Web Chapter 28 The basic supplies required for sample collection are listed in Web Box 28-1. Whereas 1- to
Collection of Specimens for Cytology
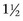 -inch needles are adequate for many clinical cases, longer spinal needles often are required for intracavitary aspirates to access deeper tissues. The recommended needle size varies with the tissue type and expected exfoliation. Large 18- to 20-gauge needles may be needed for lesions that are unlikely to exfoliate well, such as bone aspirates, whereas most lesions and organs can be aspirated adequately with 21- to 25-gauge needles. The recommended syringe size is typically between 6 and 12 ml, which provides adequate suction and also is a comfortable fit for the operator’s hands.
-inch needles are adequate for many clinical cases, longer spinal needles often are required for intracavitary aspirates to access deeper tissues. The recommended needle size varies with the tissue type and expected exfoliation. Large 18- to 20-gauge needles may be needed for lesions that are unlikely to exfoliate well, such as bone aspirates, whereas most lesions and organs can be aspirated adequately with 21- to 25-gauge needles. The recommended syringe size is typically between 6 and 12 ml, which provides adequate suction and also is a comfortable fit for the operator’s hands.
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


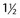 -inch needles (20- to 25-gauge needles)
-inch needles (20- to 25-gauge needles)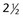 – to
– to 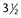 -inch spinal needle with stylet
-inch spinal needle with stylet