1 How to obtain the perfect biopsy
Obtaining a diagnosis is one of the most important steps in the management of the cancer patient. Obtaining a biopsy before the surgical procedure is performed is best clinical practice in the majority of cases as it provides a pre-treatment diagnosis, helps the clinician plan the surgery and can provide the owner with a more accurate prognosis. There are a number of methods of obtaining samples from the tumour and the choice is based on a number of factors including:
With the exception of diagnostic cytology, all other techniques listed in Table 1.1 involve tissue sampling and histological interpretation.
CLINICAL CASE EXAMPLE 1.1 – CYTOLOGY
Clinical history
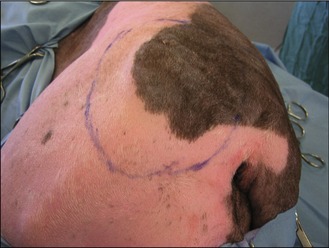
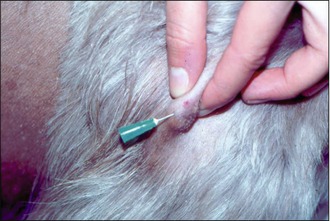
Four months previously, the dog had presented with a 2-cm mass located in the region of the left hip. The veterinary surgeon had excised the mass; however, the owners declined histopathology so the diagnosis was not determined. The mass gradually recurred and the dog was referred for further evaluation and treatment (Fig. 1.1).
Diagnostic options
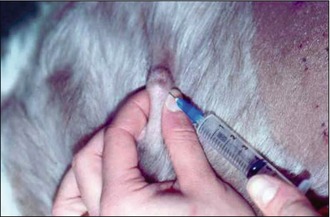
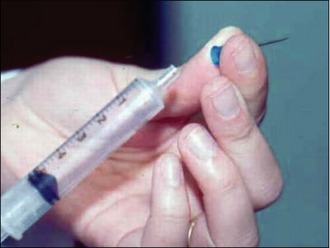
To perform a fine needle aspirate the only tools required are:
The smears should be air dried and the sample should be thin enough to dry within 1 minute. The best cytology stains to use in clinical practice are the Romanowsky stains (such as Wright’s stain, Giemsa stain and May-Grünwald-Giemsa stain) as these provide clear detail of both the nuclear and cytoplasmic structures. They are relatively quick to prepare and these stains will also stain bacteria if they are present. The ‘rapid’ stain kits such as ‘Diff-Quik’ are highly useful and obviously extremely convenient but it is important to realize that these may not always stain the granules within mast cells clearly, thereby leading to potential confusion in the diagnosis. In such a situation, Toludine blue stain will identify mast cell granules. The use of Toludine blue may be especially useful in poorly differentiated mast cell tumours in which the granularity can be low.
Diagnosis
The specimen obtained in this case was of diagnostic quality and a diagnosis of a mast cell tumour (Fig. 1.8) was made.
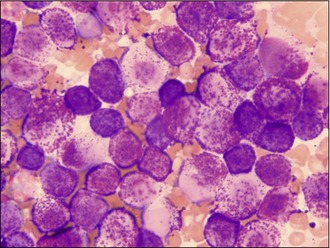
Figure 1.8 Case 1.1 Mast cell tumour aspirate, revealing the classical ‘granular’ appearance of the cytoplasm
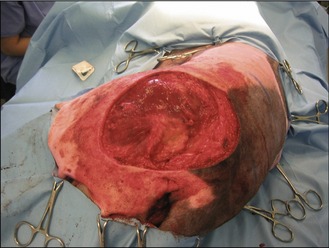
For this case a fine needle aspirate provided an accurate diagnosis although providing a definitive grade for this tumour type is not possible by cytology alone as explained above. However, by submitting the entire tumour after excision, a tumour grade can be assigned and the completeness of surgical excision evaluated. In this and similar cases, knowledge of the tumour type preoperatively allows careful surgical planning. Additionally, appropriate staging of the disease can be performed by means of thoracic radiographs, local lymph node assessment and hepatic and splenic ultrasound examination. The mass was excised with 2-cm lateral margins and one fascial plane deep and the wound deficit was repaired using a local transposition flap (Figs 1.9 and 1.10).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree