CHAPTER 88 Medical Management of Horses After Colic Surgery
The prognosis for survival in horses undergoing colic surgery has improved dramatically during the past 10 to 15 years, with short-term survival rates of horses recovered from general anesthesia being as high as 90%. Although much of the improvement in survival rate can be attributed to early referral and surgical intervention, improvements in general anesthesia and postoperative medical management have also been important. With development of the specialty of equine emergency and critical care, clinicians are beginning to focus more on monitoring and treatment of the critically ill postoperative colic patient. Providing adequate cardiovascular and metabolic support in addition to analgesia is vital for healing of the damaged gastrointestinal tract and surgical site. Diagnostic tests and treatment should be goal directed to optimize treatment success and minimize expense. Meticulous medical records should be maintained for postoperative colic patients. Horses should be monitored closely with physical examination findings and various monitoring tools.
MONITORING
Physical Examination
Horses should be examined every 2 to 6 hours after colic surgery, depending on the severity of illness and the time since surgery. Observations should be recorded on a form similar to the one in the example provided (Figure 88-1).
Laboratory Data
Serum creatinine concentration can be measured to assess organ and tissue perfusion. If a horse that has undergone colic surgery is persistently azotemic (Table 88-1), either the IV fluid rate is inadequate (prerenal azotemia) or the horse is in renal failure. Renal failure can be diagnosed on the basis of isosthenuria, urinary casts, and decreased urinary fractional excretion of sodium.
Table 88-1 Values of Measurements Used for Monitoring Cardiovascular and Metabolic Status in Healthy Adult Horses
| Variable | Reference Value |
|---|---|
| Serum or plasma creatinine concentration | <1.8 mg/dL |
| Serum triglyceride concentrations | <50 mg/dL |
| Blood lactate concentration | <2 mmol/L |
| PcvO2 | 45 ± 4.7 mm Hg |
| ScvO2 | 65%-75% |
| Systolic arterial pressure (direct) | 126-168 mm Hg |
| Diastolic arterial pressure (direct) | 85-116 mm Hg |
| Mean arterial pressure (direct) | 110-133 mm Hg |
| Systolic arterial pressure (indirect) | 98-125 mm Hg |
| Diastolic arterial pressure (indirect) | 54-82 mm Hg |
| Mean arterial pressure (indirect) | 70-96 mm Hg |
| Central venous pressure | 7-12 cmH2O |
| Colloid oncotic (osmotic) pressure | 19-31 mm Hg |
Direct, Directly measured; indirect, indirectly measured; PcvO2, central venous partial pressure of oxygen; ScvO2, central venous oxygen saturation.
Blood and urine glucose concentration should be monitored, particularly in horses receiving dextrose-containing fluids. An insulin constant-rate infusion should be considered in some horses with persistent hyperglycemia and glucosuria. Blood glucose concentration should be maintained from 80 and 120 mg/dL. The renal threshold for glucose is 160 to 180 mg/dL. Serum triglyceride concentration should be measured in horses at risk for developing hyperlipemia-hyperlipidosis (see Table 88-1).
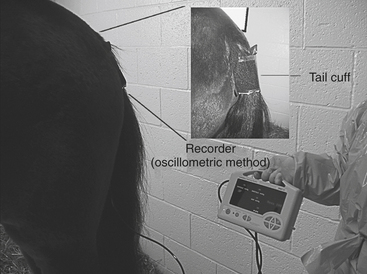
Arterial Blood Pressure
Arterial pressure, particularly mean arterial pressure (MAP), can be used to estimate organ and tissue perfusion. Horses may have low MAP (hypotension) as a result of hypovolemia or activity of the systemic inflammatory response syndrome (SIRS), which can be a response to endotoxemia. Arterial blood pressure can be measured directly or indirectly. Direct measurement of arterial pressure is performed with an arterial catheter, pressure transducer, and continuous recorder. Indirect arterial pressure is most commonly measured by means of the oscillometric method, with an occlusion cuff placed over the coccygeal (tail), dorsal metatarsal, median, or palmar digital (limb) artery (Figure 88-2; see Table 88-1). The internal inflatable bladder length should be 80% of the tail or limb circumference, and the bladder width should be 20% to 25% and 40% to 50% of the tail and limb circumferences, respectively. For measured pressures to be considered reliable, the heart rate recorded by the oscillometric method should be the same as that obtained on physical examination. The accuracy of indirect blood pressure measurements can be improved by taking serial measurements, with the horse minimally restrained in a quiet environment and the head maintained in a resting position.