Chapter 1 Diseases of the Cardiovascular System
The cardiovascular system plays an important role in maintaining homeostasis throughout the body. It performs this function by regulating the flow of blood through miles of vessels and capillaries. It is in the capillaries that vital nutrients are transported into the body cells and removal of waste materials from the cells occurs.
ANATOMY AND PHYSIOLOGY
The Pump
At the center of the cardiovascular system is the heart, a four-chambered pump designed to contract, pumping blood to all parts of the body. Specialized cardiac muscle cells located in the sinoatrial (SA) node generate electrical impulses that spread, through a specialized conduction system, resulting in simultaneous contraction of the cardiac muscle cells. This contraction pushes blood into the arterial vessels and returns blood to the heart from the veins. This electrical activity can be measured as it moves across the surface of the body, using an electrocardiograph (Fig. 1-1). The electrocardiographic instrument measures the electrical activity generated by the heart by the placement of electrodes at specific points on the body surface. Each mechanical contraction of the heart is preceded by an electrical wave front that stimulates heart muscle contraction. This electrical wave front begins at the SA node and travels to the muscle cells of the ventricle through the cardiac conduction system. These wave fronts are recorded as the electrocardiogram (ECG). Figure 1-2 shows a normal ECG of a dog. Figure 1-3 represents the normal pathway for electrical conduction through the heart.
Figure 1-1 Using an electrocardiograph.
(From Edwards NJ: Bolton’s handbook of canine and feline electrocardiography, ed 2, Philadelphia, 1987, WB Saunders, by permission.)
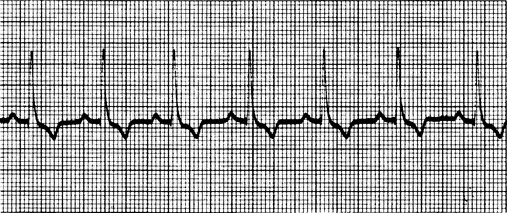
Figure 1-2 Normal electrocardiogram of a dog.
(From Edwards NJ: ECG manual for the veterinary technician, Philadelphia, 1993, WB Saunders, by permission.)
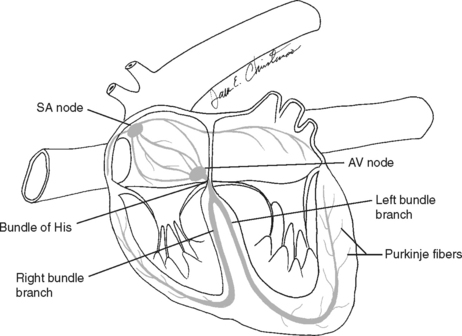
Figure 1-3 Normal pathway for electrical conduction through the heart. AV, atrioventricular; SA, sinoatrial.
(From McBride DF: Learning veterinary terminology, ed 2, St Louis, 2002, Mosby, by permission.)
Many cardiac diseases involve a failure of this pump to function properly. Congestive heart failure, cardiomyopathy, valvular disease, and congenital malformations can all affect the pumping efficiency of the heart, and ultimately the function of the entire body.
The Vessels
Connected to the pump are a series of vessels. Arteries carry oxygenated blood at high pressure (the systolic blood pressure) to arterioles and onto capillaries where exchange of nutrients and gases occurs. Blood then moves into venules, through veins, and is returned to the right side of the heart. Arteries are capable of dilation and constriction, routing blood to areas where it is needed and away from those areas not in need. Constriction serves to increase blood pressure, and dilation serves to decrease it.
CARDIOMYOPATHIES
Canine Dilated Cardiomyopathy
TREATMENT
Feline Dilated Cardiomyopathy
Before the late 1980s, feline DCM was one of the most frequent cardiac diseases reported in cats. After the association of the disease with taurine deficiency, additional taurine was added to commercial diets, and the incidence of the disease significantly decreased. The pathologic condition is similar to DCM in dogs. Evidence has been found of a genetic predisposition to DCM in cats fed taurine-deficient diets.
