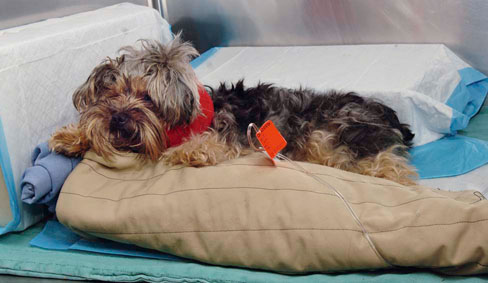
Nursing care is essential during treatment of the physical rehabilitation patient. As discussed in depth in Chapter 7, muscles and tendons quickly atrophy with immobilization or reduced activity, and other tissues such as cartilage, ligaments, and bones also undergo rapid changes during the postoperative period. Starting physical rehabilitation as soon as it is safe can minimize these changes. There are several considerations one should consider when working with a postoperative patient, some of which may be severely injured and therefore recumbent or nonambulatory. Taking care of a patient that is nonambulatory or weakly ambulatory can be emotionally and physically challenging. It may be best to start with basic functions, such as learning to stand and walk again. Nursing care during the critical postoperative period in the hospital can be achieved by veterinarians, technicians, assistants, and ultimately the owner. It is important for every person involved to understand the recovery process to provide the best supportive care possible. Rehabilitation should begin as soon as the animal is stable, and before the onset of complications that are often associated with prolonged immobility. A proactive approach to rehabilitation requires less effort, and better outcomes may be obtained than one that is in response to a developing problem. Veterinary professionals have a responsibility to act as advocates for animals when people bring their pets for care and treatment. Providing a clean, dry bed in an appropriate cage or enclosure is vital (Figure 16-1). In some veterinary hospital environments housing is limited to cages elevated above the ground or runs enclosed by fencing, thus creating a “step” in and out of the cage or run. These circumstances provide challenges for the preoperative/postoperative patient and those who care for them. It may seem elementary to consider but patients should be cared for as if they were our own pets and we should occasionally stop to consider, “If I were my patient, how would I feel? What would make me feel better?” Rubber mats may be used to provide appropriate footing in cages and kennel runs (Figure 16-2). An essential factor with any orthopedic or neurologic patient that is unable to walk is preventing decubital ulcers. Decubital ulcers develop over bony prominences such as the olecranon process, acromion process, greater trochanter, tuber ischium, and medial and lateral malleoluses. Prevention is crucial and begins with proper bedding. A helpful rule of thumb is that 1 inch of bedding should be used for every 40 pounds of body weight. Bedding should be changed daily and the cage or run cleaned daily as well. It has been well documented that frequent position changes and ample bedding decrease the incidence of pressure sores, edematous limbs, and joint and muscle stiffness.1,2 Recumbent patients should be turned every 4 to 6 hours at a minimum. Providing absorbent pads underneath the patient to facilitate the absorption of urine and feces of recumbent patients is essential to keep the patient and cage from becoming excessively soiled. Alternatively larger patients that are incontinent may be placed on fenestrated grates to allow excess urine and feces to drain away from the patient. Urine- or fecal-scalded skin typically appears red, irritated with or without serous exudate, and is caused by prolonged exposure to urine or feces (Figure 16-3). Proper nursing care should be initiated immediately if scalding occurs and consists of cleansing the area, drying thoroughly, applying creams such as Desitin (Johnson & Johnson, New Brunswick, NJ) or Skin Sake (Skin Sake, Hamburg, AR) (Figure 16-4) to help protect the skin and baby powder to the fur to absorb additional fluid. The importance of preventing the patient from licking its urine/perianal scald, wounds, and/or incisions is paramount. A no sting barrier film (Cavilon, 3M, St. Paul, MN) (Figure 16-5) also can be used to protect the skin. The Elizabethan collar, or e-collar, is very helpful to discourage patients from licking their incision. The art of a functioning e-collar lies in the sizing and fitting of the collar. Elizabethan collars are typically made of plastic, but softer cloth e-collars are also commercially available. When placed appropriately the brim of the e-collar should extend well past the patient’s nose (Figure 16-7). This ensures the patient cannot lick the incision. The collar should be comfortably snug around the patient’s neck. To check the tightness of the collar, two fingers are passed under the collar. If two fingers cannot be passed under the collar, it is too tight. If three or more fingers can be passed under the collar, it is too loose. Most commonly, e-collars are too loose and the patient has little trouble getting it off. Patients must become accustomed to daily tasks such as eating and drinking while wearing an e-collar. Food and water must be placed close enough to the patients for them to reach the bowls. Attention must be paid to the patient’s appetite and willingness to eat. Many patients have difficulty learning to eat while wearing an e-collar. In this case the e-collar can be removed, feeding should be supervised, and the collar promptly reapplied after eating or drinking. In addition many patients refuse to eat or drink during their stay at the hospital. Hand feeding or assisted syringe feeding using pureed canned food is recommended, but feeding should be slow enough to allow ample time for swallowing. Patients should wear the e-collar at all times in the hospital and at home until the sutures or staples are removed and any open wounds are adequately healed. It is important to realize that many owners may give their pets aspirin, but do not consider this as a medication because it is easily obtained over the counter. It is critical to take a thorough medication history to prevent serious side effects from drug interactions. Aspirin is a nonselective COX inhibitor and may increase gastrointestinal side effects when given with other NSAIDs or COXIB class medications.3 For the neurologic patient with intervertebral disk disease or trauma, triage begins with the history. It is vital to collect information regarding the duration of clinical signs, the time elapsed since the patient became paretic/paralyzed, and any use of steroids or other antiinflammatory medication. After a patient has lost deep pain there is a 50% chance it will regain functional mobility if surgery is performed within 24 hours. In general every hour past 24 hours from the point when deep pain is lost decreases the prognosis for a full recovery exponentially.4 It is good to remember that the loss of deep pain does not definitively preclude a dog from regaining motor function. However, dogs that do not recover deep pain, rarely regain conscious voluntary motor function and their ambulation is not normal. It is also important to remember that any dog that has intact deep pain in its affected limbs has the potential to recover function. Many patients receive local or regional nerve blocks or epidurals before or immediately after surgery. It is extremely important to monitor the urinary status of patients after receiving epidurals. A high percentage of patients retain urine following epidural morphine (Duramorph) administration. There are three suggested mechanisms that may explain urinary retention following epidural morphine administration: (1) local detrusor inhibition through opiate receptors; (2) inhibitory action on the primary micturition center in the caudal brainstem; and (3) inhibition of parasympathetic nervous activity following binding of morphine with opiate receptors in the sacral spinal cord, resulting in detrusor muscle relaxation and increased bladder filling.5,6 A 50/50 mixture of bupivacaine and Duramorph may be administered to provide more balanced analgesia. Animals that are recumbent for a very long time may be at risk for developing decubital ulcers. It may take as little as 1 hour of sustained pressure to develop a deep decubitus.7 In particular, animals with thin skin and prominent bony protuberances, such as greyhounds and whippets, are especially prone to developing decubital ulcers. Prevention of ulcers is far superior to treatment of ulcers. Therefore the skin should be assessed each day for redness of the skin or other evidence of skin breakdown. If redness or irritation of skin is noted, a “doughnut” may be made to help distribute pressure away from the area. The adept practitioner should be familiar with the characteristics and appearance of “normal” incisions, in order to properly identify questionable or compromised incisions (Figure 16-8). It is important to note the location of the incision(s), the number of incisions, number of individual sutures, and the type of incision closure(s). Effort should be made to ensure the incision(s) is clean and dry. Following surgical closure of the incision in the operating room the skin around the incision is cleaned with either warm sterile saline or hydrogen peroxide and allowed to dry. Although hydrogen peroxide may recruit free radicals and cause oxidative injury to tissue, the use of hydrogen peroxide to clean blood and other fluids from the skin around the wound has minimal effects on healing if not directly applied to the incision. For standard orthopedic procedures a nonadherent pad is applied with triple antibiotic ointment and affixed over the incision with wide adhesive tape (Figure 16-9). This temporary dressing serves to protect the incision and may be removed when the incision is sealed after 2 to 3 days if there is no drainage. Often the postoperative orthopedic patient’s incision will have slight bloody or serosanguinous discharge the day of surgery and the day following surgery. This is considered to be normal and becomes less prominent as a fibrin seal is created approximately 24 to 72 hours8 after surgery (Figure 16-10). When allowable a cold compress can be applied immediately after surgery for 15 to 20 minutes to the operative site to provide analgesia, and to decrease inflammation and edema formation.9,10 The patient’s incision should be monitored daily for any drainage or discharge, apposition of wound edges, excessive inflammation or swelling, and any missing staples or sutures (Figure 16-11). Before discharge the patient’s incision should be evaluated and cleaned again if needed. The patient should not be discharged with an actively draining or open incision (Figure 16-12). Patients with edema in a single limb should have this limb elevated to help prevent swelling by lying on the opposite side and using a rolled towel or pillow to elevate the edematous limb if tolerated. Cold compresses may be applied to the operative site immediately following surgery. Common practice is to affix a nonadherent sterile pad with adherent tape over the incision, and apply the compress directly on or around the temporary dressing. Studies of human patients have indicated that it is not effective to apply a cold compress through a thick padded bandage.11 Thus cryotherapy should be performed at the time of bandage changes if a bandage must remain on the patient. When applying a cold compress, a barrier such as a nonadherent sterile pad or very thin towel may be placed between the skin and the cold compress to maintain patient comfort (Figure 16-13). It is important to use a compress appropriately sized for the patient. For example in a small dog with a distal radius and ulnar fracture that has been repaired with a bone plate, an extra-small to small cold compress should be used. Likewise for a mastiff with a femur fracture repair, a large to extra-large cold compress would be best. For more information on types of cold compresses, the reader is referred to the chapter on superficial thermal modalities. It is important to remember that if the tissues are inflamed from disease, injury, or surgery, application of heat is contraindicated. Warm compresses may begin after the acute postoperative inflammatory phase has ceased, usually around 72 hours postoperatively or postinjury. The application of heat results in increased blood flow to the area, and possibly decreased muscle spasms. When applying a warm compress when drainage is present, always provide a barrier, such as a nonadherent sterile pad or an absorbent pad with a plastic backing, between the skin and the compress to reduce disease transmission. It is important to always test the compress before application on the patient to be certain that it is not too hot. Several commercially made compresses are available. For more information regarding types of warm compresses, see Chapter 18, Superficial Thermal Modalities. Soft tissue massage can be beneficial in reducing postoperative edema. The degree of limb edema can vary following orthopedic surgery depending on the size of the patient, the procedure performed and the severity of the injury. Ideally massage is performed following superficial thermal modalities and before assisted ambulation or controlled therapeutic exercises. It is important to begin the massage distally and work proximally to increase lymphatic and venous flow and thereby decrease edema (Figure 16-14). The edges of the incision should be avoided. The degree of pressure applied by the hand and fingers should be gentle but firm. Patients that have compromised incisions or skin grafts are not candidates for soft tissue massage. The reader is referring to the chapter on massage for more information regarding massage techniques. Standing exercises focus on strengthening shoulder, elbow, hip and stifle extensors and reeducating muscles needed for balance and proprioception. Several approaches can be used to facilitate standing. Particularly when a patient is large, assistive devices such as slings, physio rolls, mobility carts, wheelchairs, or lifts may be used to maintain a patient in a standing position (Figure 16-15). In smaller patients, the therapist can manually support the patient to maintain a standing position (Figure 16-16, A). Two common variations of this exercise are static standing and sit-to-stand exercises. Static standing is typically performed three to five times daily for 2 to 5 minutes or until the patient fatigues. It is useful to support the patient under the ischiatic tuberosity and on the cranial aspect of the stifles to facilitate a normal standing angle of the hind limbs (Figure 16-16, B). Conversely one often will need two people to maintain limb position in a tetraparetic patient even with abdominal support from a physio roll or sling system. It is important to maintain normal foot position at all times during this exercise (prevent knuckling and encourage weight bearing). It is recommended that one should perform standing exercises at the time of eating or drinking, because standing often improves while the patient is distracted by the act of eating and drinking. Assisted ambulation is paramount with orthopedic and neurologic patients. It is necessary to assist postoperative patients getting into and out of cages, and to help them up from a recumbent position, as well as while ambulating. The caregiver provides assistance when needed and also prevents the patient from falling or overloading tissues and surgical repairs (Figure 16-17). Caution should be taken if the patient is excessively painful or ill tempered. To best help these patients begin early ambulation, the patient must trust the caregiver. Therefore some time must be invested in allowing the patient to become accustomed to the assistive device and the procedures involved in placement and assisting ambulation. Numerous assisted ambulation devices such as slings and harnesses are commercially available. The practitioner should be familiar with the devices, how they work, and how to graduate a patient from one device to a more challenging device. For more on this topic see Chapter 17, Assistive Devices. Encourage owners to make everyday activities a rehabilitation exercise in disguise. For example, while a postoperative patient is standing to eat from a food bowl, an opportunity is presented to do some standing and weight-shifting exercises. Not only will the dog probably be more interested in the food than the standing or weight-shifting exercise, but the behavior is tolerated and positively reinforced with the food (Figure 16-18). Other factors can also increase infection risk, such as prolonged operative and anesthesia times, number of personnel in the operating room, prolonged hospitalization, and the experience level of personnel. The level of microbes in the air of the operating room is directly proportional to the number of people moving in the room; thus Staphylococcus infections can be reduced if traffic is limited in the operating room during surgical procedures.12 Effort should be made to reduce operative times and limit foot traffic in the operating room. Perioperative antibiotic therapy is indicated in contaminated cases (open surgeries), patients with severe soft tissue swelling or bruising as a result of trauma, or in cases with lengthy operative times (longer than 2 hours).13 Cefazolin is commonly used at a dose of 22 mg/kg given 20 minutes before surgery, and every 2 to 4 hours thereafter throughout the surgical procedure. The most common types of infectious agents seen in postoperative surgical wounds are Staphylococcus species and Escherichia coli.13 Infection can be a costly complication of orthopedic surgery and affect the ability of the patient to undergo physical rehabilitation. The patient in Figure 16-12 had a tibial plateau leveling osteotomy (TPLO) that developed a postoperative infection. Wound management was performed daily under sedation and consisted of flushing, wound debridement, and packing it with antimicrobial gauze. A drain was placed and the wound was surgically closed after 2 weeks. In human medicine the incidence of methicillin-resistant Staphylococcus aureus (MRSA) infections has increased. The term methicillin-resistant was originally used in 1961 after a Staphylococcus aureus infection in the United Kingdom was found to be resistant to methicillin.14 MRSA is a gram-positive bacterium that is resistant to methicillin and many other β-lactam antimicrobials; β-lactam antimicrobials include penicillins and cephalosporins. The major vector for transmission is unwashed hands, although MRSA can live on the patient’s skin, in the environment, and on surfaces for extended periods of time. Approximately 25% to 40% of humans are carriers of the organism, mostly in the nasal passages.15 The organism lives harmoniously with the host, but an immunocompromised individual or skin wounds can allow infection to occur. These infections can have dire consequences in humans, but so far do not appear to be life threatening in canine patients. Morbidity generally consists of incisional dehiscence, septic arthritis, and chronically draining wounds necessitating implant removal. Methicillin-resistant Staphylococcus aureus was considered solely a human pathogen for many years until it was first isolated in a dairy cow in 1972.16 Since then the veterinary medical community has seen an increase in the incidence of postoperative infections. Specifically infections caused by MRSA and more commonly, methicillin-resistant Staphylococcus pseudintermedius (MRSI), have been identified following both soft tissue and orthopedic surgeries, as well as cases of pyoderma. Although a standard method for typing and naming MRSA strains has yet to be developed, many methods of nomenclature are currently in use.15 MRSA infections in small animals are predominated by the strain USA 100, whereas MRSA infections in large animals are predominately strain USA 500.17 Although it was previously thought that MRSA could only be transmitted from human to animal, emerging evidence indicates that MRSA can be transmitted in both directions, from human to animal and from animal to human. After an animal becomes colonized it can act as a reservoir, transmitting the infection to humans or other animals.17 Although identification of carriers is paramount to prevention of MRSA and MRSI transmission, routine screening of animals is not yet practiced; therefore the possibility remains that colonized animals will go undetected in veterinary settings.18 Research is needed to determine adequate treatment regimens for MRSA- and MRSI-infected animals, control of MRSA and MRSI transmission, and environmental sanitation. In the interim practicing proper hand hygiene and sanitization is integral to the prevention of MRSA and MRSI transmission. In addition, disposable gowns, gloves, and masks should be worn to change bandages and wound dressings of afflicted patients. Signs of MRSA and MRSI in dogs include acute edema around the incision or wound, excessive scratching or licking, pimple-like lesions on the skin with larger “bumps” in the area, and drainage from the wounds. The fluid should be cultured for both aerobic and anaerobic bacteria. It generally takes 72 hours to obtain culture and antibiotic susceptibility profiles of organism(s). In this case, the client is instructed to continue on an empirical antibiotic or perhaps no antibiotic, until the results are available. During this time it is imperative that the patient be prevented from licking the wound. After culture and susceptibility results are available the patient is placed on the appropriate antibiotic therapy for 3 to 6 weeks. In serious cases the patient may need to return every other day or daily or be hospitalized for bandage changes and wound debridement (Figure 16-19).
Nursing Care of the Rehabilitation Patient
Preoperative Considerations
Clean, Dry, Safe, and Comfortable Housing
Bedding and Decubital Ulcers
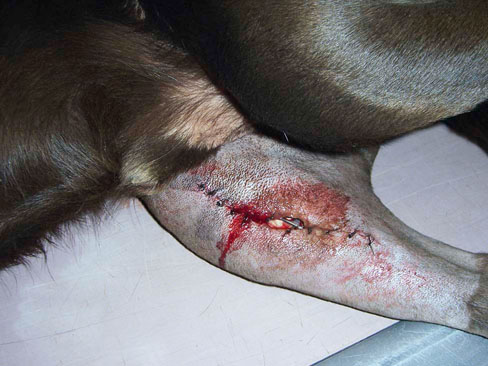
Urine/Fecal Scald
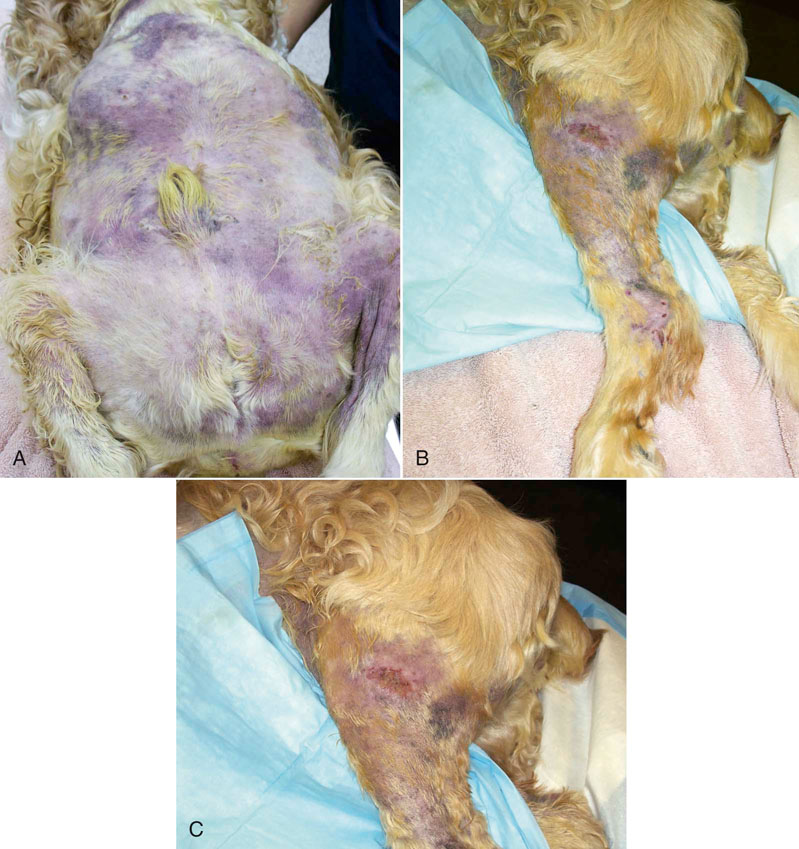
Figure 16-3 Patient with absence of deep pain with urine scald. A, View of abdomen. B, Lateral aspect of hindlimb. C, Close up view of lateral aspect of hindlimb.

Figure 16-4 Skin Sake (Skin Sake, Hamburg, AR) or other skin barrier cream can be applied to areas of skin with urine scald.
Elizabethan Collars
Medications
Understanding When, What, and How
Postoperative Considerations
Postoperative Pain Management
Managing the Recumbent Patient
Care of the Incision
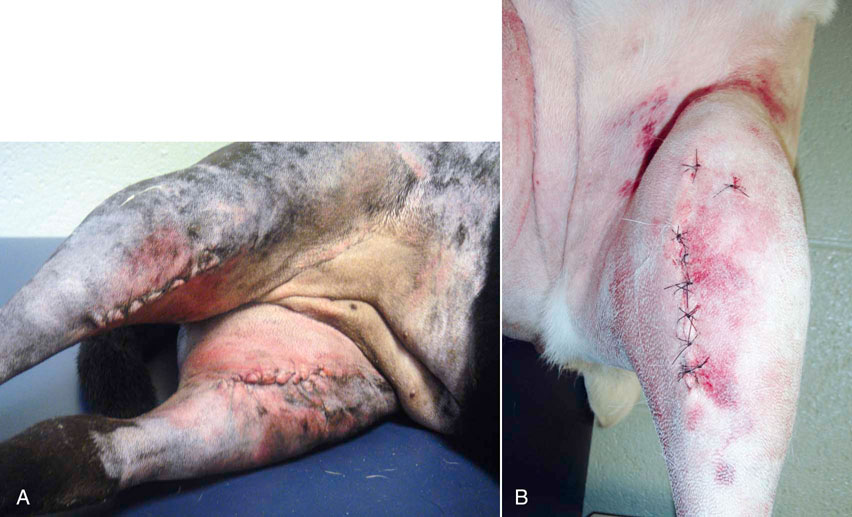
Figure 16-8 Incisions following surgery. A, One day postoperative bilateral TPLO incisions. Note that there is swelling and edema, resulting in constriction of the soft tissues by the sutures. There is also bruising. Bilateral procedures may be at greater risk for incisional complications. B, More normal one day postoperative TPLO incision.
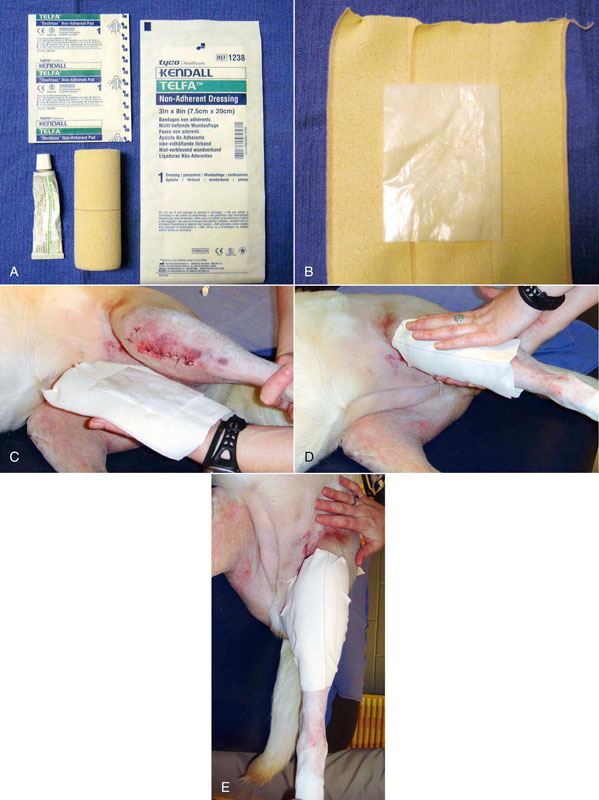
Figure 16-9 Placement of a protective dressing following surgery. A, Materials needed to apply a temporary dressing. B, A nonadherent contact dressing is placed on an adhesive bandage. Triple antibiotic ointment is applied to the pad. C and D, Application to the incision. E, Final placement of temporary protective dressing.
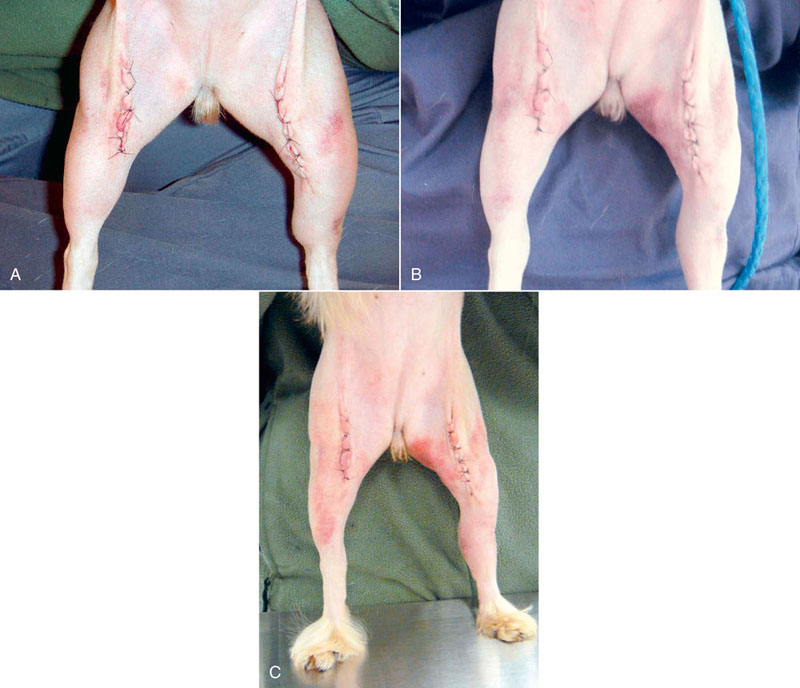
Figure 16-10 Bilateral incisions for luxating patella corrective surgery. A, 24 hours postoperatively. Note that the sutures are somewhat tight. B, 48 hours postoperatively. C, 72 hours postoperatively.
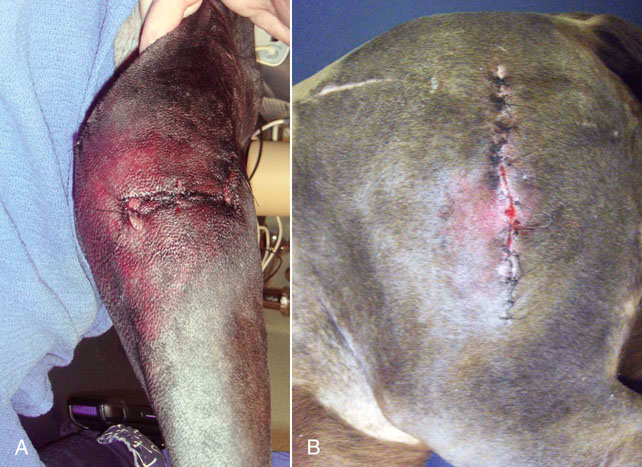
Figure 16-11 A, Postoperative stifle wound repair with marked increase in erythema and edema. B, Postoperative femur fracture repair with a compromised incision. Note area of missing sutures.
Cold Compresses
Warm Compresses
Massage
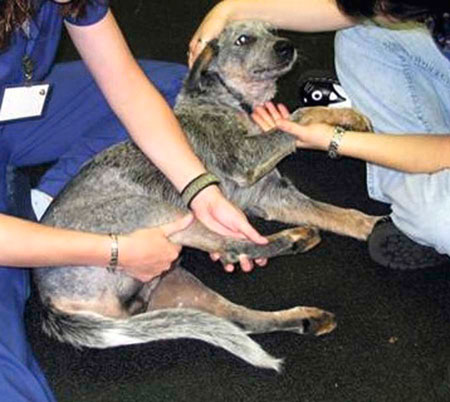
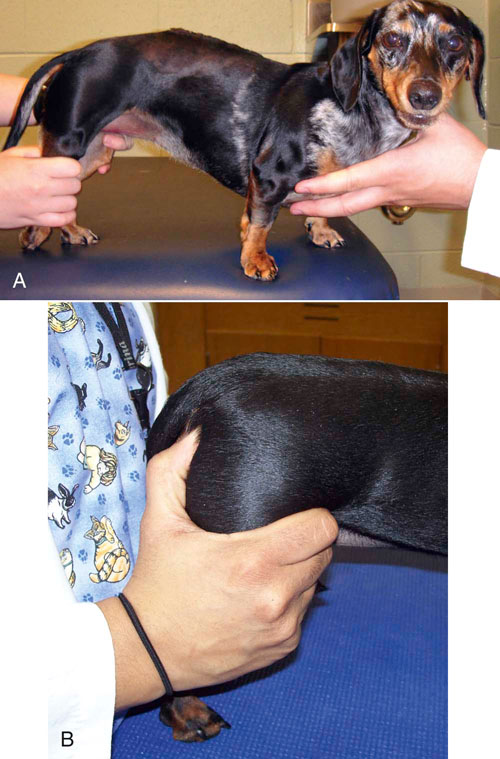
Assisted Standing
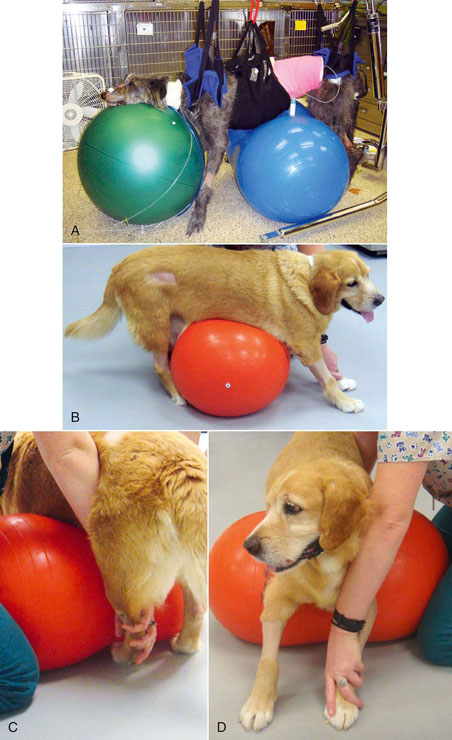
Figure 16-15 Methods of manual assistance for patients. A, Assisted standing exercise in a critically ill patient provided via Hoyer lift, slings and therapy balls. B, Assisted standing over therapy roll by a dog recovering from a cerebellar infarction. C, Note manual placement of the left hind foot during assisted standing exercises. D, Note manual placement of the left forefoot during assisted standing exercises.
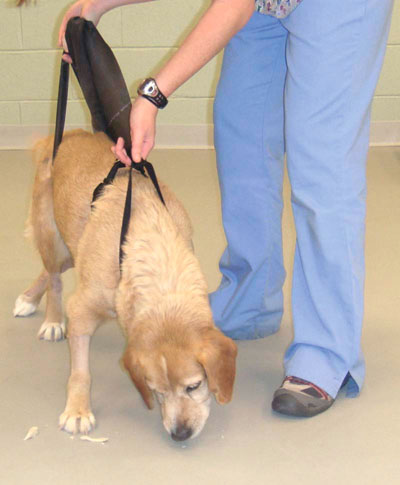
Assisted Ambulation
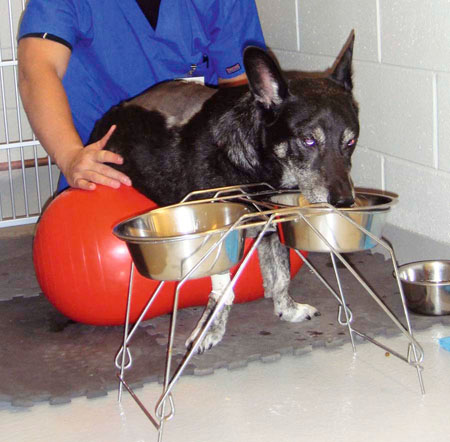
Everyday Activities as Rehabilitation Exercises
Nursing Care for the Orthopedic Patient
Postoperative Orthopedic Infections
Recognizing Signs of Infection
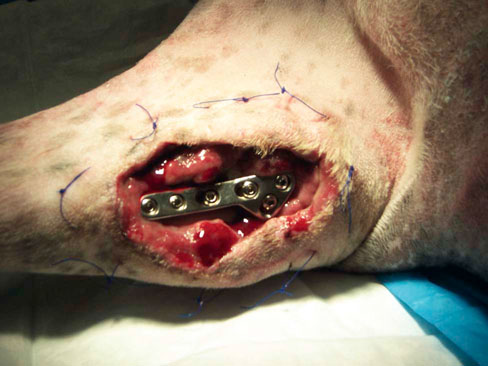
Figure 16-19 Postoperative infected TPLO after several days of daily aggressive wound management. The sutures are for securing a tie-over dressing.![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Nursing Care of the Rehabilitation Patient
Only gold members can continue reading. Log In or Register to continue


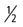 ” Eco-soft tiles, Foamtiles.com, Chandler, AZ). They can be pieced together to fit a variety of cages and are also easily cleaned and sanitized between patients.
” Eco-soft tiles, Foamtiles.com, Chandler, AZ). They can be pieced together to fit a variety of cages and are also easily cleaned and sanitized between patients.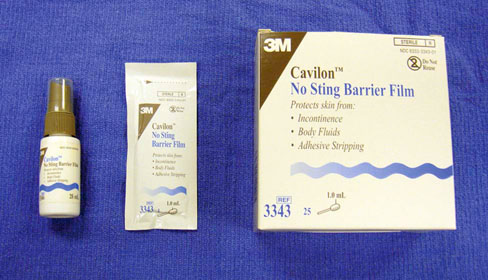

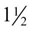 -inch needle is inserted into the joint, usually just medial to the patellar tendon. In the event that the joint is not infected, caution should be used to avoid affected skin and subcutaneous tissues while obtaining joint fluid to avoid carrying infection into the joint. Fluid is allowed to drip from the needle into a sterile collection vial. If fluid does not drain a syringe may be attached and fluid gently aspirated and collected into the proper tubes or culturettes. Normal joint fluid is clear and viscous. Abnormal joint fluid may be cloudy and red-tinged, indicating the presence
-inch needle is inserted into the joint, usually just medial to the patellar tendon. In the event that the joint is not infected, caution should be used to avoid affected skin and subcutaneous tissues while obtaining joint fluid to avoid carrying infection into the joint. Fluid is allowed to drip from the needle into a sterile collection vial. If fluid does not drain a syringe may be attached and fluid gently aspirated and collected into the proper tubes or culturettes. Normal joint fluid is clear and viscous. Abnormal joint fluid may be cloudy and red-tinged, indicating the presence 