence of spermatozoa in the ejaculate, has not been studied experimentally in SACs. In South America, domestic SAC males are used for breeding when they reach age 3 years. However, in North America, some males are used as sires when they are 2 years old. This suggests that puberty occurs earlier than 2 years. Studies in South America have demonstrated that spermatozoa are present in the vas deferens in 25% of 18-month-old male alpacas. This percentage reaches 100% when male alpacas are 22 months old.8 The presence of spermatozoa in the seminiferous tubules has been reported in 20-month-old alpacas. Anecdotal reports from North American alpaca owners about male alpacas younger than age 22 months successfully impregnating females are common. However, some males may not reach puberty even after age 3 years. In South America, males that have not been “proven” by 3 years of age are culled from reproduction.8,9
Endocrine Function of the Testicle
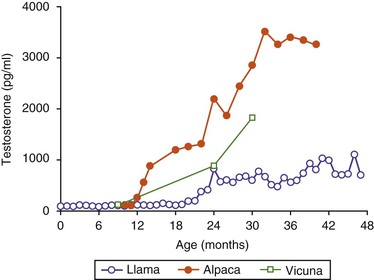
Basal testosterone concentrations in llamas (60–90 picogram per milliliter [pg/mL]) are present in blood from birth to approximately age 20 to 21 months. After age 21 months, testosterone concentrations rise quickly to reach 1000 pg/mL or more (Figure 15-2).3 Alpacas often show higher circulating testosterone levels compared with llamas. Adult testosterone levels are reached at age 2 years in alpacas and vicunas and show seasonal (monthly) variation (Table 15-2). In addition, testosterone concentrations do not vary with the altitude. Testosterone concentrations were 5.9 ± 1.8 nanogram per milliliter (ng/mL) in male alpacas maintained at sea level compared with 4.3 ± 1.2 ng/mL in male alpacas living at high altitude.10–12
TABLE 15-2
Mean and Standard Error of the Mean of Testosterone Concentrations* in Llamas, Alpacas and Vicunas in the Southern Hemisphere and During Different Seasons of the Year

*pg/mL, picogram per milliliter.
1Sumar J: Fisiología de la reproducción del macho y manejo reproductivo. In Fernandez-Baca, editor: Avances y perspectivas del conocimiento de los Camélidos Sud Americanos. Santiago, Chile, 1991, FAO (pp 111-148).
2Urquieta B, et al: Seasonal variation in some reproductive parameters of male vicuna in the High Andes on northern Chile, J Arid Environ 26:79-87, 1994.
Detachment of the Penis–Prepuce Adhesion
At birth, the glans penis cannot be exteriorized because of its adhesion to the prepuce. The process of detachment is gradual and parallels sexual development and testosterone secretion.8 It begins from the tip toward the neck of the glans penis (Figure 15-3). The penis–prepuce detachment is completed in 12% of males at age 1 year. In llamas, by age 14 to 16 months, half of the glans is free, and by age 18 to 21 months, the penis is completely free.3 In alpacas, this process may take a little longer, but by age 36 months, all males have a free glans (Figure 15-4). Detachment of the penis appears to occur earlier in males raised in North America. Suri males seem to be more precocious compared with Huacaya males and are often used as sires as early as 20 months of age.13–17
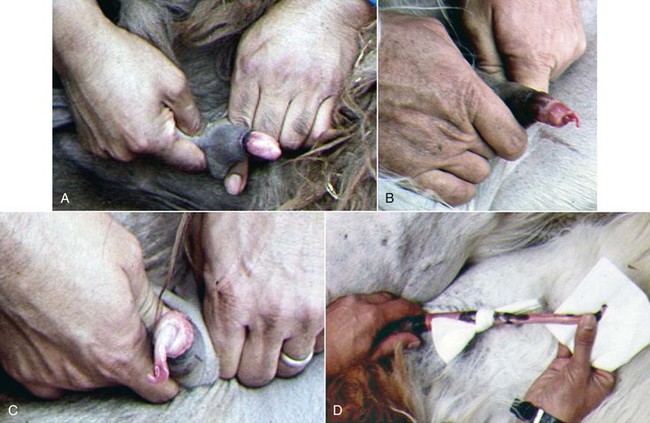
Figure 15-3 Progress of penis–prepuce detachment. A, No detachment. B, Initial detachment. C, Half way detached. D, Penis completely detached.
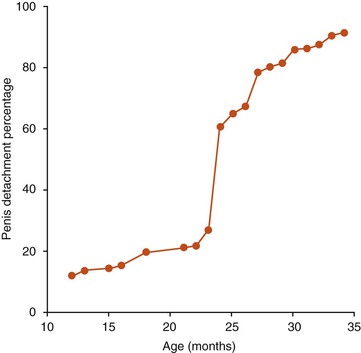
Figure 15-4 Proportion of alpaca males with complete penile–preputial detachment according to age in South America.
Treatment with testosterone (327 mg intramuscularly [IM] given 3 times at 2-week intervals) was effective in causing penis–prepuce detachment within 6 to 9 weeks.18
Mating Behavior and Copulation
Mating behavior varies slightly, depending on whether the male is introduced to a single female or to a group of females. If it is a single female, the male immediately starts chasing her, mounts her, and forces her down. When several females are available, the male randomly chases a female until she cushes if she is receptive. If the female rejects him by kicking, spitting, and running away, the male may persist for a few minutes and then moves on to other females. In a group mating system, several other receptive females may be observed sitting near the mating pair. In South America, at the beginning of the breeding season, up to seven females could be observed in the recumbent position waiting for the male (Figure 15-5). In a multi-sire, free-mating situation, breeding pairs tend to copulate in the same area without interference. However, once a male finishes copulating, he immediately starts challenging other males. To avoid this situation, males are mated in separate pens or are separated from the group as soon as they finish copulating. Males visit the dung pile and urinate following copulation.14,16,19–21
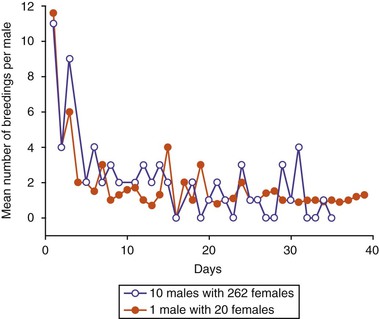
Figure 15-5 Mean number of copulatory periods per male at the beginning of the breeding season in Peru in two mating systems (Group1: 10 males with 200 females; Group 2: a single male with 20 females).
Males may continue to mate successively with any available receptive female in the group. Some males may mate up to 18 times on the first day of the breeding season. At the end of the first day of mating, males may be observed cooling down by sitting in a sternal position in water. The number of matings decreases considerably on the second day of the breeding season. (Figure 15-6). This reduction in breeding activity is maintained throughout the rest of the breeding period, even in presence of receptive females. It has been suggested that this reduction in sexual activity is caused by physical exhaustion. When the first group of males is replaced by another group of fresh males after 7 days, intense mating activity resumes as described above. Physical exhaustion does not, however, fully explain the trend in mating activity as copulatory activity increases considerably when new receptive females are introduced to the herd.
The flehmen response displayed by male llama and alpacas is different from that seen in bulls and rams.22–24 The male sniffs the perineum of the female and raises its head with its month open; however, the upper lips do not curl upward. The head is held almost in a vertical position, and the ears are pined backward. The male raises its head and may shake it rapidly. This is an indication that the vomeronasal organ is also responsive in these animals. The flehmen response is manifested when the male sniffs the female dung pile and may also be displayed when seeking a female and after copulation.
The male probes the female perineal area several times using pelvic movements until the vulvar opening is found and intromission is completed. This may take longer in young inexperienced males than in proven sires. During this phase, the penis becomes rigid, and the male makes semicircular movements of the cartilaginous process of the glans penis. Once the vulva is located and vaginal penetration has occurred, the male gets closer to the female, and positions its hocks parallel to the female’s. When penile intromission is in progress, the back of the male becomes curved and the sacral region becomes vertical to the ground, and very close to the perineum of the female. During copulation, the penis penetrates through the cervix into the uterine horns.20,21,25
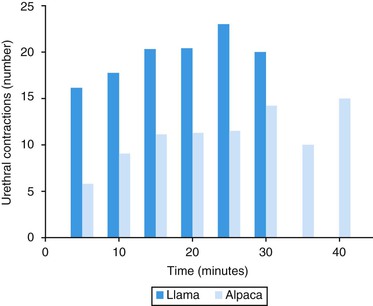
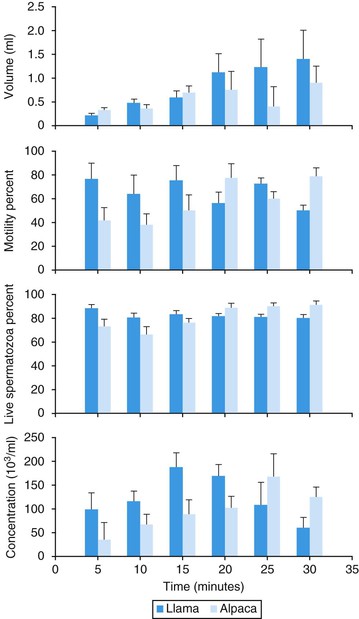
Ejaculation in these species is continuous throughout copulation and is often referred to as “dribble ejaculation.”20 Digital palpation during mating in llamas and alpacas showed that ejaculation corresponds to urethral pulses. During the first 4 minutes of copulation, 11 urethral pulses are identified on average (3.5 per minute) and are not associated with body contractions. These are followed by up to 18.5 clusters of urethral pulses, accompanied by contractions of the male’s body. Each cluster of contractions is preceded by a new repositioning of the penis into the female’s reproductive tract (Figure 15-7). A male llama ejaculates 18 to 19 times during the 22-minute copulatory session. Alpacas show fewer (mean 40.4; range 11–117) urethral contractions than llamas (mean 63; range 38–132). Ejaculate characteristics at 5-minute intervals during copulation in llamas and alpacas are shown in Figure 15-8.26,27
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree