CHAPTER 20 Herbs and Nutraceuticals
Herbal medicine, also called phytomedicine, is the use of therapeutic plants, plant parts, or plant-derived substances to aid in fighting against infections and diseases or enhancing overall health. In the United States, the herbal market exceeds $3.2 billion in sales, and 32% to 37% of Americans use herbal agents each year. This percentage is thought to be much higher in Europe, where herbal agents are more widely accepted by medical professionals. In 2005 the United States herbal market value was thought to be higher than in previous years, with garlic and echinacea being the two top-selling herbs. A United States Department of Agriculture (USDA) study revealed that approximately 70% of U.S. horse operations fed some type of supplement and that nearly 5% of those operations fed herbal supplements.
HERBAL ACTIONS AND USES
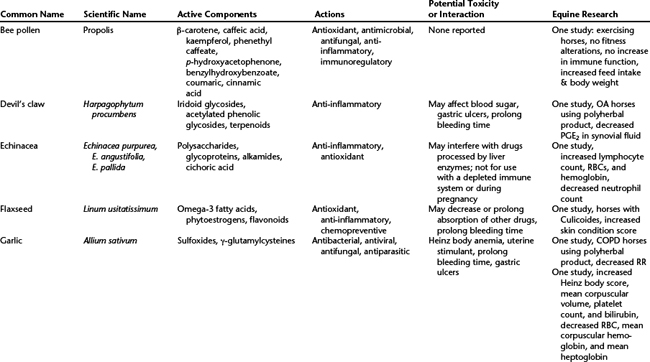
Herbal supplements that affect the immune system can be classified as adaptogens, immunostimulants, or both. Adaptogens increase resistance to physical, chemical, or biological stressors, whereas immunostimulants activate nonspecific or innate defense mechanisms against viral, bacterial, or cellular infections. Most of the studies to date in laboratory animals, humans, and other species have determined that herbal supplements do not enhance the normal immune response but may help if the immune system is compromised. Many of these effects of herbal supplements and nutraceuticals also depend on the source and processing of the supplement. Various products are available as extracts, powders, and teas, and all have different efficacies. Efficacy also varies with the portion of the plant that is used, whether it is bark, leaves, berries, or roots. The active components, actions, drug interactions, and equine research information pertaining to some of the herbs described later have been summarized (Table 20-1).
HERB-DRUG INTERACTIONS
Valerian components prolong the action of barbiturates in laboratory animal species and can interact with alcohol. They also inhibit cytochrome P450, the body’s major detoxification enzyme, which can lead to multiple drug interactions if not used with caution. These actions were enough to prompt USEF and the FEI to ban valerian-containing products. Persistent use of echinacea causes hepatotoxic effects in species other than horses, and the compound should not be administered concurrently with other drugs. Garlic can cause Heinz body anemia in horses and also causes gastrointestinal upset, allergic reactions, and dermatitis in humans. Garlic also decreases systolic and diastolic blood pressure; however, there is insufficient evidence to recommend its use for treatment of hypertension in humans. Ginger inhibits thromboxane synthetase and increases bleeding time in laboratory animals, which could be detrimental if used with anticlotting drugs like warfarin. Some of ginseng’s adverse effects in humans include hypertension, insomnia, vomiting, headache, nervousness, sleeplessness, and epistaxis. When ginseng is administered, it is advisable to discontinue use of warfarin, heparin, aspirin, and other nonsteroidal anti-inflammatory drugs (NSAIDs).