CHAPTER 4 Oral and Nasal Cavities, Pharynx, Guttural Pouches, and Paranasal Sinuses
Indications for Cytologic Examination
Oral Cavity
Clinical signs associated with pathologic conditions of the oral cavity include ptyalism, quidding, foul odor, dysphagia, and depression. Conditions of the nasal passages may result in nasal discharge, epis-taxis, dyspnea, inspiratory stridor, and foul breath. Involvement of the nasopharynx can be associated with dysphagia, dyspnea, abnormal respiratory noise, and exercise intolerance.1
Examination
Diseases of the nasal passages and nasopharynx often require endoscopic examination for adequate visualization.1 Sedation may distort the nasopharynx by relaxation of the soft tissues. Therefore, initial endoscopic examination of this area should be conducted without the aid of sedation if possible. Radiography may also help define the extent of lesions in this area.
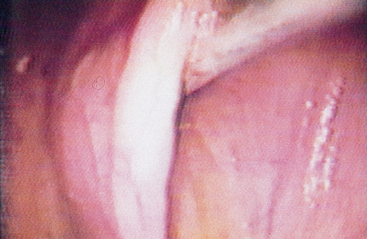
The guttural pouches can be evaluated by palpation, endoscopy, and radiography. Two different methods may be used to insert a flexible endoscope into the guttural pouch. In the first method, place a biopsy instrument or cleaning brush in the biopsy channel of the endoscope and extend it 2 or 3 cm past the end of the endoscope. Then insert the biopsy instrument or cleaning brush into the guttural pouch opening and rotate the endoscope to open the guttural pouch flap. Then advance the endoscope into the guttural pouch. With the second method, place a Chambers mare catheter into the guttural pouch and rotate to open the flap (Fig. 4-1). Pass the flexible endoscope dorsal or ventral to the catheter and into the pouch as the Chambers catheter is withdrawn.
Sample Collection
Lesions in the oral cavity, nasal passages, nasopharynx, paranasal sinuses, and guttural pouches may be defined by cytologic evaluation, histopathologic examination, and culture (bacterial, fungal). Cytologic samples from the oral cavity are usually limited to fine-needle aspirates of masses or imprints for cytologic examination from excised tissues (see Chapter 1 for a discussion of slide preparation techniques). Cytologic preparations from ulcerative lesions may be collected by imprinting, swabbing, or scraping.
Paranasal Sinuses
Though exudate from paranasal sinuses can sometimes be detected endoscopically in the middle meatus, samples for cytologic evaluation and culture should be taken directly from the involved sinus. Sinus aspiration is usually possible in a standing horse using sedation and local anesthesia. The landmarks for frontal sinus aspiration are 2.5 cm caudal to the point at which the nasal bones start to diverge and 2.5 cm lateral to the midline. For superior maxillary sinus aspiration, the landmarks are dorsal to the facial crest and about 2 cm rostral to the bony orbit. To avoid the lacrimal canal, the practitioner must not perform trephining more dorsally than a line drawn from the medial canthus to the infraorbital foramen.2
After surgical preparation and local anesthetic infiltration, make a stab incision in the skin and use a small Steinmann pin to drill into the sinus. Anatomic landmarks and the technique for trephination of paranasal sinuses are described in detail elsewhere.1 Retrieve exudate by aspiration with a male canine urinary catheter or intravenous catheter. If exudate is not easily obtained, infuse and aspirate 30 to 40 ml of physiologic saline. If sinus contents are too thick for aspiration, use the eyed end of a large suture needle for sample collection.
Sample Preparation
Cells collected by washes can be harvested by centrifugation in a clinical centrifuge using the same speed as is used for urine sedimentation. The supernatant is poured off, the pelleted material is gently resuspended, and a drop of the suspension applied to a clean glass microscope slide with an applicator or pipet. The sediment is then spread using a blood smear technique (see Chapter 1) and allowed to air dry.
Preparation of fine-needle aspirate smears and impressions of biopsied samples is covered in Chapter 1. Staining with hematologic stains is satisfactory for cytologic examination of samples from the oral cavity and upper respiratory tract. A more complete discussion on sample collection, slide preparation, and staining is given in Chapter 1.
Normal Cytologic Features
Oral Cavity and Nasal Passages
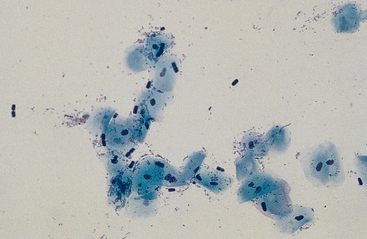
The epithelium of the mucous membranes lining the oral cavity and rostral portion of the nasal passages consists of keratinized and nonkeratinized stratified squamous epithelium; therefore, these surfaces exfoliate squamous epithelial cells (Fig. 4-2). Squamous epithelial cells appear cytologically as large flattened cells with angular borders and abundant, pale-staining cytoplasm and a condensed to pyknotic central nucleus.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree