CHAPTER 65 Equine Multinodular Pulmonary Fibrosis
Interstitial lung diseases (ILDs) in the horse are poorly understood and are often diagnostically and therapeutically problematic for the clinician. For decades a subset of ILD has been recognized in equine medicine that is characterized by substantial accumulations of collagen within the lung.
SIGNALMENT AND CLINICAL FINDINGS
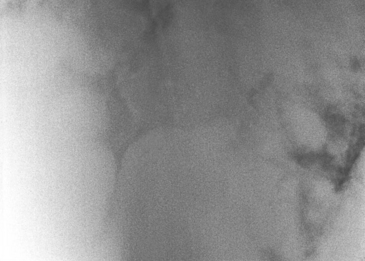
The most prominent clinical diagnostic feature of this disease is its striking radiographic appearance (Figure 65-1). The radiographic lesion is a diffuse bronchointerstitial pattern with multiple coalescing circular nodules throughout the lung field. These nodules may be so numerous that radiographically it appears as if little normal lung remains. In some cases, radiographic studies obtained 1 to 3 weeks apart reveal profound progression of the disease. The radiographic appearance of EMPF may be similar to that of the lungs in horses with fungal pneumonia or neoplasia. Radiographic findings can be in stark contrast to the overall physical appearance of the horse, depending on the chronicity of the condition. Many horses are generally in good body condition, and owners report that they have been riding these horses with little noticeable change in exercise tolerance. However, it is not infrequent for horses to be in poor body condition at initial definitive diagnosis, particularly if treatment for other respiratory conditions—such as heaves or pleuropneumonia—has been undertaken prior to diagnosis. Ultrasound examination of the thorax reveals multiple circular hypoechoic masses involving both the periphery of the lung and the pulmonary parenchyma. The masses are bilateral and are distributed throughout the lung field. Pleural effusion is generally not present.
PATHOLOGY
The gross pathologic changes associated with EMPF are recognizable and striking; gross lesions are restricted to the lower portion of respiratory tract and usually involve all lung lobes. There are two major patterns of nodular fibrosis in the lungs of EMPF horses. The most commonly reported form consists of nearly diffuse, coalescing nodules of fibrosis (Figure 65-2
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree