Chapter 100 Intracranial Hypertension
PHYSIOLOGY OF INTRACRANIAL PRESSURE
INTRACRANIAL PRESSURE
Intracranial pressure ICP is the pressure inside the cranial vault exerted by the tissues and fluids against the encasing bone. Normal ICP in the dog is 5 to 12 mm Hg, similar to that of humans for whom 20 mm Hg is an arbitrary upper limit beyond which treatment for ICH may be instituted1,3 (see Chapter 209, Intracranial Pressure Monitoring). The upper limit of ICP above which treatment is indicated for ICH has not been defined in dogs and cats. It seems reasonable to utilize the human guidelines for ICH until species-specific information is available.
Homeostatic Responses of the Brain
Volume Buffering
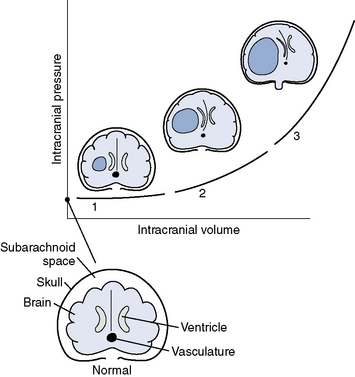
The brain is relatively noncompressible and is encased in bone, causing the volume of the intracranial contents to be fixed. Increase in the volume of one component requires a compensatory decrease in one or more of the others if ICP is to remain unchanged (Monroe-Kellie doctrine). Sources of added volume include hemorrhage, CSF accumulation, vascular congestion, cerebral edema, and decreases in venous outflow (Figure 100-1). Immediate volume buffering responses, specifically displacement of blood and CSF extracranially, are reflected by the pressure-volume curve that relates the temporal change in ICP to expanding intracranial volume.
Autoregulatory Mechanisms
Pressure Autoregulation
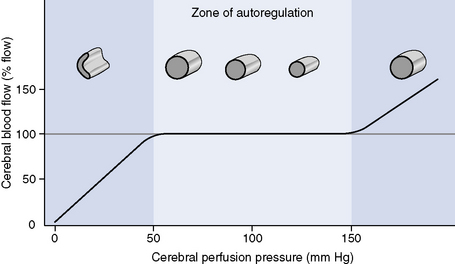
Autoregulation of CBF results from a vascular (myogenic) reflex that changes resistance of cerebral arterioles in response to changes in transmural pressure. The purpose is to prevent underperfusion or overperfusion of the brain. Normally this mechanism operates at perfusion pressures between 50 and 150 mm Hg. Outside this range, CBF becomes linear with MAP (Figure 100-2).1,4,5