TREATMENT
Feline Hypertrophic Cardiomyopathy
TREATMENT
Thromboembolism
Thrombus formation is a common and serious complication of myocardial disease in the cat. It is estimated that between 10% and 20% of cats with HCM will experience development of thrombi in the left side of the heart, which may dislodge and become trapped elsewhere in the arterial system. Cats appear to have inherently high platelet reactivity, making clot formation a more likely sequel to endothelial damage and sluggish blood flow occurring with myocardial disease. Approximately 90% of these emboli lodge as “saddle thrombi” in the distal aortic trifurcation, resulting in hind-limb pain and paresis. Rarely will a thrombus lodge at other arterial sites such as the renal artery, the coronary arteries, the cerebral arteries, or the mesenteric artery.
TREATMENT
CONGENITAL HEART DISEASE
While malformations of the heart and great vessels represent a small cause of clinical heart disease, it is important to identify them in newly acquired pets or those to be used for breeding. Technicians should be encouraged to use their stethoscopes to routinely listen to the heart. With practice, subtle changes will become noticeable, allowing the technician to note abnormalities in the patient’s record.
Many malformations have a genetic basis. Breed predilections for congenital heart disease are listed in Table 1-1. The diagnostic approach for congenital heart disease should include a good history, with special attention paid to breed, sex, and age of the patient. Clinical signs of congenital heart failure include failure to grow, dyspnea, weakness, syncope, cyanosis, seizures, and sudden death; however, many animals with congenital malformations may be asymptomatic.
Table 1-1 Canine Breed Predilections for Congenital Heart Disease
| Breed | Defect(s) |
|---|---|
| Basset hound | PS |
| Beagle | PS |
| Bichon frise | PDA |
| Boxer | SAS, PS, ASD |
| Boykin spaniel | PS |
| Bull terrier | MVD, AS |
| Chihuahua | PDA, PS |
| Chow chow | PS, CTD |
| Cocker spaniel | PDA, PS |
| Collie | PDA |
| Doberman pinscher | ASD |
| English bulldog | PS, VSD, TOF |
| English springer spaniel | PDA, VSD |
| German shepherd | SAS, PDA, TVD, MVD |
| German shorthaired pointer | SAS |
| Golden retriever | SAS, TVD, MVD |
| Great Dane | TVD, MVD, SAS |
| Keeshond | TOF, PDA |
| Labrador retriever | TVD, PDA, PS |
| Maltese | PDA |
| Mastiff | PS, MVD |
| Newfoundland | SAS, MVD, PS |
| Pomeranian | PDA |
| Poodle | PDA |
| Rottweiler | SAS |
| Samoyed | PS, SAS, ASD |
| Schnauzer | PS |
| Shetland sheepdog | PDA |
| Terrier breeds | PS |
| Weimaraner | TVD, PPDH |
| Welsh corgi | PDA |
| West Highland white terrier | PS, VSD |
| Yorkshire terrier | PDA |
AS, Aortic stenosis; ASD, atrial septal defect; CTD, cor triatriatum dexter; MVD, mitral valve dysplasia; PDA, patent ductus arteriosus; PPDH, peritoneopericardial diaphragmatic hernia; PS, pulmonic stenosis; SAS, subaortic stenosis; TOF, tetralogy of Fallot; TVD, tricuspid valve dysplasia; VSD, ventricular septal defect.
From Oyama MA, Sisson DD, Thomas WP, Bonagura JD: Congenital heart disease. In Ettinger SJ, Feldman EC, editors: Textbook of veterinary internal medicine, ed 6, vol 2, St Louis, 2005, Saunders.
Patent Ductus Arteriosus
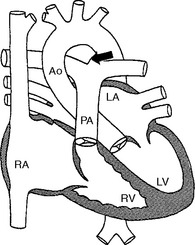
Failure of the ductus arteriosus to close after parturition results in blood shunting from the systemic circulation to the pulmonary artery. Normally the ductus carries blood from the pulmonary artery to the aorta during fetal development. The increase in oxygen tension in the blood at birth results in closure of the path in the first 12 to 14 hours of life. If the ductus remains open, blood will hyperperfuse the lung and the left side of the heart will become volume overloaded (Fig. 1-4). The resulting cardiac murmur is often referred to as a “machinery murmur”; this type of murmur is heard best over the main pulmonary artery high on the left base.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree