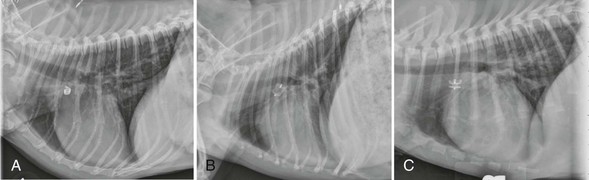
Web Chapter 64 Patent ductus arteriosus (PDA) is the persistence of a fetal communication between the descending aorta and the main pulmonary artery. Depending on the study, PDA is considered the most or second most common congenital cardiac malformation diagnosed in dogs (see Chapter 174). There is a higher incidence in females, with an odds ratio of approximately 3 : 1, although this is not evident in all breeds. Numerous breeds appear predisposed, including the bichon frise, Chihuahua, cocker spaniel, collie, English springer spaniel, German shepherd, Labrador retriever, Maltese, Pomeranian, poodle, Shetland sheepdog, Welsh corgi, and Yorkshire terrier. Without correction, the estimated 1-year mortality for PDA approaches 60%. Although subaortic stenosis is the other congenital malformation reported as the most common in many case series, many diagnosed cases of subaortic stenosis are of trivial hemodynamic significance, requiring no intervention and having little effect on an individual patient’s quality or duration of life. Therefore PDA is likely the most commonly diagnosed defect that routinely requires intervention. In a very small percentage of cases, shunt reversal (right-to-left PDA) may occur secondary to severe pulmonary hypertension, a condition that is not amenable to closure in any manner. Dogs tend to develop pulmonary hypertension early in life and relatively quickly from a large PDA, but the development of pulmonary hypertension in cats typically is more gradual. This allows some cats to benefit from closure of the ductus, despite the presence of pulmonary vascular disease. It may be possible to close a PDA successfully when there is left-to-right shunting even when pulmonary hypertension is present (Seibert et al, 2010), and this judgment is best made by considering not only pulmonary artery pressure but calculated pulmonary to systemic vascular resistance. This generally requires cardiac catheterization. A complete Doppler echocardiographic study is the test of choice for definitive diagnosis. Echocardiographic features of PDA include dilation of the left atrium, left ventricle, ascending aorta, and pulmonary artery. A portion of the PDA itself frequently can be visualized from several imaging planes, but the left cranial short-axis view usually provides the optimal image. Saunders and colleagues (2007) described the transthoracic and transesophageal echocardiographic (TEE) appearance of PDA as it relates to angiographic anatomy, demonstrating the superiority of TEE for critical visualization of ductal anatomy in the dog. Doppler echocardiography confirms continuous flow in the main pulmonary artery with reversal of flow in diastole. Mild to moderate mitral regurgitation secondary to annular dilation is a frequent finding. Depending on its severity, mitral regurgitation usually resolves following ductal ligation or occlusion. Evidence of systolic dysfunction (increased left ventricular internal diameter during systole, reduced percent fractional shortening) is common in dogs with large volumetric shunts and typically persists or appears worse following successful ligation or occlusion. Nevertheless, this finding rarely translates into long-term morbidity or mortality unless ventricular function is severely depressed or atrial fibrillation or congestive heart failure is evident. Following the initial report by Snaps and colleagues, numerous reports have detailed the use of either free-release or detachable occlusion coils. In addition, there have been two reports of the use of the human Amplatzer ductal occluder (Sisson, 2003). The high cost of this device and the requirement for transvenous delivery precludes its routine use in veterinary medicine. The Amplatzer vascular plug and now the Amplatz canine duct occluder have been evaluated in dogs (Hogan et al, 2006; Nguyenba and Tobias, 2007). Generally these are deployed using femoral artery catheterization. Most dogs can be treated successfully with one of these two devices (or with coils) provided the operator is experienced and the catheterization laboratory carries a sufficient inventory of devices. Inappropriate ductal morphology or small size most commonly precludes use of catheter-based techniques. Lower-profile devices have been developed, but the majority of centers tend to recommend surgical correction in dogs weighing less than approximately 3 kg. All catheter-delivered devices are deployed under general anesthesia using fluoroscopic guidance following angiographic evaluation of ductal morphology. Web Figure 64-1 provides lateral radiographs showing the three most commonly used occlusion devices deployed in canine clinical patients. Web Figure 64-1 Lateral radiographs of dogs with patent ductus arteriosus treated with multiple vascular occlusion coils (A), an Amplatzer vascular plug (B), and an Amplatz canine duct occluder (C).
Patent Ductus Arteriosus
Diagnosis
Treatment
Device Occlusion of Patent Ductus Arteriosus
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Chapter 64: Patent Ductus Arteriosus
Only gold members can continue reading. Log In or Register to continue
