14 Nutritional management of elite endurance horses
The sport of endurance racing is probably the most demanding of the equine athletic disciplines, with horses required to complete distances of up to 160 km in a single day (and over longer distances during multi-day races). In the past, races tended to be over longer distances and at relatively slow speeds e.g. the winner in an event in Berlin in 1892 travelled 597 km at an average speed of 8.4 km/h, but recently, particularly at the international level, the trend has been for very high racing speeds. The winner of the 2005 World Equine Endurance Championship race in Dubai, for example, covered the 160 km distance at an average speed of 22.5 km/h (~14 miles/h) and the winner of the 2010 World Equestrian Games (WEG) race in Lexington competed at an average speed of 21 km/h (~13 miles/h). In addition, the top three finishers of the 2010 WEG competition completed the final loop at speeds of 30 km/h (~18 miles/h)! These high work rates pose several challenges for the endurance horse. First, high energy demands may result in depletion of substrate stores, particularly muscle and liver glycogen, resulting in a decline in performance. Second, as the evaporation of sweat is the major mechanism for heat dissipation during exercise, there is a substantial loss of body water and electrolytes (especially sodium and chloride). Failure to mitigate the resultant dehydration and electrolyte losses via replacement strategies is another potential reason for poor performance and elimination from the race. Additionally, dehydration and electrolyte imbalances may increase risk for the development of metabolic problems including heat stress, synchronous diaphragmatic flutter, ileus and rhabdomyolysis. In a survey of horses that participated in 16 CEI or CEN events in France, depending on the ride 27.8–69% (mean 50%) were disqualified and between 4.6–17.6% (mean 10.5%) had to be treated (Robert 2004). The frequency of elimination for lameness (~41%) and for metabolic reasons (~40%) was very similar in this survey. The most common clinical diagnoses were dehydration (~26%), colic (~17%), exhaustion syndrome (~13%) and tying up (~13%). It can be argued that appropriate nutritional management may help reduce the incidence of metabolic problems that result in race disqualification and/or the need for veterinary intervention.
Energy metabolism
Body condition score (BCS) reflecting body fat content, (see Chapter 22) may influence performance during endurance rides. In one study of horses that competed in a 240 km (150 mile) ride, over 2 days, the average BCS (scale 1–9) was 4.7 and the percentage of body fat, estimated from ultrasound assessment (see Chapter 22) of rump fat thickness, was 7.8% (Lawrence et al 1992). Among the top finishers the estimated total fat was ~6.5% of body mass, whereas the fat content of non-finishers averaged ~11%. In another study, mean BCS of horses that completed the 160 km (100 mile) Tevis cup ride was 4.5 (measurement taken pre-ride), while all horses with a BCS less than three failed to finish (Garlinghouse & Burrill 1999). Horses eliminated for metabolic reasons had a mean pre-ride BCS of 2.9 as compared to a BCS of 4.5 in those disqualified for nonmetabolic problems such as lameness. The authors of this study suggested that, at least in more difficult rides such as the Tevis Cup, thin horses (i.e., BCS <3) might be at a disadvantage because of lower energy reserves (and potentially lower muscle mass) when compared to horses with higher body condition. Over-conditioned horses (BCS >6) also could have problems due to the extra weight carried plus the insulating effect (impairment to heat dissipation) of the additional subcutaneous adipose tissue (Garlinghouse & Burrill 1999, Langlois & Robert 2008). Similarly, in a recent study (Barnes et al 2010) of an Australian 160 km ride, horses that were eliminated for metabolic reasons weighed less pre-ride than those that completed (and interestingly appeared more dehydrated with greater electrolyte depletion at the mid-ride checkpoint). In general, feeding programs for endurance horses should target a BCS of around 4–4.5 on the 9-point scale. There are many potential pitfalls in trying to determine accurately, using theoretical equations, an individual’s energy requirements (see Chapter 5). It is therefore important to appreciate the value in the functional “energy” check that the assessment of an individual animal’s body condition provides.
The capacity for endurance exercise is dependent on the availability of substrate for the synthesis of adenosine triphosphate (ATP), the cell’s energy “currency.” Stored energy, in the form of muscle and liver glycogen, intramuscular and adipose triglycerides, along with glucose and fatty acids derived from the feed ingested during longer rides are used for ATP synthesis in working tissues. It has been estimated that a 450-kg horse has around 1400–2800 g muscle triglyceride, 40,000 g adipose triglyceride, 3000–4000 g muscle glycogen (1–2% skeletal muscle weight) and 100–200 g liver glycogen (Harris 1997). Fat stores are therefore comparatively larger, and it is currently thought depletion of muscle and liver glycogen stores (Farris et al 1998, Lacombe et al 2001) combined with fluid and electrolyte disturbances (Pösö et al 2004) are factors that contribute to fatigue in endurance racing.
Traditional aerobic conditioning endurance training is associated with an increase in skeletal muscle oxidative capacity and a decrease in anaerobic metabolism as well as alterations in fiber type and mitochondrial respiratory function (Serrano et al 2000, Votion et al 2010). Further work is needed to understand the type of training best suited to achieve the higher speeds of recent elite competitions. At low to moderate work intensities, oxidative phosphorylation (aerobic metabolism) of glucose and fatty acids is an efficient mechanism for regeneration of ATP. At higher workloads (e.g., canter and gallop), however, ATP demand cannot be met by oxidative metabolism alone and non-oxidative breakdown (anaerobic metabolism) of glycogen and glucose (fatty acids cannot be metabolized by anaerobic mechanisms) also contributes to ATP resynthesis. However, anaerobic glycolysis is far less efficient in terms of ATP yield per gram of glucose metabolized and also results in lactic acid accumulation within skeletal muscle, that may also contribute to the development of fatigue (Pösö et al 2004). Historically, aerobic metabolism of fatty acids was thought to predominate during endurance rides undertaken at an average speed of 12–14 km/h (8–9 miles/h) – with increasing contributions from anaerobic metabolism for only brief periods of time, e.g., during controlled “sprints” that some riders use at the beginning or at the end of the ride or when hill climbing. However, this assumption may not apply to modern, elite level endurance racing wherein the more sustained periods of high speed running will invoke a larger contribution from anaerobic metabolism and, therefore, greater demands for use of the more limited body glycogen stores. Even with the low rate of glycogen utilization during submaximal exercise (<15 km/h), if such exercise is continued for several hours, muscle glycogen stores may be depleted by more than 50–75% (Snow et al 1982). Therefore, there is increasing interest in the application of strategies (e.g. diet, training, racing strategy) that might reduce the rate or extent of glycogen utilization.
Key Points
• In general, feeding programs for endurance horses should target a BCS of around 4 – 4.5 on the 9 point scale
• Fatigue during endurance racing is likely to be due to depletion of muscle and liver glycogen stores combined with fluid and electrolyte disturbances
• The increasing speeds now expected at certain international rides challenge traditional concepts of the training and feeding of endurance horses
Energy requirements
Energy requirements for horses are most commonly expressed as megacalories (Mcal) or megajoules (MJ) of digestible energy (DE), where 1 Mcal = 4.184 MJ. For an endurance horse, energy needs depend not only on work duration and intensity but also on environmental conditions, terrain, weight of the rider and tack, ability of the rider, and fitness of the horse, etc. (Harris 1997, NRC 2007). In general terms, the requirement is the sum of maintenance requirements plus an allowance for the work being undertaken. Maintenance DE requirements for a 450 kg endurance horse (NRC 2007) are around 13.6–16.3 Mcal/day (~57–68 MJ/day). Training or competition requirements depend on the weight of the horse plus rider and tack as well as the speed of work (Harris 1997, NRC 2007). As illustrated in Table 14-1 (adapted from Pagan & Hintz 1986) a 450 kg horse (plus 75 kg for the rider and tack) performing a 3 h training ride at a medium trot (~250 m/min) would have an estimated additional DE requirement of 15 Mcal (63 MJ) giving a total requirement of ~29–31 Mcal (~120–130 MJ) for that day. The National Research Council (NRC) publication on nutrient requirements for horses (NRC 2007), uses a multiplier (1.9) over maintenance in order to estimated the requirements for elite endurance horses, which are included in the very heavy work load category. In this category maintenance requirements are taken at the elevated level, which might reflect the increased voluntary activity of such animals or perhaps help compensate for any extra energy costs associated with restoring and maintaining body energy stores. This also gives an estimated requirement value of ~31 Mcal/day (129 MJ/day) for a 450 kg horse. In a US survey of aspiring international level competitors (Crandell 2002) average estimated daily DE intake (from forage and supplementary feeds) during training was 24 Mcal (~100 MJ) for horses with an average body weight (BW) of 460 kg. However, many of the horses surveyed were kept at pasture and the estimate did not account for DE intake from pasture forage.
Table 14-1 A Guide to Potential Digestible Energy (DE) Requirements above Maintenance at Various Speeds (Adapted from Pagan & Hintz 1986: DE (kJ per kilogram of Horse, Rider and Tack) = 4.184 × {[e(3.02+0.0065y) – 13.92] x 0.06}/0.57 Where y is the Speed (Meters per Minute) and 0.57 Accounts for the Efficiency of Utilization of DE)
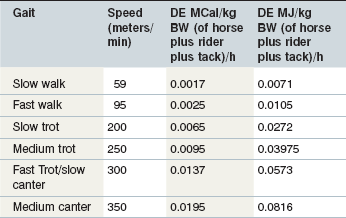
Horses are individuals and therefore actual DE requirements may deviate from calculated estimates, and vary widely among horses of similar bodyweight undertaking the same amount and type of work. From a practical aspect, weekly or biweekly monitoring and assessment of bodyweight and/or BCS (see Chapter 22) will enable the feeding program to be individually tailored.
Meeting energy requirements
The four main sources of energy (Harris 1997) in horse feeds are:
1. Fermentable fiber (includes cellulose, hemicellulose and pectins): these components are not digestible by mammalian enzymes but can be fermented by microorganisms predominantly located in the hindgut. Speed of fermentation as well as site likely influence the energy value to the horse.
2. Hydrolyzable carbohydrates (e.g., simple sugars and starches): these can be digested by mammalian enzymes to hexoses which are absorbed from the small intestine (SI) or, if they “escape” digestion in the SI, they are rapidly fermented in the hindgut, i.e., cecum and colon.
3. Oils/fats: although horses evolved on diets with low concentrations of oils and fats, they are able to digest and utilize up to 20% of the diet (dry matter basis) as oil, if suitably introduced (see further comments below).
4. Protein: protein is not primarily fed as an energy source because metabolism of amino acids to useable energy is inefficient relative to metabolism of carbohydrates and fats (see below for specific comments re protein requirements to support exercise activity and recovery).
Different feeds and feedstuffs contain differing amounts of gross energy. The efficiency of conversion from gross to useable or net energy also differs widely (Martin-Rosset et al 1994), both between the feedstuffs and between individual animals. Vegetable oils provide proportionally more net energy than the cereal grains (~2.5 times as much DE as maize/corn and three times as much as oats). Cereal grains, in turn, provide more net energy than hay, while the net energy from hay is twofold higher than that available from straw (Martin-Rosset et al 1994).
Forage should be the foundation
Forage should be the foundation of the diet for all horses. In the aforementioned US survey of feeding practices (Crandell 2002), about 80% of the horses had 24 h pasture turnout (with additional preserved forage provided some of the year). On average, 78% of the ration was forage which is higher in comparison to other sports horses (e.g. the ration of racehorses may be no more than 30% forage). Although there is currently no agreed upon requirement for dietary fiber, the inclusion of some long stem roughage in the diet is thought to be important for maintenance of hindgut function and health as well as for reduction in risk of gastric ulceration and abnormal behaviours (Goodwin et al 2002, Shirazi-Beechey 2008) and the authors currently recommend a minimum of 1.5% BW on a dry matter (DM) basis of long stem roughage or chopped long fiber (see also Chapter 26). Fiber may provide energy during an endurance ride via absorption of volatile fatty acids (acetate, propionate) produced by fermentation that continues long after the fiber has been ingested. Propionic acid from hindgut fermentation of fiber is also an important substrate for gluconeogenesis (Ford & Simmons 1985).
Some fiber sources (e.g., beet pulp, soya hulls) may augment the size of the large intestinal fluid reservoir, which may represent as much as 8–10% of bodyweight and 10–20% of total body Na, K and Cl (Meyer & Coenen 1989, Warren et al 2001, Parsons et al 2011). This may assist in the maintenance of hydration during exercise by acting as a reservoir for both water and electrolytes (Meyer & Coenen 1989). Extrapolating from studies in ponies (Meyer & Coenen 1989) during low intensity exercise, approximately 10 liters of water, 19 g of Na and 10 g of Cl may be absorbed from the gastrointestinal tract of horses during 2–3 h of exercise, potentially offsetting some of the sweat water and electrolyte losses that occur during endurance rides (Geor & Harris 2005). Recent work has suggested that differences in dietary fiber type, which affect total body water, may also influence core temperature during endurance type exercise (Spooner et al 2007). However, the potential benefits of a high-fiber diet with respect to improved water and electrolyte balance must be weighed against possible energetic disadvantages associated with an increase in hindgut weight (bowel ballast: Kronfeld 2001a, Chapter 26). The optimal mix of dietary fiber types is not known but it is common for endurance horses to consume a variety of fiber sources, including long stem hay, beet pulp and soya hulls (Kronfeld 2001b). Recent work has suggested that organic matter, DM and neutral detergent fiber digestibility may be higher after endurance conditioning but these adaptations were not systematically associated with a longer total tract mean retention time nor an increase in microbial fibrolytic activity (Goachet et al 2010).
Key Points –
Forage feeding
Practical recommendations include:
• Feeding preferably 1.5–2.0 kg (DM basis) per 100 kg bodyweight of long stem roughage or chopped long fiber
• Avoiding overly mature forages, due to reduced digestibility and possibly reduced water reservoir capacity.
• Selecting hay with a low to moderate protein content (i.e., grass hay with crude protein [CP] of 8–14%) rather than a legume hay (often with CP >20%) because of concerns regarding a high protein intake (see below). A good quality grass hay or a grass/alfalfa mix in which alfalfa is no more than 30% of the mix is recommended
• Avoiding high intakes of calcium-rich forages (i.e., alfalfa) due to the perceived increased risk of synchronous diaphragmatic flutter (“thumps”) during endurance rides when horses are fed a high calcium ration
• Appropriate vitamin/mineral and possibly protein fortification will be required especially in highly forage based rations
Supplemental energy sources
Cereal grains
Many endurance horses are Arabian, at least in part, and tend to be “easy keepers”. However, even good quality pasture/preserved forage may not be sufficient to maintain bodyweight and condition during endurance training and competition. Therefore, some cereals are commonly required – the average amount in the aforementioned survey (Crandell 2002) was 2.27 kg/day. However, even higher quantities are likely to be required by horses engaged in heavier training and higher level racing.
Starch, a hydrolyzable carbohydrate, is the principal component of cereal grains (~50% of oats and 70% of corn). Recommendations with respect to starch intake per meal have been reduced for various reasons (link with increased risk of equine gastric ulcer syndrome, insulin response, etc.), in recent years from 4 to 1 g/kg BW or even less under specific clinical circumstances (Potter et al 1992, Kienzle 1994, McLean 2000, De Fombelle et al 2001, Luthersson et al 2009, Vervuert et al 2009, see Chapters 8 and 26), although this may depend to some extent on the source and processing of the starch. It is also advisable to limit the size of individual grain-based meals to no more than 1.5 kg (for a 450 kg horse). Some nutritionists also advocate providing grain-based meals separately from a large meal of long fibrous hay due to potential concerns of lower prececal starch digestibility when these feeds are ingested together (Pagan & Harris 1999), although no effect on the glycemic response has been reported with the addition of short chopped fiber (Harris et al 2005).
Highly digestible fiber sources
As mentioned, the feeding of sugar beet pulp and soya hulls to horses has gained popularity recently (Crandell et al 1999, Palmgren Karlsson et al 2002). When compared to more traditional fiber sources like hay, these highly digestible or “super fibers” contain less indigestible material (e.g., lignin) and higher amounts of non-starch polysaccharides, pectins and gums which can be digested to a large extent within the time period that they remain within the gastrointestinal tract (see Chapter 17). This translates to a higher energy yield. In addition, there may be beneficial effects on hydration due to improved water holding and releasing capacities (Warren et al 2001). In addition, work with resting horses showed that replacing hay with sugarbeet pulp resulted in a decreased water intake and retention but an increase in the proportion of water lost via the urine. During exercise it is possible that these increased urinary losses may be more easily and “safely” reduced providing more water to support sweat losses.
Vegetable oils
Energy-rich vegetable oils contain no starch or sugar and potentially may provide other advantages, including reduced heat production (important in conditions of high heat and humidity) (see Box 14.1), reduced amount of feed needed to achieve desired energy intake, and possible behavioural advantages (Harris 1997, Harris & Harris 1998, Holland et al 1996, Harris & Kronfeld 2003, NRC 2007). Reduced bowel ballast, due to the substitution of some cereals by oil, helps to balance to a certain extent the recommendation for a high fiber intake (which creates more bowel ballast) in endurance horses. Of perhaps more interest is the potential for oil supplemented diets to provide a direct performance advantage. More than 30 years ago, it was reported that horses fed a diet containing 12% fat (9% added corn oil) and ridden 67 km over mountainous terrain for 8–10 h performed better and had higher blood glucose concentrations at the end of the ride than horses fed the control diet (3% fat) (Slade et al 1975). Subsequent studies have demonstrated that oil supplementation is characterized by a dose-dependent increase in the activity of lipoprotein lipase and, in some reports, an increase in the activity of skeletal muscle citrate synthase and beta-hydroxy acyl-CoA dehydrogenase (Orme et al 1997, Dunnett et al 2002). These alterations in enzyme activities may result in increased uptake and oxidation of free fatty acids in skeletal muscle (Pagan et al 2002). Horses fed a diet supplemented with oil to provide 25–30% of DE had a lower respiratory exchange ratio (Dunnett et al 2002, Pagan et al 2002) and decreased glucose utilization (Pagan et al 2002) during low intensity exercise (~ 25–35% VO2max) than horses fed a control (nonsupplemented) ration. Investigators that used compartmental modeling techniques to evaluate glucose kinetics also observed that horses adapted to and fed an oil- and fiber-rich complementary feed utilized less glucose during exercise (4 m/s on a treadmill) when compared to horses fed a starch-rich complementary feed (Treiber et al 2008). Theoretically, enhancement of lipid oxidation and sparing of plasma glucose utilization should result in muscle glycogen sparing and could perhaps improve (or prolong) endurance performance. Preliminary work also suggests that adaptation to fat- and fiber-rich feeds lowers serum insulin concentration during endurance rides in association with improved performance (Hess et al 2007). Overall, however, the effect of diet composition on performance during endurance races currently remains uncertain (Harris 1997, NRC 2007).
Box 14.1
If you need to supply 10 MJ of maintenance net or useable energy then (based on Harris 1997) as a guide to the amounts of feed needed and the amount of waste heat the horse needs to remove:
• 10 MJ would require feeding 1.8 kg of timothy hay and would produce 3.8 MJ waste heat from metabolism plus 1.1 MJ of fermentation heat = 4.9 MJ
• 10 MJ would require feeding 1.14 kg of oats and would produce 2.6 MJ waste heat from metabolism plus around 0.4 MJ of heat of fermentation = 3 MJ
• 10 MJ would require feeding 0.36 kg of oil and would produce 1.44 MJ waste heat from metabolism and nothing from fermentation = 1.44 MJ
The ideal type (including fatty acid profile) and amount of vegetable oil for supplementation of horse diets has not been determined. Corn oil tends to be one of the more palatable oils (Holland et al 1998) but several vegetable oils show acceptable palatability providing they are fresh, not rancid and of a good quality – preferably human grade. Individual horse preferences also occur. Any supplemental oil should be introduced slowly (over 14–21 days) to avoid intestinal disturbances as the capacity to hydrolyse lipids requires time to adapt (Kronfeld et al 2004). However, the equine pancreas has high lipase activity (Lorenzo-Figueras et al 2007) which might explain why horses are able to digest and utilize up to 20% or more of the diet as oil (Kronfeld et al 2004) if introduced appropriately. Supplementation with soya oil (15% DM intake; ~ 35% of daily energy intake) was reported to decrease fiber digestibility in one study (Jansen et al 2007); however the evidence is conflicting as several other studies (analyzed in Kronfeld et al 2004) suggested no effect of fat supplementation on fiber digestibility (see Chapter 7).
Practical recommendations for endurance horses in hard training/competition are that vegetable oil should be 5–8% of the total diet (with 10% as an upper limit) on an as fed basis. An alternative recommendation is to feed up to 1 ml/l kg BWt/day. For reference, 450 ml of oil (~420 g) provides about 3.4 Mcal (14 MJ) of DE. This daily amount should be divided into two to three portions and mixed in with the other complementary feed. Supplemental oil should be introduced gradually (e.g., starting at 50 ml/day) and to obtain optimal metabolic benefits of dietary oil, it has been recommended that the oil be fed for several weeks if not months before competition (Pagan et al 2002). High fat diets are commonly linked with reduced exercise performance in humans, but it is important to note that “normal” baseline human diets tend to contain >10% fat whereas the additional of 500 ml of oil to a 9 kg DM ration in a 450 kg endurance horse is only just >5%.
With the exception of vitamin E (variable amounts), vegetable oils do not provide other nutrients. Indeed, adding oil to an existing ration has the potential to create multiple imbalances, because it adds energy but no accompanying micronutrients, vitamins or amino acids. Therefore, it is recommended to feed a diet in which the inclusion of oil has been balanced with respect to other essential nutrients. Alternatively a vitamin/mineral supplement may be needed to achieve the desired balance and ensure that nutrient requirements are met. Although oil supplementation may enhance vitamin E absorption in horses (Siciliano & Wood, 1993) studies in humans suggest that the requirement for vitamin E increases with increasing dietary polyunsaturated fatty acid content (Wardlaw 1999). The increased fat oxidation that occurs during submaximal exercise following fat supplementation (Dunnett et al 2002) is likely to increase the production of peroxyl free radicals and hence the need for additional dietary antioxidants. Based on studies in other species, it has been estimated that the vitamin E requirement is 0.6 mg α-tocopherol per g linoleic acid and around 3 mg α-tocopherol per g of omega-3 polyunsaturated fatty acid (NRC 1987). Currently, the author (P.H.) recommends an additional 1–1.5 IU per 1 ml of added vegetable oil.
Key Points
• Supplementary energy if required can be provided by a mixture of highly digestible fibers, cereal grains and vegetable oil
• Although not proven to improve performance, it is currently recommended that up to 1ml/kg BW vegetable oil may provide a number of metabolic advantages. Additional Vitamin E should be provided at consistency of 1–1.5 IU/1 ml supplemental oil
• If required any additional starch should be provided at <1 g/kg BW starch/meal
Muscle glycogen storage
During exercise, horses appear to rely more heavily on carbohydrate for energy transduction (from muscle glycogen and blood glucose) than humans (Jose-Cunilleras et al 2006). Suboptimal liver and muscle glycogen content at the onset of exercise may therefore reduce performance (Snow et al 1982, Lacombe et al 1999), However, the carbohydrate based nutritional strategies employed by human marathon runners to raise muscle glycogen levels (exercise-linked depletion followed by a high carbohydrate meal) do not appear to be of any value in the horse. Several studies have shown that diet has minimal impact on the slow rate of muscle glycogen restoration in the horse following glycogen depleting exercise (Essen-Gustavsson et al 1991, Pösö et al 2004, Lacombe et al 2004, Jose-Cunilleras et al 2006). See Chapter 2 for more details.
A recent study reported that oral administration of a hypotonic electrolyte solution after prolonged moderate intensity exercise enhanced the rate of muscle glycogen storage when compared to no fluid treatment (Waller et al 2009). The authors suggested that post-exercise dehydration may be one factor that contributes to the slow muscle glycogen replenishment in horses. This may be a very important consideration in the endurance horse.
Timing of feeding relative to exercise
There is evidence from several studies that the timing and composition of a meal consumed before exercise can influence metabolic responses and possibly distribution of body fluids in horses (Harris & Graham-Thiers 1999). In particular, hyperglycemia and insulinemia associated with digestion and absorption of grain meals affects the mix of substrates utilized during a bout of exercise. Insulin is a potent inhibitor of lipolysis and fatty acid oxidation in skeletal muscle, and also promotes glucose uptake into muscle via recruitment of the glucose transporter protein GLUT4 to the sarcolemma. Thus, an elevated circulating insulin concentration at exercise onset will suppress free fatty acid availability as well as lipid oxidation and increase reliance on carbohydrate stores (including plasma glucose) for energy transduction. The resultant decrease in plasma glucose concentrations that occurs when horses are exercised 2–3 hr following a grain meal tends to be relatively transient (Pagan & Harris 1999). However, plasma free fatty acids concentrations and lipid oxidation rate may remain lower in fed animals when compared to the fasted state throughout certain types of exercise (Jose-Cunilleras et al 2002). The impact of feeding may be more complex in endurance horses because they are also offered carbohydrate-rich feeds at rest stops during rides. In this circumstance exercise-associated alterations in hormones (e.g., increased catecholamines, decreased insulin) may counterbalance the effect of any hormonal changes induced by feeding.
Large meals (hay or grain/concentrate or mixtures) may result in a decrease in plasma volume, reflected by an increase in plasma protein concentration, as a result of a fluid shift into the lumen of the gastrointestinal tract (Pagan & Harris 1999) and should therefore be avoided shortly prior to exercise. Although the effects of pre-exercise grain feeding on endurance exercise performance in the field have not been reported, the potential acceleration in carbohydrate oxidation and suppression of fat oxidation lead to a current recommendation that grain or concentrate based meals should not be fed within 3 h of a race.
Protein nutrition
Additional protein over maintenance requirements may be needed with exercise and training because of the accompanying muscular development, the need for muscle repair and to replenish nitrogen lost in sweat (~20–25 g/kg sweat loss). The precise protein requirements for exercise, however, are unknown and the current NRC recommendations (NRC 2007) are 1.26 g CP/kg BW/day for maintenance plus between 0.089 and 0.354 g CP/kg BW/day depending on the exercise level and sweat nitrogen loss (see also Chapter 6). The author’s (P.H.) current recommendations are to feed 2.0–2.5 g CP/kg bodyweight/day (providing stable hygiene is good and water intake is not restricted). There is some evidence that dietary protein level may alter urea metabolism in horses. It has been estimated that a change in dietary CP from 10% to 15% of DM intake would increase water requirement by approximately 5% because of an obligate increase in urine production for clearance of endogenous urea (Kronfeld 1996). Higher protein diets may also be undesirable because of the effects of excess dietary protein on heat production, acid-base balance (especially as speeds increase), and possibly respiratory health (due to ammonia accumulation in confinement housing) (Graham-Thiers et al 2000). However, in Standardbred trotters it has recently been reported that feeding a forage only diet with a high CP intake (16.6%) increased glycogen and leucine concentrations in the muscle of trained animals compared with a lower CP forage (12.5%) and had no effect on plasma lactate and blood pH during exercise tests performed to mimic a race. The authors concluded that the higher protein intake might be beneficial for muscle recovery following intensive exercise (Essen-Gustavsson et al 2010). It is important to note that the responses were not compared with those from animals fed a forage cereal/sweet feed based diet and these were trotters rather than endurance horses.
It has in fact been recommended (Meyer 1987) that athletic horses should not be fed more than 2 g of digestible protein/kg BW/day (around 3–4 g CP/kg BW/day depending on the feedstuffs). Some potential advantages during more short term exercise (Graham-Thiers et al 2000) have been demonstrated with a restricted protein diet (7.5% CP with added lysine and threonine) but this has not been proven in the field to date. Further work is needed with respect to the optimal protein requirements for endurance horses racing under different environmental conditions and speeds.
The quality and nature of the protein fed is important, especially in growing horses and those in hard or repetitive work (NRC 2007, see also Chapter 6). Recent work in humans suggests that the timing and type of protein/amino acid supplementation may influence protein synthesis and degradation in muscle (Phillips 2011). However, detailed information for the endurance horse is not currently available. At the present time it is therefore recommended that as a start, the lysine and possibly threonine content of the diet of actively exercising horses at least should be considered. A recommended allowance (PH) for lysine in hard working endurance horses is 0.08–1.0 g/kg BW/day. Soya bean meal or flakes are a good source of lysine. The amount of additional lysine needed will depend on the hay and pasture being fed – for example, alfalfa and other legumes have higher lysine content than many meadow hays and grasses.
Branch chain amino acids (BCAA)
Supplementation with BCAA (valine, leucine, and isoleucine) has been advocated as a strategy to improve performance. One theory suggests that BCAA supplementation increases the concentration of trichloroacetate intermediates (anaplerosis) available for condensation with acetyl-CoA, enabling an increase in the turnover rate of the cycle. A second theory is that BCAA may attenuate fatigue during exercise by limiting the rise of free, unbound plasma tryptophan which results in elevated brain serotonin and development of central fatigue. However, this theory has been disputed (Grimmett & Sillence, 2005) and research in the horse has failed to show any beneficial effect of BCAA supplementation on exercise performance (Casini et al 2000, Steffanon et al 2000
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


