Chapter 10 Diseases of the Ocular Posterior Segment
Evaluation of the posterior segment of the equine eye is an essential part of an ophthalmic, prepurchase, and general physical examination. Initial examination includes a history with respect to vision, determination of menace response and pupillary light reflex (PLR), and evaluation of clarity of transmitting media (cornea, aqueous, lens, vitreous). The resting pupil size and direct and consensual PLRs are evaluated. Menace response should be evaluated in various visual fields: anterior, central, and posterior. Although it is possible to examine portions of the fundus without dilation, for proper examination of the posterior segment of the equine eye, mydriasis, a darkened environment, and proper equipment are essential. Diagnostic mydriasis is achieved by using topical 1% tropicamide (Mydriacyl), which has an onset of action of 15 to 25 minutes and a duration of action of 8 to 12 hours. As a solution, 0.2 mL of tropicamide is easily sprayed on the cornea with a tuberculin syringe and the hub of a 25-gauge needle. If required, sedation and nerve blocks can be performed to facilitate examination. Blocking the auriculopalpebral nerve will facilitate examination by allowing the clinician to elevate the superior eyelid (see Chapter 1).
Examination of the Ocular Fundus
Direct and Indirect Ophthalmoscopy
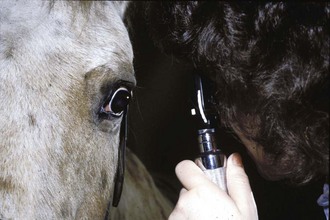
Both direct and indirect ophthalmoscopy should be used for examination of the equine posterior segment. Indirect examination is performed initially for an overview of the fundus, and direct ophthalmoscopy is preferred for detailed examination of the optic nerve and retinal blood vessels. Indirect examination with a 20-diopter (D) condensing lens and a Finnoff transilluminator will provide a more panoramic but less magnified view of the fundus, with lateral and axial magnifications of ×0.79 and ×0.84, respectively.1 Indirect examination provides better visualization of the peripheral retina and can also be performed by using an indirect headset that provides stereopsis and leaves the examiner a free hand to elevate the superior eyelid (Figs. 10-1 to 10-3). Indirect examination allows visualization of the fundus through cloudy transmitting media such as aqueous flare or lenticular sclerosis, which is often not possible with direct examination. A virtual, upside down, reversed image is created. If a headset is used, it will provide the examiner with illumination and stereopsis. Alternately, a device called a PanOptic ophthalmoscope (Welch Allyn, Skaneateles Falls, NY) can be used. The PanOptic is an indirect monocular ophthalmoscope that provides an erect image with a wider field of view but less magnification than that provided by direct examination (Fig. 10-4) (for more information, see Chapter 1).

Figure 10-4 Examination of the posterior segment, using the Welch-Allyn PanOptic monocular indirect ophthalmoscope.
(Photograph courtesy Dr. Dennis Brooks, University of Florida.)
Direct examination provides the greatest magnification, with lateral and axial magnifications of ×7.9 and ×8.4, respectively (Fig. 10-5).1 Direct ophthalmoscopy should be used for detailed examination of the optic nerve, retinal blood vessels, and peripapillary region. For examination of the posterior segment, the direct ophthalmoscope is set to 0 D, and the ophthalmoscope is held approximately 2 cm from the horse’s eye (Fig. 10-6). The optic nerve should be in focus for most examiners at this setting. Changing the diopter settings will allow the clinician to focus vitread or more posterior to determine whether an abnormality is elevated or depressed, respectively. The image obtained with a direct ophthalmoscope is upright and magnified (Fig. 10-7). Abnormalities of the cornea, aqueous, lens, and vitreous make direct examination difficult. In such instances, indirect examination should be performed.
Biomicroscopy
Biomicroscopy should also be used for examination of both the anterior segment and the anterior third of the vitreous for vitreous syneresis, asteroid hyalosis, debris, membranes, vascular remnants, and hyalitis (see Chapter 1 for more information). A yellow-green discoloration of the vitreous may be observed and is indicative of serum staining and previous intraocular inflammation.
Posterior segment examination should include evaluation of the vitreous, tapetal and nontapetal fundus, retina, retinal blood vessels, and optic nerve. Variations in the normal equine fundus are common, and familiarity with these variations is essential.2–9 Additional posterior segment diagnostic tests may include ocular ultrasonography,10 fluorescein angiography,11 computed tomography (CT), electroretinography (ERG),12 and visual evoked potential (VEP) (see Chapter 1 for more information).
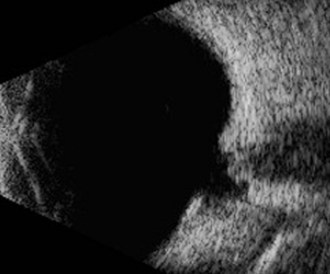
Ultrasonography
Ultrasonography is indicated when opacities of the transmitting media prevent examination of the posterior segment. Typically, cataracts and hyphema are the most common indications for ultrasound examination of the posterior aspect of the globe.10 A 5- to 20-MHz ultrasound probe will provide diagnostic images of the posterior globe. The 5-MHz probe provides less near-field resolution and greater penetration of the orbit, whereas the 20-MHz probe provides excellent resolution of the anterior segment, but the posterior eye wall is the limit of its penetration in the equine eye. Direct corneal contact with the use of methylcellulose gel is the technique of choice, provided the cornea is intact. Imaging can be performed through the eyelid if the cornea is compromised (see Chapter 1 for more information).
Electroretinography
While the ERG has traditionally been performed using general anesthesia, a technique for obtaining a flash photopic and scotopic ERG in a standing sedated horse has been described, making this test more easily obtained and associated with less risk and expense.12 An ERG is indicated to evaluate retinal function prior to cataract surgery and to assess retinal health and aid in diagnosis in congenital stationary night blindness, glaucoma, equine motor neuron disease, chorioretinitis, optic neuritis, and ocular trauma.
Fluorescein Angiography
The technique and results of fluorescein angiography have been described in the normal horse.11 Two successive angiographic phases were described: the choriocapillary phase, starting at 46.95 ± 9.48 seconds, and the retinal vascular phase, starting at 47.79 ± 10.38 seconds. The retinal vascular phase was divided in three parts: filling phase, maximum fluorescence point, and fading phase. During the filling phase, the dye progressed into the retinal vessels, obtaining maximum fluorescence at 59.79 ± 10.39 seconds, termed the maximum fluorescence point. The fading phase started immediately following the maximum fluorescence point. During this phase, vascular fluorescence decreased to complete reduction at 74.76 ± 9.81 seconds. Areas of delayed choroidal filling, the presence of short retinal vessels in the ventral region of the optic disc, and a particular filling of the optic disc were also observed.11 Fluorescein angiography may have value in evaluating diseases of the retina, choroid, retinal pigment epithelium, and optic nerve (see Chapter 1 for more information).
Clinical Anatomy and Physiology
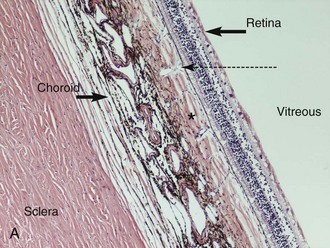
The posterior segment of the eye includes the vitreous, retina, choroid, sclera, and optic nerve (Fig. 10-8). The anterior-most extent of the posterior segment is the ora ciliaris retinae where the retina attaches to the pars plana of the ciliary body. The anterior extension of the ora ciliaris varies by quadrant of the eye and is important because it has significance for glaucoma and vitreoretinal surgery.14 The ora is located more anteriorly in the nasal and ventral quadrants and further posterior in the dorsal and temporal quadrants.14 This makes the dorsal and temporal locations the areas of choice for surgical approaches to the posterior segment and for transscleral and endocyclophotocoagulation.
The vitreous body is predominately composed of type II collagen fibrils, hyaluronic acid, and water. It is divided into cortical, intermediate, and central zones and has a volume of approximately 28 mL in the adult horse.15,16 Transrectal ultrasound of the dimensions of the fetal vitreous body has been used to predict parturition date in light horse mares and ponies.17 Normally the vitreous is transparent and uniform in appearance. Abnormalities of the vitreous generally result in loss of uniformity, with debris, membranes, and collagen fibrils appearing to float and swirl in a liquid medium. In addition, changes in vitreous color are often noted as a result of inflammation with the vitreous and the lens appearing yellow-green in color as a result of chronic inflammation. On ultrasonography, the vitreous is normally anechoic with increased vitreous echogenicity, indicating degeneration, inflammation, hemorrhage, mass lesions, or retinal detachment. This is evidence that the vitreous may have a role as an effector in some instances of equine recurrent uveitis (ERU), and pars plana vitrectomy has been advocated to control recurrent inflammation in ERU.18
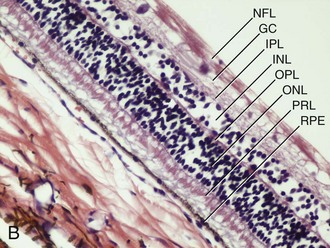
The horse has a paurangiotic (partially vascularized) retina with 30 to 60 small retinal vessels radiating from the margin of an elliptical optic disc. These vessels are visible for a distance of 1 to 2 disc diameters but are less prominent dorsal and ventral to the optic nerve (Fig. 10-9). The remainder of the equine retina is avascular, being supplied from the underlying choroid. The thickness of the NSR varies from 250 mm medial to the optic disc to 80 mm at the ora ciliaris.19 The majority of the equine retina is less than 130 mm in thickness to allow for oxygen diffusion from the underlying choroid.19 The choroidal or vascular tunic is external to the NSR. It is composed of the larger choroidal vessels, the tapetum, choriocapillaris, and melanocytes. In the equine eye, the choroid is responsible for the majority of retinal nutrition because the retina is paurangiotic.
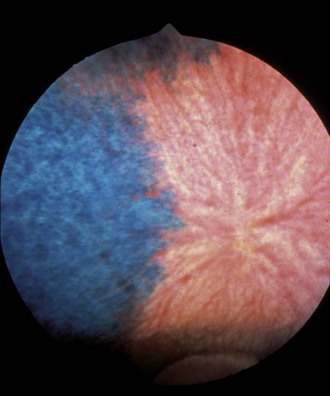
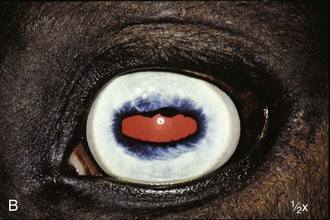
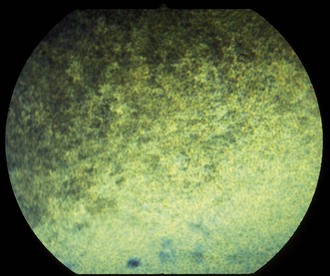
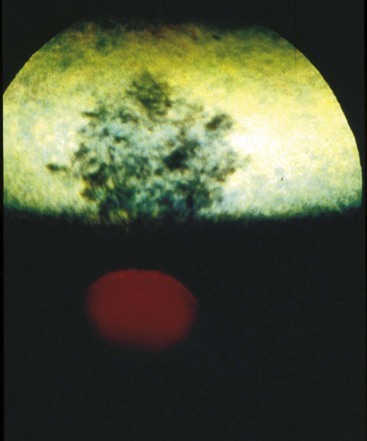
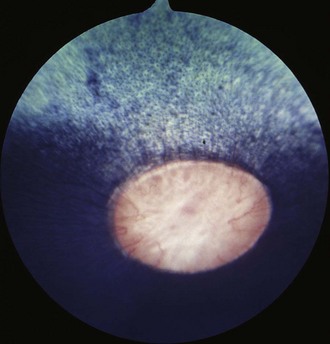
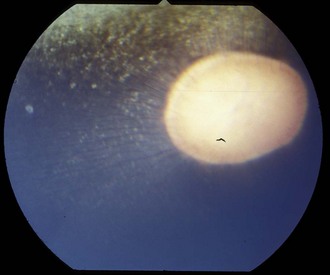
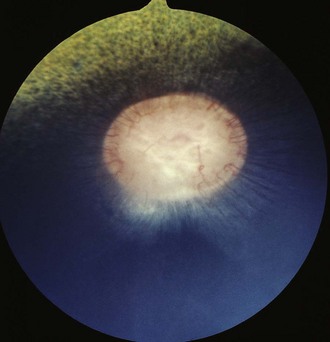
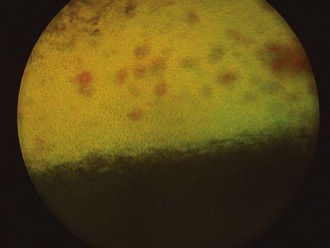
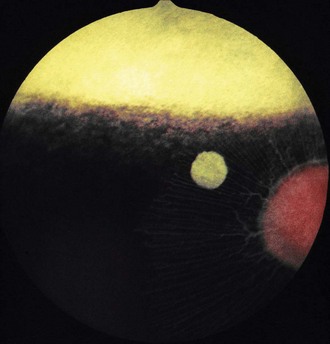
The fundus is divided into the dorsal tapetal and ventral nontapetal regions. The fibrous tapetum is situated in the dorsal choroid and is responsible for the characteristic yellow-green color of this portion of the fundus. Variations in tapetal color are related to iris and coat color and include yellow, orange, and blue-green.3,6,8 Dark bay and brown horses usually have a blue-green tapetum with a dark nontapetal region (Figs. 10-10 and 10-11). Lighter chestnuts and palominos may have a yellow tapetum with a less pigmented nontapetal region (Figs. 10-12 and 10-13). Gray and white horses generally have a yellow tapetum with a lightly pigmented or nonpigmented nontapetal fundus. This may allow visualization of the choroidal vasculature and sclera. Finally, in color-dilute horses, the tapetum may be hypoplastic or absent.21–22 Tapetal hypoplasia can be generalized or confined to a portion of the tapetum, most often immediately dorsal to the optic disc (Figs. 10-14 to 10-16). Color and thickness of the tapetum and choroidal pigment vary not only between horses but also within horses from right to left eyes. Near the periphery of the tapetum, it is not unusual to see focal areas of RPE pigmentation obscuring the underlying tapetum (Fig. 10-17). In addition, random foci of RPE pigmented freckles or nevi may appear throughout the tapetum (Fig. 10-18). These must be differentiated from postinflammatory hyperpigmentation or choroidal melanoma. The tapetum of the horse is penetrated by small choroidal arterioles that serve to supply the retina via the choriocapillaris. These vessels are seen end-on on fundic examination and appear as uniformly distributed dark dots in the tapetum called the stars of Winslow (Fig. 10-19). The ultrastructure of the equine RPE and choriocapillaris have been described.23 The function of the tapetum is to enhance vision in low-light conditions by allowing reflected light the opportunity to stimulate the photoreceptors a second time. However, although the tapetum improves the threshold of light detection, it also causes glare and thereby decreases visual acuity in bright light.20
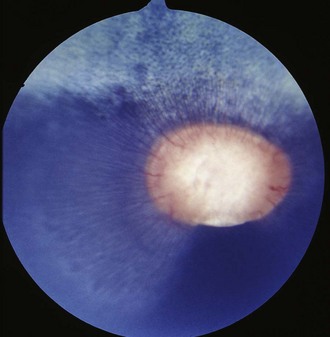
Figure 10-10 Normal fundic photograph of a dark bay horse with a blue tapetum.
(Photograph courtesy Drs. Ingo Walde and Barbara Nell, University of Vienna.)
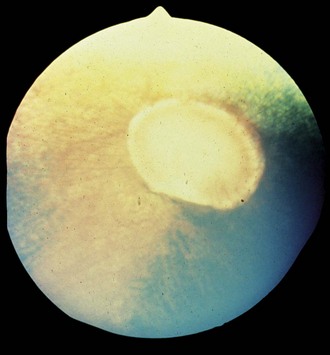
Figure 10-13 Normal fundus of a palomino horse, demonstrating a yellow tapetum and area of choroidal hypopigmentation.
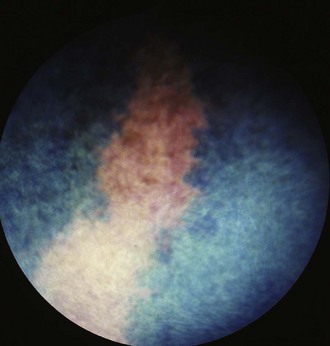
Figure 10-16 Normal fundic photograph with a linear area of tapetal hypoplasia.
(Photograph courtesy Drs. Ingo Walde and Barbara Nell, University of Vienna.)
The ventral nontapetal fundus is generally dark brown or black but can appear lighter or nonpigmented, depending on coat color.6,8 In color-dilute horses, the tapetum may be hypoplastic or absent, and the choroid and RPE may demonstrate partial or complete albinism. This can affect one or both eyes and can even be observed to vary within an eye. Choroidal vasculature and vortex veins may be visible, especially in association with tapetal hypoplasia and ocular albinism (Figs. 10-20 and 10-21).3 These vessels may appear as radiating, dark striae seen through a normal tapetum or as red vessels in a subalbinotic to albinotic eye.
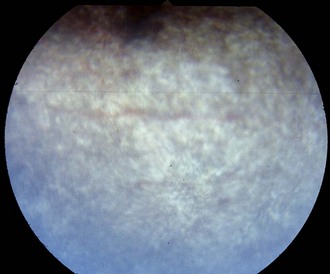
Figure 10-20 Normal nontapetal fundus of a color-dilute horse. The underlying choroidal vasculature can be seen.
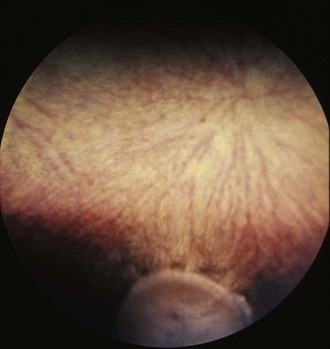
Figure 10-21 Normal fundic photograph of a color-dilute horse with tapetal hypoplasia.
(Photograph courtesy Drs. Ingo Walde and Barbara Nell, University of Vienna.)
Focal areas of nonpigmentation or depigmentation are often observed in the peripapillary nontapetal fundus (Fig. 10-22). These are often normal variations, but because previous intraocular inflammation can also result in depigmentation, a complete ophthalmic examination should be performed to rule out prior disease. In addition, infection with equine herpesvirus 1 (EHV-1) has been reported to result in focal white chorioretinal lesions in the nontapetal fundus (see discussion of chorioretinitis).24
The retina consists of the NSR and RPE. The photoreceptors, rods and cones, are located in the sensory retina. The rods predominate and are responsible for achromatic low-light vision, whereas the cones are responsible for vision in bright light and color vision. The horse lacks a fovea but has an area centralis, which serves as the area of maximum visual acuity and has an increased density of retinal ganglion cells. The equine area centralis is a horizontal band of 1 mm by 22 mm and is located approximately 3 mm dorsotemporal to the optic nerve.25,26 The concentration of ganglion cells in the area centralis is 4000 to 6000 cells/mm2 as compared with less than 500 cells/mm2 in the dorsal and ventral retina.27,28 In addition to regional differences in ganglion cell density, it has been shown that variation in ganglion cell density within the area centralis also occurs by breed of horse and skull conformation.29 Evaluation of the equine inner retina has demonstrated the presence of bipolar, amacrine, horizontal, and Müller cells in percentages of 44%, 24%, 1%, and 29%, respectively.30 Specific markers for these and other retinal cells have been identified for the equine retina.31 The horse has dichromatic vision, with a short-wavelength cone (spectral peak of 425 nm) and a middle-wavelength cone (spectral peak of 540 nm).32,33 This translates into a blue-gray and yellow-green visual spectrum. The horse has been shown to be able to discriminate color from grays and to demonstrate color-discrimination performance behavior.34 The visual field of the horse is approximately 350 degrees, with a small area—65 to 70 degrees—of binocular overlap.5 Please see Chapter 11 for more information on equine vision.
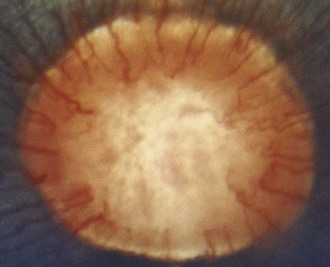
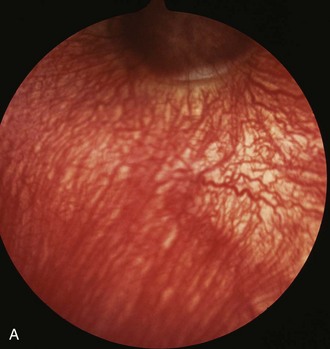
The optic disc is normally oval, salmon pink, and situated slightly temporal and ventral in the nontapetal fundus. The optic nerve will fill the view of a direct ophthalmoscope; it measures 3 to 5 mm vertically and has a horizontal diameter of 5 to 8 mm.20 The optic nerve in foals may appear more round. The surface of the optic nerve is often irregular and honeycombed as a result of the axons exiting at the fibrous lamina cribrosa. Ventrally, the optic nerve may appear notched (see Fig. 10-9). The equine optic nerve is composed of approximately 1 million axons, with larger axons found at the periphery of the optic nerve as compared with the central portion.26,35 These large axons are preferentially lost in equine glaucoma.35 Although the optic nerve is myelinated, myelin does not typically extend to the intraocular portion of the nerve. If myelin does extend into the eye, it is most often associated with retinal vessels. Myelin extending along the nerve fiber layer axons appears as light gray striations and is more common in aged horses (Fig. 10-23). Radiating from the margin of the optic nerve are 30 to 60 small arterioles and venules. They cannot be distinguished from one another and may be absent at the 6 o’clock region of the nerve. These retinal vessels travel 1 to 2 optic disc diameters into the retina before they disappear. Occasionally, a small vessel may be seen to emerge from the central optic nerve and course across its surface. The margin of the optic nerve may have a peripapillary ring associated with changes in thickness of the tapetum or pigmentation of the RPE or choroid. This circumpapillary ring can be dark or hypopigmented. This is a normal variation, provided that the overlying retinal vessels do not deviate or attenuate while crossing this region. Changes in vessel direction or thickness suggest a coloboma or degeneration. The axons of the retinal ganglion cells exit the eye through the scleral lamina cribrosa. The adult equine lamina cribrosa is a complex, multilayered structure composed of myelinated optic nerve axons, capillaries, astrocytes, and an extracellular matrix.36 The immunohistochemistry of the extracellular matrix of the normal equine lamina cribrosa has been described. It has been suggested that this macromolecular structure of the equine lamina cribrosa may make it resistant to changes associated with elevations of intraocular pressure and offer protection to the optic nerve axons.36 This is supported clinically by the preservation of vision in horses despite often chronic elevations in intraocular pressure.
Impact of Disease of the Ocular Posterior Segment on the Equine Industry
The impact of posterior segment disease is significantly greater than has been appreciated, and veterinarians are strongly encouraged to make examination of the posterior segment a routine part of a physical and prepurchase examination. In a recent survey of 204 racing Thoroughbred horses in Australia, ocular abnormalities were observed in 138 (67.6%) of the horses examined. Of these findings, potential vision-threatening abnormalities were noted in 15 horses (7.4%) and non–vision-threatening abnormalities in 117 (57.4%). The most frequent abnormalities involved the retina and lens, with abnormalities of the retina noted in 117 (57.4%) of the horses examined.13 If a complete examination of the posterior segment is required, the pupil should be pharmacologically dilated. It should be noted on a prepurchase examination whether the pupil was or was not dilated for the ophthalmic examination.
Diseases of the Ocular Fundus
Congenital Diseases
Congenital abnormalities of the equine posterior segment are uncommon. They can be seen alone or in association with multiple congenital anomalies such as anterior segment dysgenesis, congenital megaloglobus, the multiple congenital ocular anomalies (MCOA) syndrome (i.e., the Rocky Mountain horse [RMH] syndrome), and other such ocular anomalies. In one study, the incidence of congenital ocular abnormalities was 0.5% of all equine cases presented.37
Retinal Hemorrhages
Subretinal and intraretinal hemorrhages may be noted in neonatal foals (Fig. 10-24).38 In one study of 167 neonatal Thoroughbred foals, 27 (16%) were found to have retinal hemorrhages.38 Most hemorrhages were bilateral, and females were more commonly affected than males.38 Hemorrhages were punctate in 36% and “splash-like” in 56%; the number of hemorrhages per eye ranged from 1 to 20.38 Such hemorrhages may be the result of intraocular vascular hypertension with capillary rupture during parturition.20 Hemorrhages will resolve over a few weeks with no visual or residual abnormalities.38
Colobomas
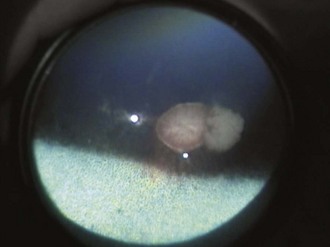
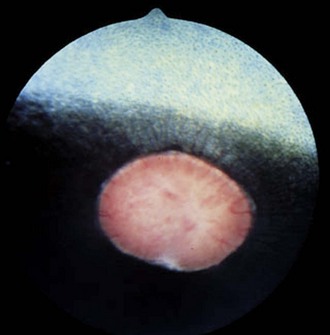
Colobomas are absences of or defects in normal ocular tissue and typically occur in the area of the embryonic optic fissure, which is ventral to slightly ventronasal; if colobomas are noted elsewhere, they are considered atypical. Colobomas can be unilateral or bilateral and can affect the iris, ciliary body, lens, retina, choroid, optic disc, and sclera.3,21,22,39–42 They can occur alone or in association with multiple congenital ocular anomalies. One report of a Thoroughbred with bilateral optic disc colobomas also included mention of microphthalmos, microcornea, and iris and lens abnormalities.43 In a survey of 204 Thoroughbred racehorses in Australia, optic nerve coloboma and “unusual optic nerve head anatomy” was described in 2 horses.13 Bilateral colobomas involving the iris, ciliary body, lens, choroid, retina, and optic nerve have been described clinically and using fluorangiography in a donkey.42 Posterior segment colobomas have been found to affect the optic disc in horses, most commonly the Quarter Horse.39–41 Others report a predisposition in the Appaloosa.20 Optic nerve colobomas accounted for 1.8% of all congenital ocular anomalies in one study.37 Small colobomas must be distinguished from the small 6 o’clock notch and peripapillary ring that can be normal in some horse optic nerves (see Fig. 10-9).20 Severe colobomas involving the optic disc have been associated with vision deficits, specifically in the Quarter Horse (Fig. 10-25).39–41
Clinical Appearance and Diagnosis
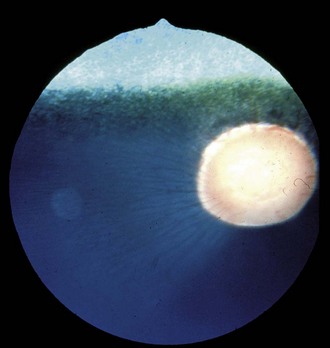
Additionally, a “window defect” involving the RPE has been described as an RPE coloboma (Figs. 10-26 and 10-27).3,20 This abnormality occurs most commonly in the nontapetal fundus adjacent to the optic disc and appears as a sharply demarcated focal area of decreased pigmentation. Such an abnormality has no known visual or genetic significance.
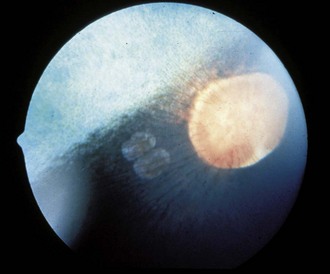
Figure 10-26 Two “window defects,” or retinal pigment epithelium colobomas, are noted adjacent to the optic nerve.
Optic Nerve Hypoplasia
Optic nerve hypoplasia can be unilateral or bilateral and is a rare congenital abnormality.44 Optic nerve hypoplasia accounted for 0.9% of all congenital ocular anomalies in one study.37
Clinical Appearance and Diagnosis
In optic nerve hypoplasia, the optic nerve appears pale, is smaller than normal, and may be depressed or recessed; retinal vessels are difficult to visualize.20,45 Numbers of retinal ganglion cells are reduced, the pupil is dilated, and the pupillary light response is reduced or absent.20 Nystagmus and decreased to absent vision are also observed.
Retinal Dysplasia
Retinal dysplasia is a congenital malformation of the retina, with the formation of folds or rosettes that appear clinically as retinal folds or larger areas of geographic dysplasia. Retinal dysplasia can be developmental or occur as a result of inflammation. Retinal dysplasia accounted for 3.5% of all congenital ocular anomalies in one study.37
Clinical Appearance and Diagnosis
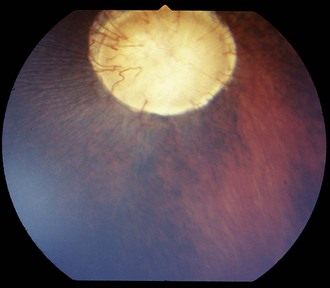
On ophthalmoscopic examination, dysplasia can appear as gray or hyperpigmented areas caused by retinal disorganization and RPE proliferation, respectively (Fig. 10-28). Histologically, the NSR is folded and disorganized, often forming rosettes (Fig. 10-29). The rosettes can be single or multiple and unilateral or bilateral. Retinal folds can be associated with in utero infections and have been attributed to various viral infections. In other species, they are also seen in association with multiple congenital ocular anomalies, which are inherited. One example of multiple congenital anomalies with associated retinal dysplasia in the horse is the MCOA syndrome.46 In addition, congenital megaloglobus has been seen with associated severe retinal dysplasia. The retinal dysplasia in one case was associated with a cartilaginous choristoma of the choroid (D. Wilkie, unpublished data) (Figs. 10-30
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree